The Looming Threat of Antimicrobial Resistance: A Global Healthcare Challenge
Rising rates of drug-resistant infections, driven by “superbugs” – bacteria, viruses, fungi, and parasites that have evolved to withstand antimicrobial drugs – pose a significant and growing threat to global public health. This week, reports indicate a concerning acceleration in resistance patterns, potentially reversing decades of progress in treating common infections and complicating routine medical procedures. The issue is particularly acute in low- and middle-income countries, but increasingly impacts healthcare systems worldwide.
The escalating crisis isn’t simply about infections becoming harder to treat; it’s about a potential return to a pre-antibiotic era where even minor injuries could prove fatal. The economic burden is also substantial, with longer hospital stays, increased healthcare costs, and lost productivity all contributing to a strain on global economies. Understanding the mechanisms driving this resistance, and the strategies to combat it, is paramount.
In Plain English: The Clinical Takeaway
- Superbugs are evolving: Bacteria are changing, making our current medicines less effective.
- This impacts everyone: Simple infections could become life-threatening again.
- Prevention is key: Good hygiene and responsible antibiotic use are crucial to slowing the spread.
The Mechanisms of Resistance and Emerging Threats
Antimicrobial resistance (AMR) isn’t a spontaneous event. It’s a consequence of evolutionary pressure. Bacteria, through genetic mutations and horizontal gene transfer (the sharing of genetic material between bacteria), develop mechanisms to evade the effects of antimicrobial drugs. These mechanisms include enzymatic degradation of the drug, alteration of the drug target, reduced drug uptake, and increased drug efflux (pumping the drug out of the cell). The Forbes article highlights the growing concern with carbapenem-resistant organisms (CROs), a family of bacteria resistant to one or both of the last-resort antibiotics, carbapenems.
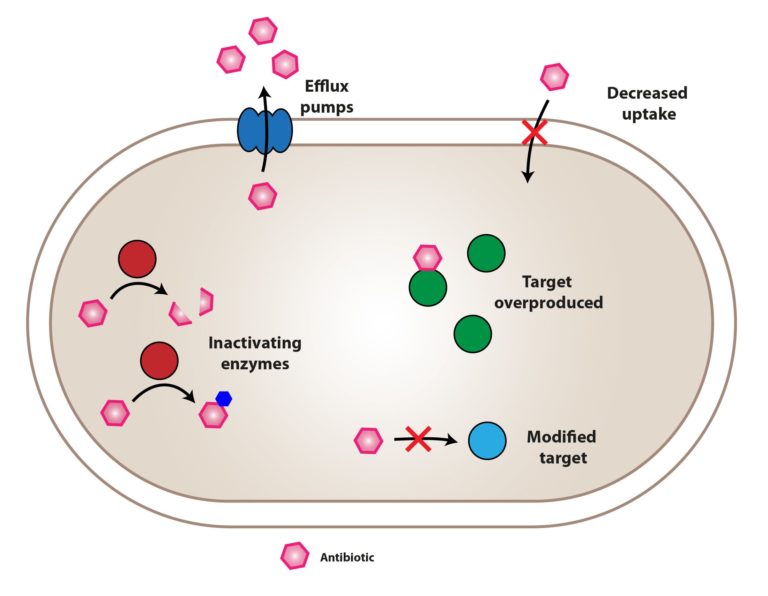
However, the threat extends beyond CROs. Methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), and extensively drug-resistant Mycobacterium tuberculosis (XDR-TB) continue to pose significant challenges. The rise of antifungal resistance, particularly in Candida auris, is alarming due to its ability to cause severe, often invasive infections and its propensity for healthcare-associated outbreaks. The underlying process involves selective pressure – when antibiotics are used, susceptible bacteria are killed, leaving resistant strains to proliferate. This represents exacerbated by overuse and misuse of antibiotics in both human medicine and agriculture.
