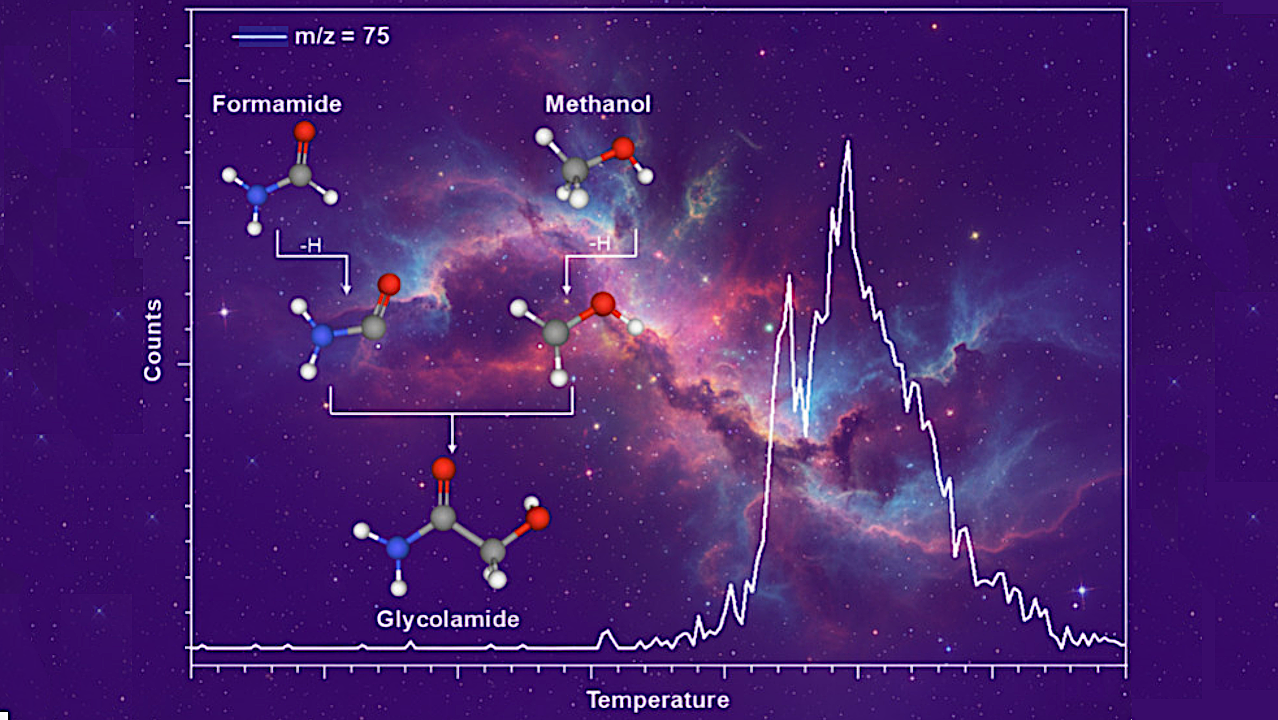
Astrobiologists have identified a nonequilibrium synthesis pathway for glycolamide (NH2COCH2OH) on interstellar nanoparticles, providing a critical chemical bridge to amino acids. This discovery, detailed via astrobiology.com, explains how complex organic molecules form in the harsh vacuum of space, fundamentally altering our understanding of prebiotic chemistry and cosmic evolution.
Let’s be clear: this isn’t just another “we found a molecule in space” press release. This is a fundamental shift in the thermodynamic narrative of the universe. For decades, the “equilibrium” model suggested that the chemistry of the interstellar medium (ISM) was too sluggish, too cold, and too diluted to produce the precursors of life without a massive external energy kick. We were looking for a spark; it turns out the engine was already idling.
The core of this discovery lies in the nonequilibrium aspect. In a standard chemical equilibrium, reactions move toward the lowest energy state. But on the surface of interstellar nanoparticles—essentially cosmic dust grains—the rules change. These grains act as catalytic substrates, lowering the activation energy required for synthesis. By bypassing the traditional thermodynamic hurdles, the synthesis of glycolamide becomes not just possible, but probable.
The Catalytic Engine: Why Nanoparticles Change the Math
To understand the “how,” we have to look at the surface science. Interstellar grains aren’t just rocks; they are complex lattices of silicates and carbonaceous material. When molecules like hydrogen cyanide (HCN) and formaldehyde (H2CO) adsorb onto these surfaces, they aren’t floating in a void—they are locked into a geometric orientation that favors specific bond formations.

