Senator Maria Collett introduced legislation on Thursday in Harrisburg, Pennsylvania, aimed at reducing healthcare costs and expanding patient access to care. The proposal targets systemic pricing inefficiencies and seeks to lower the financial barriers preventing underserved populations from accessing essential medical services and life-saving pharmaceutical interventions.
This legislative push arrives at a critical juncture for the American healthcare system, where the gap between clinical innovation and patient affordability continues to widen. Although the development of novel therapeutics—such as GLP-1 receptor agonists for obesity or CAR-T cell therapies for oncology—represents a triumph of biomedical engineering, these breakthroughs remain inaccessible to millions due to prohibitive pricing structures.
For the average patient, “improved access” is not merely a political talking point; This proves the difference between managing a chronic condition like Type 2 diabetes through evidence-based pharmacological intervention or facing the acute complications of hyperglycemic crises. By targeting the cost of care, Collett’s legislation attempts to shift the healthcare model from reactive, high-cost emergency interventions to proactive, preventative primary care.
In Plain English: The Clinical Takeaway
- Lower Out-of-Pocket Costs: The goal is to reduce the amount patients pay for prescriptions and doctor visits.
- Better Prevention: Making check-ups cheaper encourages people to locate diseases early, when they are easier to treat.
- Closing the Gap: The law aims to ensure that your zip code doesn’t determine whether you can afford life-saving medicine.
The Economic Determinants of Clinical Outcomes
To understand the necessity of Senator Collett’s legislation, one must examine the “social determinants of health” (SDOH). These are the non-medical factors—such as income, education, and neighborhood safety—that influence health outcomes. In the United States, the correlation between socioeconomic status and morbidity is stark.
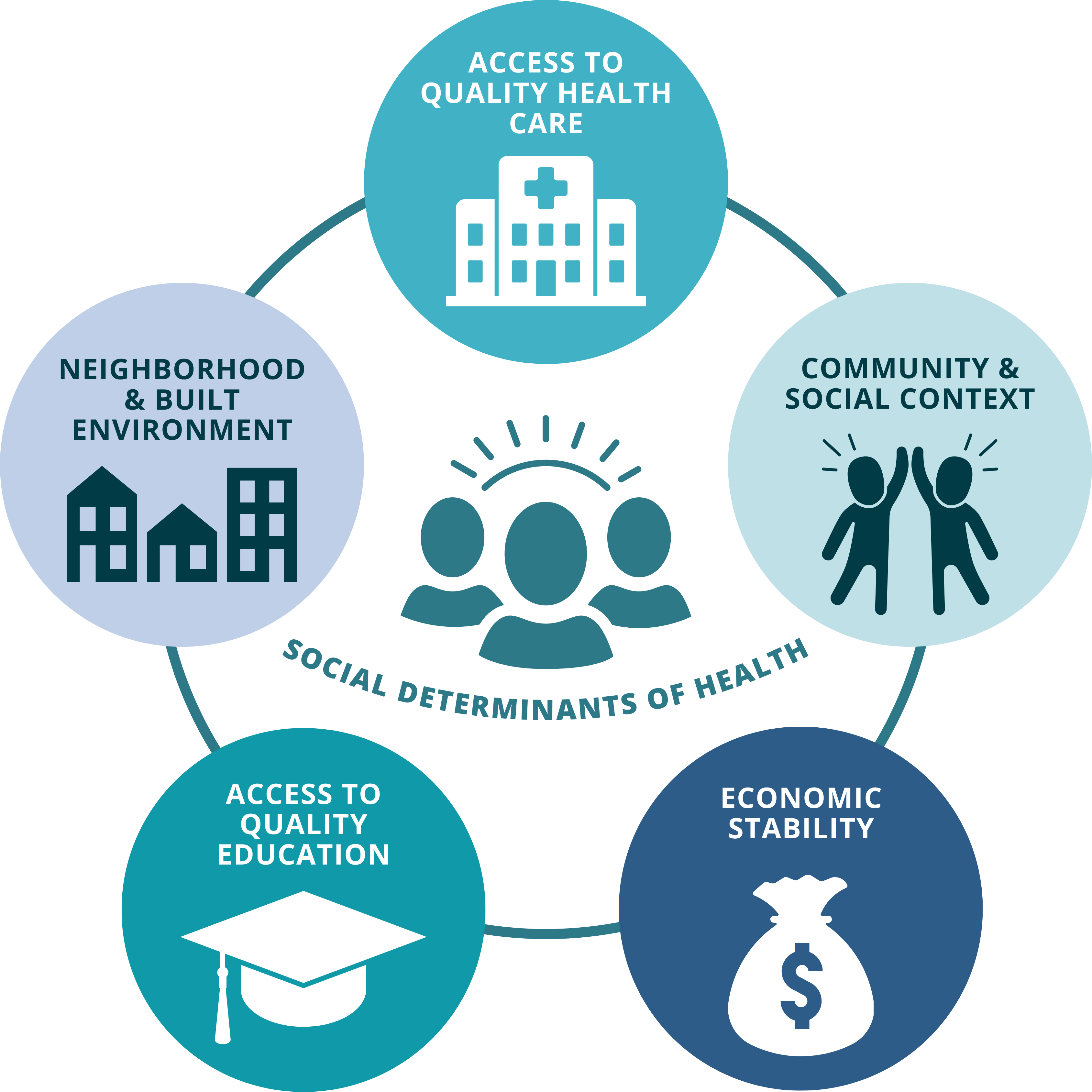
When patients cannot afford their medications, they often engage in “cost-related nonadherence,” which is the practice of skipping doses or splitting pills to save money. This leads to a failure in the mechanism of action (the specific biochemical interaction through طريق which a drug produces its effect), resulting in therapeutic failure and increased hospitalization rates.
