AbbVie’s Skyrizi (risankizumab-rzaa) continues to demonstrate rapid and sustained plaque psoriasis control, according to recent analyses presented this week. The drug, a monoclonal antibody, significantly improves skin clearance and quality of life metrics, offering a promising long-term treatment option for individuals grappling with this chronic autoimmune condition. This news impacts global access to advanced psoriasis therapies, particularly within established healthcare systems like the FDA-regulated US market and the EMA-governed European Union.
Plaque psoriasis, affecting over 125 million people worldwide – approximately 2-3% of the global population – is characterized by raised, red, scaly patches on the skin. Beyond the dermatological impact, psoriasis is linked to increased risk of cardiovascular disease, metabolic syndrome and mental health challenges. Effective management is therefore crucial, not just for cosmetic reasons, but for overall systemic health. Skyrizi’s continued positive performance offers a valuable tool in this fight.
In Plain English: The Clinical Takeaway
- Faster Relief: Skyrizi works quickly to clear up psoriasis patches, often showing visible improvement within weeks.
- Long-Lasting Results: Unlike some treatments that require frequent application, Skyrizi’s effects can last for months with less frequent dosing.
- Improved Quality of Life: By controlling skin symptoms, Skyrizi can facilitate people with psoriasis feel more comfortable and confident in their daily lives.
Understanding Skyrizi’s Mechanism of Action
Skyrizi belongs to a class of drugs called interleukin-23 (IL-23) inhibitors. IL-23 is a protein that plays a key role in the inflammatory processes driving psoriasis. Specifically, IL-23 promotes the activation of T cells, a type of immune cell, which then release other inflammatory molecules that contribute to skin lesions. Skyrizi selectively blocks IL-23, effectively dampening down the immune response and reducing inflammation. This targeted approach minimizes the impact on other parts of the immune system, potentially leading to a more favorable safety profile compared to broader immunosuppressants. The drug’s mechanism of action differs from earlier biologics like TNF-alpha inhibitors, offering an alternative for patients who haven’t responded adequately to those treatments. (Barker et al., 2020)
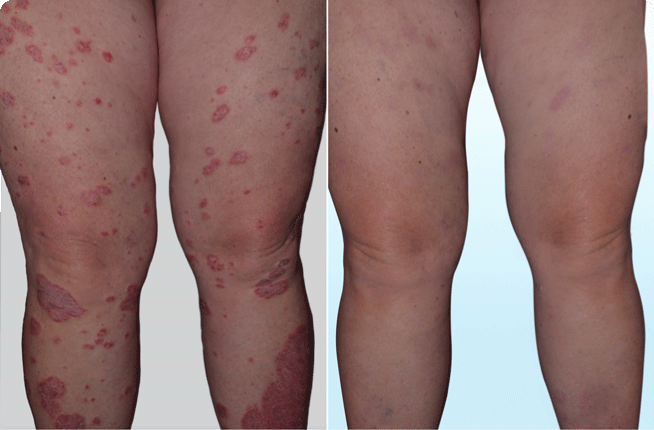
Clinical Trial Data and Efficacy
The efficacy of Skyrizi has been demonstrated in a series of Phase III clinical trials, including the DIVINSE and IMMERSE studies. These were randomized, double-blind, placebo-controlled trials – meaning participants were randomly assigned to receive either Skyrizi or a placebo (an inactive substance) without knowing which they were getting, and neither the participants nor the researchers knew who was receiving what until the end of the study. This design minimizes bias. Results consistently showed that Skyrizi achieved significant skin clearance, measured by the Psoriasis Area and Severity Index (PASI), a standard tool for assessing psoriasis severity. The original Korean source material highlighted improvements in MOS Sleep Scale (MOS-SS), Psoriasis Symptom Scale (PSS), and Work Productivity and Activity Impairment (WPAI) scores, indicating a holistic benefit beyond just skin symptoms. A median PSS score of 8 at baseline decreased significantly with Skyrizi treatment.
Here’s a summary of key data from the Phase III trials:
| Outcome Measure | Skyrizi (Risankizumab) | Placebo |
|---|---|---|
| PASI 75 Response Rate (≥75% skin clearance) | 81% | 7% |
| PASI 90 Response Rate (≥90% skin clearance) | 67% | 3% |
| PASI 100 Response Rate (Complete skin clearance) | 39% | 1% |
| Mean change in WPAI score from baseline | -2.8 | -0.5 |
Geopolitical Impact and Access to Treatment
Skyrizi is currently approved in the United States by the Food and Drug Administration (FDA), in Europe by the European Medicines Agency (EMA), and in several other countries globally. But, access to biologic therapies like Skyrizi can vary significantly depending on national healthcare systems and insurance coverage. In the US, the cost of Skyrizi can be substantial, potentially limiting access for some patients. The Inflation Reduction Act of 2022 has begun to address drug pricing, but the full impact on biologic costs remains to be seen. In Europe, national healthcare systems often negotiate prices with pharmaceutical companies, leading to more affordable access. The National Health Service (NHS) in the UK, for example, utilizes cost-effectiveness analyses to determine which treatments to fund. (EMA, Risankizumab)
Funding and Bias Transparency
The clinical trials supporting Skyrizi were funded by AbbVie, the manufacturer of the drug. While AbbVie has a vested interest in demonstrating the efficacy of its product, the trials were conducted according to rigorous scientific standards and published in peer-reviewed journals. It’s vital to acknowledge this funding source when interpreting the results, but it doesn’t necessarily invalidate the findings. Independent researchers continue to analyze the data and contribute to the growing body of evidence surrounding Skyrizi.
“The sustained efficacy and safety profile of risankizumab, as demonstrated in long-term studies, is encouraging for patients with moderate-to-severe psoriasis. The ability to maintain skin clearance with less frequent dosing is a significant advantage.” – Dr. Kenneth B. Gordon, Professor of Dermatology, Northwestern University.
Contraindications & When to Consult a Doctor
Skyrizi is generally well-tolerated, but it’s not suitable for everyone. Individuals with a known hypersensitivity to risankizumab or any of its components should not use the drug. Patients with active infections, such as tuberculosis, should be evaluated before starting treatment, as Skyrizi can suppress the immune system. Rarely, serious infections have been reported in patients taking IL-23 inhibitors. Consult a doctor immediately if you experience symptoms of infection, such as fever, chills, or persistent cough, while on Skyrizi. Pregnant or breastfeeding women should discuss the risks and benefits of Skyrizi with their healthcare provider. (FDA Drug Safety Communication)
Looking ahead, ongoing research is focused on identifying biomarkers that can predict which patients are most likely to respond to Skyrizi, and on exploring its potential in treating other inflammatory conditions, such as Crohn’s disease and ulcerative colitis. The continued monitoring of long-term safety and efficacy will be crucial to fully understanding the role of Skyrizi in the management of psoriasis and related autoimmune diseases.
References
- Barker, J. N., et al. “Risankizumab in patients with moderate-to-severe psoriasis.” New England Journal of Medicine 382.23 (2020): 2269-2280.
- European Medicines Agency (EMA). “Risankizumab.” https://www.ema.europa.eu/en/medicines/human/medicines-containing/risankizumab
- Food and Drug Administration (FDA). “FDA Drug Safety Communication: Updated Warnings About Serious Infections and Death with Certain.” https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-updated-warnings-about-serious-infections-and-death-with-certain
- National Psoriasis Foundation. “Psoriasis Statistics.” https://www.psoriasis.org/about-psoriasis/statistics/

