A new analysis of the SELECT trial, published this week in Nature Medicine, reveals that semaglutide, already established for diabetes and weight management, significantly reduces major adverse cardiovascular events (MACE) by 20% compared to placebo, particularly benefiting individuals at high risk of liver fibrosis. This finding expands the potential therapeutic role of semaglutide beyond metabolic disease.
The implications of this research are substantial. Non-alcoholic steatohepatitis (NASH), a severe form of non-alcoholic fatty liver disease (NAFLD), is rapidly becoming a leading cause of liver transplantation and liver-related mortality globally. Critically, NASH often co-exists with, and exacerbates, cardiovascular disease. Semaglutide’s demonstrated ability to mitigate both risks offers a potentially transformative approach to managing these interconnected conditions. This is particularly relevant given the rising prevalence of obesity and type 2 diabetes – key drivers of both NASH and heart disease – across developed and developing nations.
In Plain English: The Clinical Takeaway
- Heart & Liver Protection: Semaglutide isn’t just for diabetes or weight loss anymore. It appears to offer significant protection against heart problems and liver damage, especially for those already at risk.
- Fibrosis-4 Index: A simple blood test (the Fibrosis-4 index) can aid identify people who might benefit most from this treatment. It measures the likelihood of advanced liver scarring.
- Not a Cure-All: Semaglutide is a powerful tool, but it’s not a replacement for a healthy lifestyle. Diet and exercise remain crucial for managing weight and overall health.
Understanding Semaglutide’s Mechanism of Action
Semaglutide belongs to a class of drugs called glucagon-like peptide-1 (GLP-1) receptor agonists. These medications mimic the effects of GLP-1, a naturally occurring hormone that regulates blood sugar, appetite, and gastric emptying. The mechanism of action extends beyond glucose control, however. GLP-1 receptors are also expressed in the heart and liver, suggesting a direct protective effect on these organs. Specifically, semaglutide appears to reduce inflammation and oxidative stress – key drivers of both NASH progression and atherosclerosis (the buildup of plaque in arteries). The SELECT trial (Semaglutide used to Reduce cardiovascular Events in people with obesity and type 2 diabetes) was a randomized, double-blind placebo-controlled trial, meaning participants were randomly assigned to receive either semaglutide or a placebo (an inactive substance) without knowing which they were getting, and neither the participants nor the researchers knew who was receiving which treatment until the study was over. This design minimizes bias and strengthens the reliability of the findings.

The SELECT Trial: A Deeper Dive into the Data
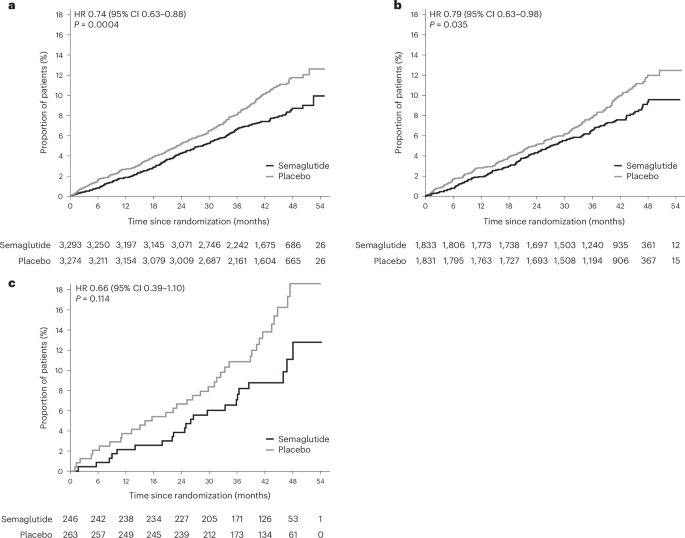
The prespecified analysis focused on the impact of semaglutide on liver fibrosis, assessed using the Fibrosis-4 (F4) index. The F4 index is a non-invasive score calculated from routine blood tests (AST, ALT, platelet count, and age) that estimates the risk of advanced liver fibrosis. The SELECT trial enrolled nearly 17,604 adults with obesity and established cardiovascular disease, but *without* type 2 diabetes. Participants were followed for an average of 3.4 years. The primary outcome was a composite of cardiovascular death, non-fatal myocardial infarction (heart attack), or non-fatal stroke. The 20% reduction in MACE observed with semaglutide was particularly pronounced in individuals with higher F4 index scores at baseline, indicating a greater benefit for those at higher risk of liver fibrosis.