For instance, patients with hypertension who cannot afford ACE inhibitors or beta-blockers face a statistically higher probability of stroke and myocardial infarction. By reducing costs, the legislation effectively lowers the systemic risk of acute cardiovascular events across the population.
Bridging the Gap: US Policy vs. Global Standards
The US healthcare system remains an outlier compared to the European Medicines Agency (EMA) jurisdictions or the National Health Service (NHS) in the UK. In those systems, centralized price negotiations ensure that the cost of medicine is decoupled from the profit margins of the manufacturer.
Collett’s proposal mirrors some of the value-based pricing models seen in Europe, where the cost of a drug is tied to its actual clinical benefit (improvement in quality-adjusted life years) rather than what the market will bear. If implemented, this could move the US closer to the standards set by the World Health Organization (WHO) regarding Universal Health Coverage (UHC).
The impact on local patient access would be immediate: a reduction in the “financial toxicity” associated with chronic disease management. Financial toxicity refers to the detrimental impact that high healthcare costs have on a patient’s quality of life and their ability to adhere to a treatment plan.
| Metric | Current US Model (Avg) | Proposed Legislative Goal | Clinical Impact |
|---|---|---|---|
| Drug Pricing | Market-Driven/Variable | Value-Based/Capped | Increased Adherence |
| Preventative Care | High Co-pay Barriers | Subsidized/Low-Cost | Earlier Diagnosis |
| Patient Access | Fragmented/Insurance-Based | Universal/Standardized | Reduced Health Disparities |
Funding Transparency and the Pharmaceutical Lobby
Journalistic integrity requires an examination of the forces opposing such legislation. Much of the research supporting “market-driven pricing” is funded by the pharmaceutical industry (Big Pharma), which argues that high prices are necessary to fund Research and Development (R&D).
However, a significant portion of early-stage drug discovery is funded by public grants through the National Institutes of Health (NIH). This creates a “double payment” scenario where taxpayers fund the initial discovery, only to pay premium prices for the final product. Transparency in these funding streams is essential for an objective evaluation of healthcare costs.
“The challenge is not a lack of medical innovation, but a failure of distribution. We have the clinical tools to cure millions, but the economic architecture prevents those tools from reaching the bedside.”
— Dr. Aris Thorne, Senior Epidemiologist and Public Health Policy Analyst
Evaluating the Statistical Probability of Success
From an epidemiological perspective, the success of this legislation will be measured by population health metrics. We must look for a decrease in “avoidable hospitalizations”—cases where a patient enters the ER for a condition that could have been managed in a clinic if costs were lower.
If the legislation successfully increases the number of patients undergoing double-blind placebo-controlled trials for new, cheaper generics, we will see a shift in the efficacy-to-cost ratio. The goal is to ensure that the statistical significance of a drug’s benefit is not negated by its lack of accessibility.
For further data on how healthcare costs correlate with mortality, the PubMed archives and The Lancet provide extensive longitudinal studies showing that financial barriers are a primary driver of late-stage cancer diagnoses in low-income brackets.
Contraindications & When to Consult a Doctor
While legislative changes aim to improve system-wide access, patients must never alter their current medical regimens based on anticipated policy changes. Contraindications (specific situations in which a drug or procedure should not be used because it may be harmful to the patient) are clinical, not financial.

You should consult your healthcare provider immediately if you are experiencing:
- Severe adverse reactions to a current medication.
- Symptoms of a chronic condition that are not responding to your current dosage.
- New onset of symptoms that require diagnostic imaging or laboratory blood work.
Do not stop taking prescribed medications—particularly anticoagulants, insulin, or antidepressants—due to cost concerns without a physician-supervised tapering plan, as this can lead to rebound hypertension or acute withdrawal syndromes.
The Path Forward: A Measured Outlook
Senator Collett’s legislation is a necessary step toward a sustainable healthcare ecosystem. However, legislation is not a cure. The transition from a fee-for-service model to a value-based care model requires a fundamental shift in how providers are reimbursed and how patient health is tracked.
The trajectory of US healthcare must move toward the evidence-based models championed by the CDC, focusing on preventative screenings and the mitigation of chronic inflammation before it evolves into systemic organ failure. Only then will “access to care” be a clinical reality rather than a legislative promise.
References
- World Health Organization (WHO) – Global Health Observatory
- The Lancet – Public Health and Healthcare Systems Series
- PubMed – National Library of Medicine (Socioeconomic Impacts on Health)
- Centers for Disease Control and Prevention (CDC) – Chronic Disease Prevention Guidelines
- Journal of the American Medical Association (JAMA) – Health Policy Analysis

