Recent studies have shed light on the potential of bispecific T cell engagers (TCEs) in treating treatment-refractory autoimmune connective tissue diseases, particularly anti-synthetase syndrome (ASyS) and diffuse cutaneous systemic sclerosis (dcSSc). As traditional therapies often fall short for patients with severe manifestations of these conditions, innovative therapies like blinatumomab and teclistamab are being explored as promising alternatives.
Patients suffering from ASyS and dcSSc frequently experience debilitating symptoms and complications that significantly impact their quality of life. These diseases are characterized by immune-mediated damage to multiple organ systems, leading to severe muscle weakness, pulmonary involvement, and extensive skin fibrosis. Current treatment options are limited, especially for those who have not responded adequately to conventional therapies such as methotrexate, rituximab (RTX), and other immunomodulatory drugs.
At the University Hospital of Düsseldorf, a compassionate leverage program has been initiated to administer bispecific T cell engagers to eligible patients. This program follows strict ethical guidelines, including compliance with the Declaration of Helsinki and local regulations, ensuring that patients provide informed consent before participation.
Patient Profiles and Treatment Challenges
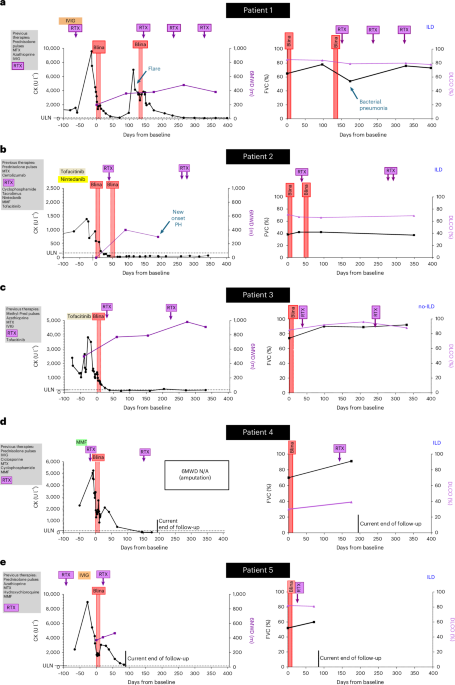
Five patients with ASyS and ten with dcSSc were enrolled in the study, all of whom had failed multiple rounds of previous therapies. Each patient with ASyS demonstrated positivity for anti-Jo1 autoantibodies, with three of the five also exhibiting autoantibodies against Ro-52. Notably, these patients had previously not responded to at least four different immunomodulatory medications.
For instance, Patient 1, a 65-year-old woman diagnosed with anti-Jo1+ ASyS, presented with severe muscle weakness, dyspnea, and laboratory findings that indicated elevated creatine kinase levels. Despite undergoing extensive treatment, including methotrexate, azathioprine, high-dose intravenous immunoglobulin (IVIG), and rituximab, she continued to experience recurrent flares and developed interstitial lung disease (ILD) characterized by a non-specific interstitial pneumonia (NSIP) pattern.
Similarly, Patient 2, a 36-year-old man, faced rapid progression of ILD and myocardial involvement despite treatment with RTX, cyclophosphamide, and tacrolimus. His treatment journey exemplifies the complexities and challenges associated with managing severe autoimmune diseases, as traditional therapies failed to provide adequate control over his symptoms.
Therapeutic Approach with Bispecific T Cell Engagers
The administration of TCEs, specifically blinatumomab and teclistamab, represents an innovative approach aimed at engaging T cells to target and eliminate autoreactive B cells responsible for disease progression. Blinatumomab, a bispecific T cell engager, is designed to redirect T cells towards B cells, leading to their destruction. This therapy has been shown to induce rapid depletion of B cells, with studies indicating a 90% reduction within four hours post-infusion.
In this compassionate use program, patients received a structured treatment regimen: blinatumomab was administered via continuous intravenous infusion for five days, followed by an increased dose over the next week. Teclistamab, was administered subcutaneously with the aim of achieving sustained T cell activation against B cell targets.
Monitoring and Future Implications
All patients were closely monitored throughout their treatment, with assessments including pulmonary function tests, laboratory evaluations, and imaging studies to track disease progression and response to therapy. Initial findings from the treatment indicate a potential for improved management of symptoms, although long-term efficacy and safety data are still needed to fully evaluate the impact of bispecific T cell engagers.
The innovative use of bispecific T cell engagers in autoimmune connective tissue diseases marks a significant step forward, particularly for patients who have exhausted conventional treatment options. The compassionate use program at the University Hospital of Düsseldorf not only provides hope for these patients but also contributes to the growing body of evidence supporting the efficacy of targeted therapies in autoimmune diseases.
As the research progresses, healthcare professionals and researchers alike will continue to monitor the outcomes of these treatments, aiming to establish clear guidelines and protocols for incorporation into standard care practices. The results from these studies could pave the way for broader applications of TCEs in various autoimmune disorders, enhancing treatment options for patients globally.
For those affected by autoimmune connective tissue diseases, ongoing research and clinical trials are crucial. We see essential to remain informed about emerging therapies and to engage with healthcare providers regarding the latest treatment options available.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always consult a healthcare professional for medical concerns.