Researchers have developed a scalable sequencing method using RNA “barcodes” to map neuronal connectivity with single-synapse precision. By transforming brain mapping into a genomic task, this breakthrough allows scientists to identify previously unknown cellular links in mice, potentially accelerating the detection and treatment of complex neurological disorders.
For decades, the “connectome”—the comprehensive map of neural connections in the brain—has remained the holy grail of neuroscience. Traditional methods, such as electron microscopy, provide immense detail but are agonizingly slow and computationally expensive. This new approach shifts the paradigm: instead of looking at the brain through a microscope, researchers are now “reading” the brain as if it were a genetic code.
This is not merely a technical victory; it is a clinical gateway. By understanding exactly how neurons communicate, One can pinpoint where the “wiring” fails in conditions like Alzheimer’s, Parkinson’s and schizophrenia. For patients, So a future where neurodegenerative diseases are caught not by the appearance of plaques or atrophy, but by the earliest detectable disruptions in synaptic architecture.
In Plain English: The Clinical Takeaway
- Faster Mapping: Scientists can now map brain connections using the same high-speed technology used for DNA sequencing.
- Precision Medicine: This allows for the identification of specific “broken” connections in the brain, rather than treating the brain as a single organ.
- Early Warning: It could lead to diagnostic tools that detect brain diseases years before physical symptoms appear.
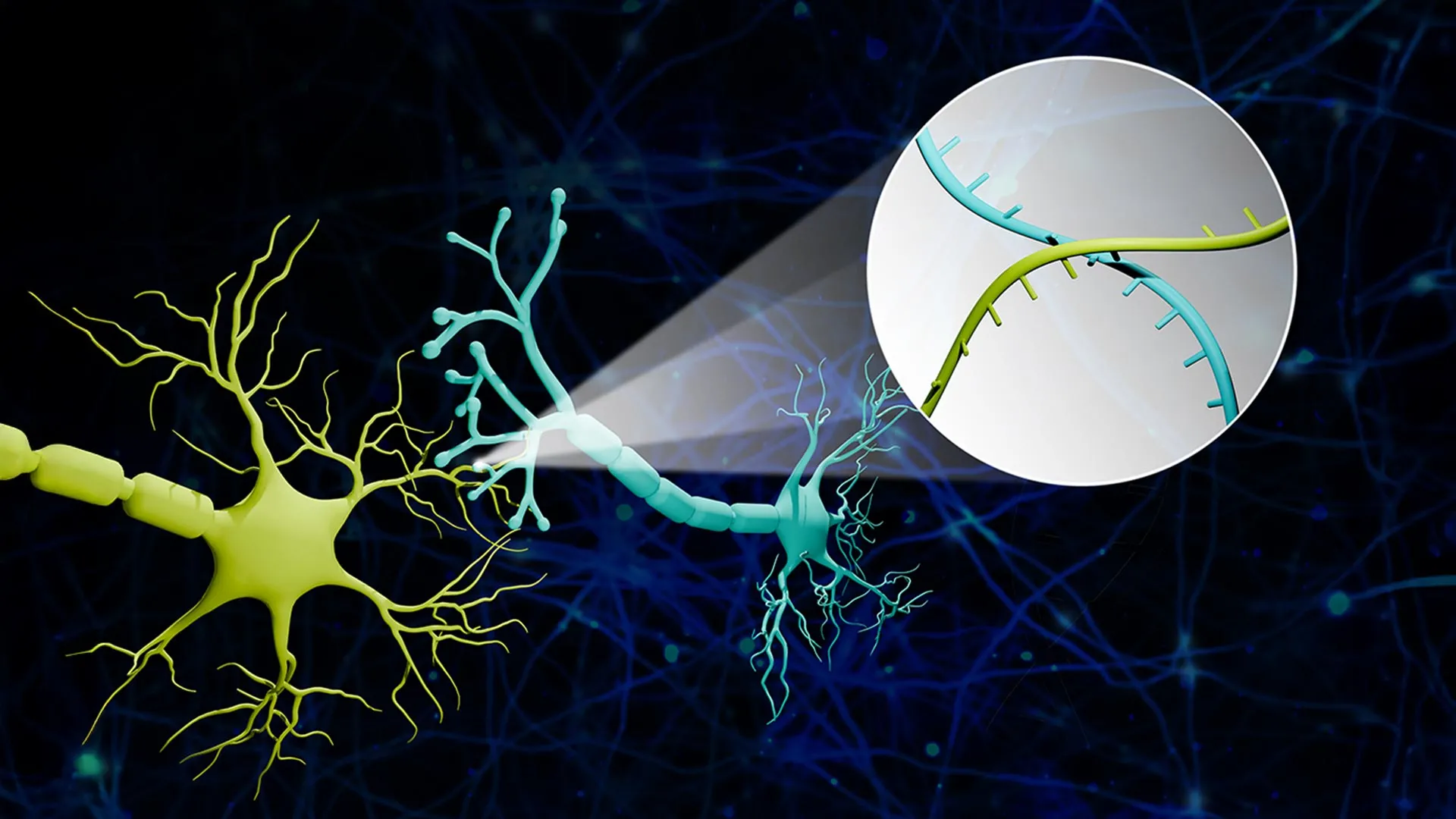
The Molecular Mechanism: How RNA Barcoding Decodes the Synapse
The core of this breakthrough lies in the mechanism of action—the specific biochemical process through which the technology works. Researchers utilize viral vectors to deliver unique RNA sequences (the “barcodes”) into neurons. These barcodes are then transported across the synapse—the microscopic gap between neurons—to the receiving cell.