This is essentially a natural version of heterogeneous catalysis. The nanoparticle surface stabilizes the transition state of the reaction, allowing glycolamide to form even at temperatures approaching absolute zero. If this were happening in a gas phase, the collision probability would be statistically insignificant. On a grain, the surface acts as a concentrator.
The synthesis of glycolamide is the “missing link” since it is a direct precursor to glycine, the simplest amino acid. If you can synthesize glycolamide in the ISM, you’ve effectively solved the “delivery problem” of prebiotic chemistry. We aren’t waiting for amino acids to form on a young Earth; we are importing them from the galactic cloud.
The 30-Second Verdict: Why This Disrupts Astrobiology
- Thermodynamic Bypass: Proves that complex organics can form without high-energy planetary environments.
- Prebiotic Pipeline: Establishes a direct chemical route from simple interstellar gases to amino acid precursors.
- Universal Scalability: Suggests that the building blocks of life are a standard feature of galaxy formation, not a planetary fluke.
Bridging the Gap: From Cosmic Dust to Biological Code
If we treat the universe as a giant computation engine, the synthesis of glycolamide is like discovering a new primitive operation in the cosmic assembly language. It bridges the gap between simple nitriles and the complex polymers required for life. This isn’t just about chemistry; it’s about the information density of the universe.
The implications for the “Rare Earth” hypothesis are devastating. If the precursors to life are synthesized ubiquitously on nanoparticles across the Milky Way, then the “bottleneck” for life isn’t the availability of materials—it’s the environment that allows those materials to organize. We move from asking “Where do the ingredients come from?” to “Where does the recipe secure executed?”
“The realization that nonequilibrium processes on grain surfaces can drive the synthesis of prebiotic molecules suggests that the chemical complexity of the universe is far more aggressive than our previous equilibrium models predicted. We are seeing a universe that is actively ‘primed’ for life.”
To put this in perspective, consider the difference between a random walk and a guided path. Equilibrium chemistry is a random walk. Nonequilibrium synthesis on nanoparticles is a guided path. The surface chemistry provides the “logic gate” that directs the reaction toward glycolamide rather than wasting energy on useless side-products.
Comparing the Synthesis Pathways
To understand the efficiency gain, we have to compare the traditional gas-phase expectations with the nanoparticle-mediated reality. The difference isn’t marginal; it’s logarithmic.
| Feature | Gas-Phase Equilibrium | Nanoparticle Nonequilibrium |
|---|---|---|
| Reaction Rate | Extremely Low (Collision-limited) | High (Surface-catalyzed) |
| Energy Barrier | High Activation Energy | Lowered via Surface Adsorption |
| Product Yield | Trace amounts of simple organics | Significant precursors (Glycolamide) |
| Temperature Req. | High thermal energy required | Effective at < 20K |
This shift in understanding aligns with recent data from the James Webb Space Telescope (JWST), which has been detecting complex organic molecules in protoplanetary disks. We are seeing the “shipping manifests” of these nanoparticles arriving in new solar systems in real-time.
The Macro Implications: A Universe of Chemical Determinism
When we look at the broader ecosystem of cosmic evolution, this discovery pushes us toward a model of chemical determinism. If the laws of physics and the properties of interstellar dust inevitably lead to the production of amino acid precursors, then life is not an accident—it is a predictable outcome of galactic chemistry.
This connects directly to the work being done in NASA’s Astrobiology program and the search for biosignatures. We are no longer looking for “miracles”; we are looking for the expected result of a nonequilibrium system. The “Information Gap” here was the assumption that the void of space was chemically inert. We now realize it’s a laboratory.
From a technical standpoint, the next step is simulating these nanoparticle surfaces using high-fidelity molecular dynamics. We need to move beyond the basic models and start looking at the specific mineralogy of these grains. Are they amorphous silicates? Crystalline carbon? The specific “hardware” of the nanoparticle determines the “software” of the chemical reaction.
The takeaway is simple: the universe is not a cold, dead place. It is a churning, nonequilibrium reactor that has been synthesizing the precursors of life for billions of years. We aren’t the exception; we are the result of a very efficient, very old, and very cosmic manufacturing process.