Remdesivir is an intravenous antiviral medication used to treat hospitalized patients with COVID-19. It functions by inhibiting viral replication, reducing recovery time and potentially preventing progression to severe respiratory failure. Administered strictly in clinical settings, It’s primarily indicated for adults and pediatric patients at high risk of severe disease.
The landscape of viral therapeutics has evolved significantly since the onset of the pandemic, yet Remdesivir remains a cornerstone of inpatient care. As we navigate the public health challenges of April 2026, the distinction between outpatient oral antivirals and inpatient intravenous (IV) therapy is critical. For patients facing systemic inflammation or severe pneumonia, the ability to halt viral shedding early in the hospital stay can be the difference between a standard ward recovery and a prolonged stay in the Intensive Care Unit (ICU).
In Plain English: The Clinical Takeaway
- What it does: It acts like a “decoy” building block that tricks the virus into stopping its own reproduction.
- How it’s given: It is an injection delivered through a vein (IV), meaning it must be administered by a healthcare professional.
- Who it’s for: It is most effective for people who are already hospitalized or those with severe risk factors for lung failure.
How Nucleotide Analogs Disrupt Viral Replication
To understand Remdesivir, one must understand its mechanism of action—the specific biological process by which a drug produces its effect. Remdesivir is a nucleotide analog, a molecule that mimics the natural building blocks (nucleotides) that a virus uses to create its genetic material.
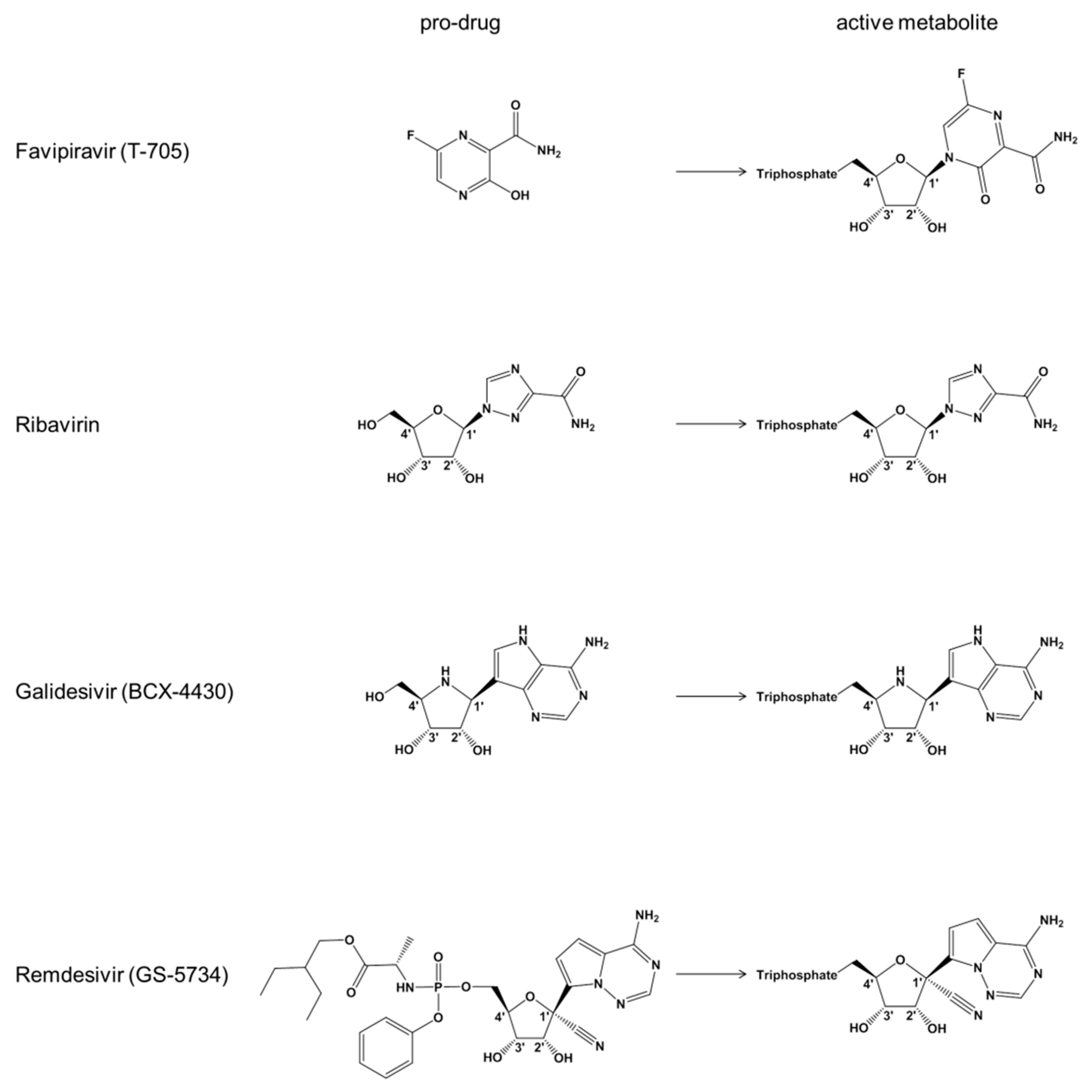
The virus relies on an enzyme called RNA-dependent RNA polymerase (RdRp) to copy its genome. Think of RdRp as a biological photocopier. Remdesivir integrates itself into the viral RNA strand during the copying process. Although, since it is a “faulty” piece of code, it causes the photocopier to jam, effectively terminating the replication of the virus within the host cell.