Global Epidemiology and Regional Impacts
The World Health Organization (WHO) estimates that in 2019, AMR was directly responsible for an estimated 1.27 million deaths globally, and contributed to nearly 5 million deaths. The highest burden of AMR is observed in low- and middle-income countries, particularly in sub-Saharan Africa and South Asia, where access to diagnostics, appropriate antibiotics, and infection prevention and control measures are limited.
In the United States, the Centers for Disease Control and Prevention (CDC) estimates that AMR causes at least 35,000 deaths annually and contributes to over 2.8 million infections. The European Centre for Disease Prevention and Control (ECDC) reports similar trends across Europe, with increasing rates of resistance to key antibiotics. The impact on healthcare systems is substantial, leading to longer hospital stays, increased treatment costs, and higher mortality rates.
The regulatory landscape varies significantly. The FDA in the US is actively working to incentivize the development of new antibiotics through programs like the Generating Antibiotic Incentives Now (GAIN) Act. The EMA in Europe has implemented similar measures. However, the economic challenges of antibiotic development – the relatively low return on investment compared to other pharmaceutical areas – remain a significant barrier.
“The speed at which antimicrobial resistance is increasing is deeply concerning. We demand a coordinated global effort to address this threat, focusing on responsible antibiotic use, improved diagnostics, and the development of new antimicrobial agents.” – Dr. Tedros Adhanom Ghebreyesus, Director-General of the World Health Organization (WHO), stated in a recent press briefing.
Funding and Bias Transparency
Much of the research on antimicrobial resistance is funded by governmental agencies, such as the National Institutes of Health (NIH) in the US and the Medical Research Council (MRC) in the UK. However, pharmaceutical companies are also increasingly investing in AMR research, often driven by regulatory incentives. It’s crucial to acknowledge potential biases in industry-funded research, and to critically evaluate the methodology and results of such studies. A recent study published in The Lancet Infectious Diseases, funded by the Wellcome Trust, highlighted the need for greater transparency in AMR research funding. The Lancet Infectious Diseases
Data on Carbapenem Resistance
| Region | Carbapenem Resistance Rate (Klebsiella pneumoniae) – 2024 | Mortality Rate Associated with CRO Infections |
|---|---|---|
| North America | 15-25% | 20-40% |
| Europe | 10-20% | 15-35% |
| Asia | 30-60% | 30-50% |
| Africa | 40-70% | 40-60% |
Contraindications & When to Consult a Doctor
While AMR doesn’t directly present contraindications for individuals, it underscores the importance of responsible antibiotic use. Individuals with weakened immune systems, chronic illnesses, or recent hospitalizations are at higher risk of developing infections with resistant organisms.

Consult a doctor immediately if you experience:
- Fever that doesn’t respond to over-the-counter medication.
- Signs of infection (redness, swelling, pain, pus) that worsen despite treatment.
- Symptoms of a severe infection, such as difficulty breathing, confusion, or organ failure.
Avoid self-treating with antibiotics and always complete the full course of antibiotics prescribed by your doctor, even if you start to feel better.
The Future of AMR: Innovation and Prevention
Combating AMR requires a multifaceted approach. Developing new antibiotics is crucial, but it’s not a silver bullet. Research into alternative therapies, such as phage therapy (using viruses to infect and kill bacteria), immunotherapy (boosting the body’s immune response), and antimicrobial peptides, is gaining momentum. Improved diagnostics are also essential for identifying resistant organisms quickly and accurately, allowing for targeted treatment.
However, prevention remains the most effective strategy. This includes promoting good hygiene practices (handwashing, sanitation), implementing robust infection prevention and control measures in healthcare settings, and reducing the unnecessary use of antibiotics in both human medicine and agriculture. Global collaboration and coordinated action are essential to address this complex and evolving threat. The challenge is significant, but not insurmountable.
References
- World Health Organization – Antimicrobial Resistance
- Centers for Disease Control and Prevention – Antibiotic Resistance
- European Centre for Disease Prevention and Control – Antimicrobial Resistance
- “Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis” – The Lancet
- “Antimicrobial Resistance: A Global Threat” – JAMA