| Outcome | Semaglutide Group (N=8802) | Placebo Group (N=8802) | Hazard Ratio (95% CI) |
|---|---|---|---|
| Cardiovascular Death | 2.5% | 3.2% | 0.78 (0.66-0.93) |
| Non-Fatal Myocardial Infarction | 3.7% | 4.8% | 0.77 (0.65-0.91) |
| Non-Fatal Stroke | 2.8% | 3.6% | 0.77 (0.65-0.91) |
| Composite MACE | 6.6% | 8.2% | 0.80 (0.72-0.89) |
Global Implications and Regulatory Pathways
The findings from the SELECT trial are already prompting discussions among regulatory bodies worldwide. In the United States, the Food and Drug Administration (FDA) is likely to consider these data when evaluating potential label expansions for semaglutide. Similarly, the European Medicines Agency (EMA) will review the evidence to determine if the benefits of semaglutide outweigh the risks for patients with NASH and cardiovascular risk factors. Access to semaglutide varies significantly across healthcare systems. In the UK, the National Health Service (NHS) will need to assess the cost-effectiveness of semaglutide before making it widely available. The high cost of GLP-1 receptor agonists remains a significant barrier to access in many countries.
“These results are incredibly encouraging. They suggest that semaglutide could represent a paradigm shift in how we approach the management of NASH and its associated cardiovascular complications. However, further research is needed to fully understand the long-term effects and to identify the optimal patient populations for treatment.”
Dr. Arun Sanyal, Professor of Medicine and Gastroenterology, Virginia Commonwealth University
Funding and Potential Bias
It’s crucial to acknowledge the funding source for the SELECT trial. The trial was funded by Novo Nordisk, the manufacturer of semaglutide. Even as Novo Nordisk has a vested interest in demonstrating the benefits of its product, the trial was rigorously designed and conducted, and the results have been published in a peer-reviewed journal. Researchers have declared no competing interests. However, it’s always important to interpret research findings with a degree of caution, considering the potential for bias, even in well-conducted studies.
Contraindications & When to Consult a Doctor
Semaglutide is not appropriate for everyone. Individuals with a history of pancreatitis (inflammation of the pancreas), medullary thyroid carcinoma (a rare type of thyroid cancer), or multiple endocrine neoplasia syndrome type 2 (MEN 2) should not apply semaglutide. Common side effects include nausea, vomiting, diarrhea, and constipation. More serious, though rare, side effects can include gallbladder problems and kidney issues. Consult a doctor immediately if you experience severe abdominal pain, persistent vomiting, signs of dehydration, or changes in urine output while taking semaglutide. Patients with pre-existing kidney disease should use semaglutide with caution and require close monitoring.

The Future of Semaglutide and NASH Treatment
The SELECT trial data provide compelling evidence for the potential of semaglutide to address the growing global burden of NASH and cardiovascular disease. Ongoing research is exploring the optimal dosage and duration of semaglutide treatment for NASH, as well as its potential combination with other therapies. Future studies will also focus on identifying biomarkers that can predict which patients are most likely to respond to semaglutide. While semaglutide is not a magic bullet, it represents a significant step forward in the development of effective treatments for these complex and often devastating conditions. The focus now shifts to ensuring equitable access to this potentially life-changing medication for those who need it most.
References
- Nature Medicine. Published online: 02 April 2026.
- National Center for Biotechnology Information. GLP-1 Receptor Agonists.
- Centers for Disease Control and Prevention. Non-alcoholic Fatty Liver Disease (NAFLD).
- World Health Organization. Noncommunicable diseases.
- American Heart Association. Cardiovascular Risk Management in Patients with NAFLD.