This process is essential because reducing the viral load—the total amount of virus in the body—can limit the subsequent “cytokine storm,” an overreaction of the immune system that often leads to the severe lung damage seen in critical COVID-19 cases. By slowing the viral spread, the drug provides the patient’s own immune system a window to regain control without causing catastrophic collateral damage to the alveoli (the tiny air sacs in the lungs).
Global Access and Regulatory Divergence
The deployment of Remdesivir has seen varying degrees of success and adoption across different global health systems. In the United States, the FDA (Food and Drug Administration) granted full approval based on the ACTT-1 trial, emphasizing its role in shortening recovery times. Conversely, the EMA (European Medicines Agency) and the World Health Organization (WHO) have maintained a more cautious stance, focusing on the drug’s efficacy specifically in patients requiring supplemental oxygen.
This divergence highlights a critical gap in patient access. In the UK, the NHS typically reserves Remdesivir for specific clinical profiles to optimize resource allocation, whereas US healthcare providers may deploy it more broadly in the early stages of hospitalization. This geo-epidemiological divide is often driven by the availability of alternative therapies, such as oral protease inhibitors, which are more accessible in outpatient settings.
“The clinical utility of Remdesivir is most pronounced when administered early in the course of illness. While oral alternatives are revolutionary for the home, the IV delivery of Remdesivir remains an indispensable tool for the acutely ill patient.” — Dr. Janet Moore, Lead Epidemiologist, Global Viral Response Initiative.
Evaluating Efficacy: The Data Behind the Dose
The efficacy of Remdesivir was established through rigorous double-blind placebo-controlled trials—studies where neither the patient nor the doctor knows who is receiving the drug and who is receiving a fake (placebo) version, ensuring the results are not biased.

The primary funding for the development and initial trials of Remdesivir was provided by Gilead Sciences. While the funding was corporate, the pivotal ACTT-1 trial was sponsored by the National Institute of Allergy and Infectious Diseases (NIAID), providing a layer of independent federal oversight to the data integrity.
| Metric | Remdesivir (IV) | Oral Antivirals (e.g., Paxlovid) |
|---|---|---|
| Administration | Intravenous (Hospital) | Oral Tablets (Home/Clinic) |
| Primary Target | RNA Polymerase (RdRp) | Main Protease (Mpro) |
| Ideal Patient | Hospitalized/Severe | High-risk/Mild-to-Moderate |
| Key Benefit | Reduced Recovery Time | Reduced Hospitalization Rate |
Funding Transparency and the Evolution of Care
Journalistic integrity requires acknowledging that Remdesivir’s journey from a broad-spectrum antiviral to a COVID-specific treatment was accelerated by significant investment from Gilead Sciences. While the PubMed indexed results from the ACTT-1 trial showed a statistically significant reduction in recovery time (10 days vs. 15 days), subsequent studies like the WHO Solidarity trial suggested a more modest impact on overall mortality.
This nuance is vital. Remdesivir is not a “cure-all” but a tactical tool. Its value lies in its ability to stabilize a patient, preventing the need for mechanical ventilation. When used in conjunction with corticosteroids (like Dexamethasone), the synergy significantly improves survival rates in patients requiring oxygen.
Contraindications & When to Consult a Doctor
Remdesivir is not suitable for all patients. Because the drug is cleared through the kidneys, those with severe renal impairment—specifically a glomerular filtration rate (eGFR) below 30 mL/min—may be at risk of toxicity. Monitoring of liver enzymes (ALT and AST) is mandatory; a significant spike in these levels may indicate drug-induced liver injury, requiring immediate cessation of treatment.
Patients and caregivers should seek immediate medical intervention if they observe the following during or after treatment:
- Signs of Anaphylaxis: Sudden swelling of the face, lips, or tongue, and difficulty breathing.
- Hepatic Distress: Yellowing of the skin or eyes (jaundice) and dark-colored urine.
- Respiratory Decline: An increase in shortness of breath despite oxygen therapy.
As we move further into 2026, the trajectory of antiviral therapy is shifting toward personalized medicine. We are seeing a transition from “one-size-fits-all” protocols to genomic sequencing, where doctors choose the antiviral based on the specific mutation of the virus present in the patient. Remdesivir continues to be a vital part of this arsenal, providing a high-potency option for those whose illness has progressed beyond the reach of oral medications.
References
- National Institute of Allergy and Infectious Diseases (NIAID) – ACTT-1 Trial Data.
- The Lancet – World Health Organization (WHO) Solidarity Trial results.
- The New England Journal of Medicine (NEJM) – Efficacy and Safety of Remdesivir in Adults.
- Centers for Disease Control and Prevention (CDC) – Guidelines for Treatment of COVID-19.
- PubMed – Pharmacokinetics and Mechanism of Nucleotide Analogs.