By sequencing the RNA in the receiving neuron, scientists can identify exactly which “sender” neuron it is connected to. This effectively turns a spatial problem (where is the wire?) into a sequencing problem (what is the code?). This method allows for the mapping of thousands of links simultaneously, a feat that would take years using traditional double-blind tracing methods.
The precision here is critical. By achieving single-synapse resolution, researchers can distinguish between a general region of activity and a specific functional circuit. This is the difference between knowing a city has electricity and knowing exactly which light switch controls which bulb.
From Murine Models to Global Clinical Application
Although the current data is derived from murine (mouse) models, the implications for human healthcare systems are profound. In the United States, the FDA is increasingly prioritizing “biomarker-driven” drug development. If these RNA barcodes can be adapted for human tissue or organoids, we could see a new class of diagnostic tests for neurological dysfunction.
In Europe, the EMA and the NHS in the UK are shifting toward personalized medicine. A map of a patient’s specific neural circuitry could allow clinicians to target deep brain stimulation (DBS) with surgical precision, reducing the side effects associated with “off-target” stimulation of healthy brain tissue.
The funding for this research typically stems from a hybrid of public grants (such as the NIH in the US or the ERC in Europe) and private philanthropic ventures. Transparency in funding is essential here to ensure that the pursuit of “brain-mapping” does not prioritize commercial viability over patient safety and ethical considerations regarding cognitive privacy.
“The ability to translate connectivity into a sequence allows us to apply the full power of genomic tools to the brain’s architecture, potentially uncovering the ‘dark matter’ of neural circuits that drive psychiatric illness.”
| Feature | Traditional Electron Microscopy | RNA Barcoding Technique |
|---|---|---|
| Resolution | Ultra-high (Nanometer) | High (Single-Synapse) |
| Scalability | Low (Slow/Manual) | High (Automated Sequencing) |
| Throughput | Few hundred connections | Thousands of simultaneous links |
| Primary Use | Structural Anatomy | Functional Connectivity Mapping |
Bridging the Gap: Impact on Neurodegenerative Pathology
The most immediate application of this technology is in the study of proteinopathies—diseases where misfolded proteins (like amyloid-beta or tau) disrupt brain function. By mapping the “hidden wiring,” researchers can see how these toxic proteins travel along specific neural pathways, effectively “mapping the spread” of the disease in real-time.
This allows for the development of targeted therapeutics that can block these pathways. Instead of a systemic drug that affects the whole brain, we may move toward “circuit-specific” interventions. This reduces the risk of contraindications—situations where a drug’s side effects outweigh its benefits—by limiting the drug’s impact to the diseased circuit.
the data suggests a significant reduction in the “information gap” regarding how the prefrontal cortex communicates with the amygdala, which is central to understanding anxiety and PTSD. By identifying the specific barcodes of these circuits, pharmacological interventions can be tuned to the individual’s unique neural architecture.
Contraindications & When to Consult a Doctor
It is critical to note that RNA barcoding is currently a research tool and not a clinical treatment. There are no approved “brain-mapping” injections for human patients at this time. Patients should be wary of any “wellness clinic” claiming to offer neural rewiring or connectivity mapping via RNA technology.
Consult a board-certified neurologist immediately if you or a loved one experience the following “red flag” neurological symptoms:
- Sudden, unexplained memory loss or cognitive disorientation.
- Rapidly progressing tremors or loss of motor coordination.
- Acute changes in personality or mood that interfere with daily functioning.
- Severe, persistent headaches accompanied by vision changes or numbness.
The Future of the Connectome
We are entering an era where the brain is no longer a “black box.” The transition from anatomical observation to digital sequencing is a leap similar to the transition from hand-drawn maps to GPS. While we are still years away from a full human connectome, the scalability of RNA barcoding provides the first realistic timeline for achieving it.
As we refine these tools, the focus must remain on ethical implementation. The ability to map the brain’s wiring is a double-edged sword; while it offers hope for curing dementia, it also raises questions about the nature of identity and privacy. As a physician, my priority is the translation of this data into bedside care—ensuring that the “hidden wiring” discovered in the lab leads to tangible healing in the clinic.