The Role of the Gut Microbiome in Gastrointestinal Cancer Development
The increasing incidence of gastrointestinal cancers, including colorectal, pancreatic, and gastric cancers, particularly among younger individuals and minority populations, highlights the urgency of understanding their underlying causes. Emerging research emphasizes the critical role of the gut microbiome in influencing cancer risk through various mechanisms, including microbial metabolites, immune modulation, and inflammatory pathways.
Recent findings suggest that interactions between host metabolism and gut microbiota can significantly impact tumor initiation and progression in gastrointestinal malignancies. While many associations have been identified through observational and preclinical studies, the challenge remains to establish definitive causal links in human populations.
The Gut Microbiome: A Complex Ecosystem
The gut microbiome is a dynamic ecosystem that can undergo dysbiosis, a condition characterized by reduced microbial diversity and the proliferation of pathogenic bacteria. This shift often results in chronic low-grade inflammation, which is a precursor to cancer development. Dysbiosis is marked by the depletion of beneficial butyrate-producing bacteria like Roseburia and Lachnospiraceae, alongside an increase in pro-inflammatory species such as Fusobacterium nucleatum and enterotoxigenic Bacteroides fragilis.
This inflammatory milieu activates pro-inflammatory pathways, notably nuclear factor kappa B (NF-κB) and signal transducer and activator of transcription 3 (STAT3), leading to disruption of epithelial integrity and heightened cell proliferation. Dysbiosis compromises the intestinal barrier, allowing bacterial translocation and perpetuating a tumor-promoting environment.
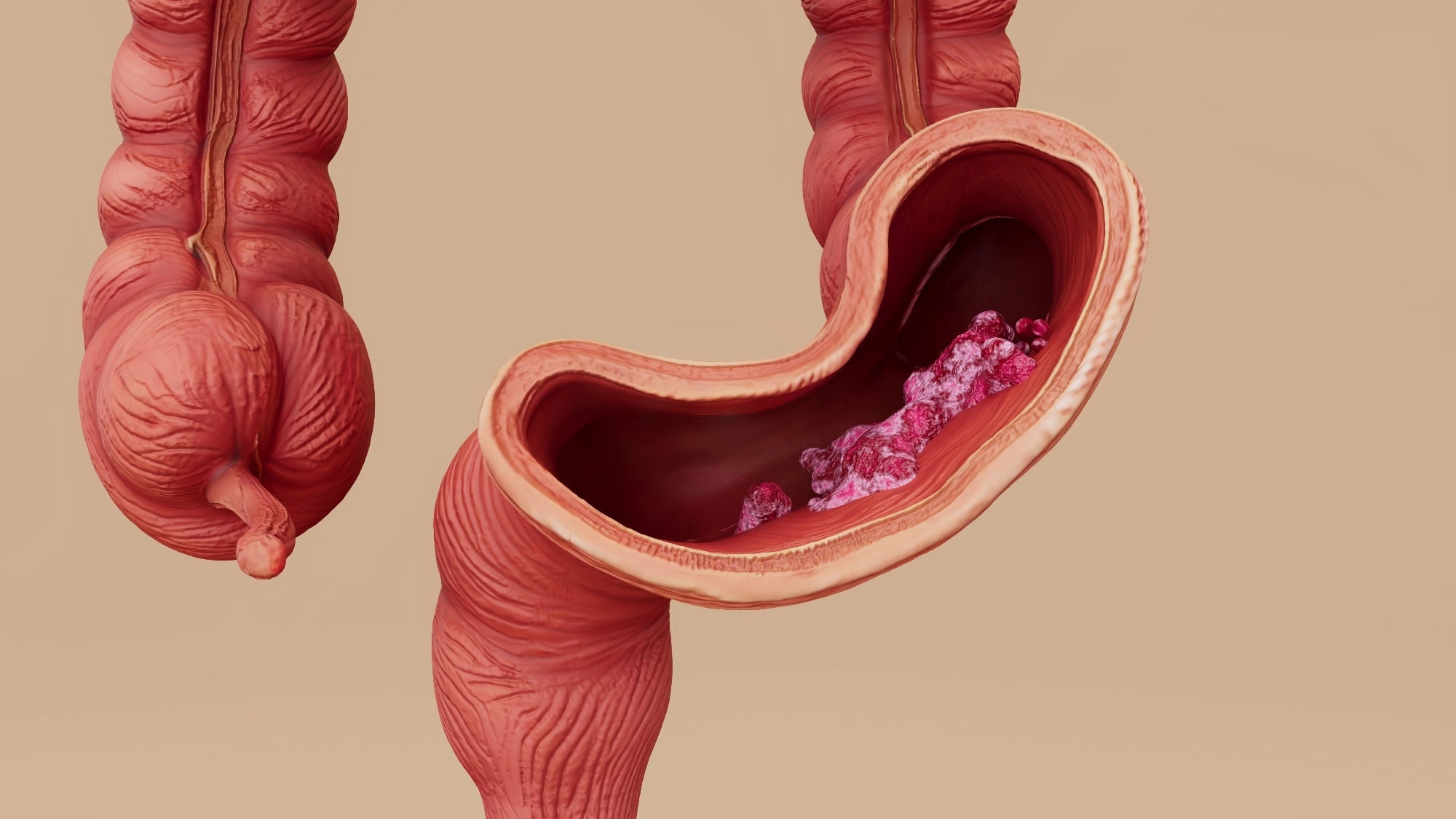
Microbial Dysbiosis and Colorectal Cancer
Research indicates that microbial dysbiosis contributes to colorectal cancer (CRC) by promoting a prolonged immune response that results in continuous epithelial injury. Specific strains of bacteria, including pathogenic Escherichia coli and Bacteroides fragilis, produce toxins that can induce DNA damage and genomic instability. For example, pks+ E. Coli generates colibactin, a genotoxin that damages DNA, while Fusobacterium nucleatum enhances tumorigenesis by activating oncogenic signaling pathways.
The presence of these bacteria not only correlates with cancer development but is also associated with distinct molecular subtypes of CRC, such as the CpG island methylator phenotype (CIMP) positivity and microsatellite instability (MSI).
Helicobacter pylori and Gastric Cancer
Among the various bacteria implicated in cancer, Helicobacter pylori stands out as a well-established risk factor for gastric cancer, classified as a Group I carcinogen. Long-term infection with H. Pylori leads to chronic gastritis and inflammation, resulting in oxidative DNA damage and an environment conducive to tumor development. This progression is characterized by precancerous changes, including atrophic gastritis and intestinal metaplasia, which heighten the risk of gastric adenocarcinoma.
H. Pylori manipulates host immune responses through virulence factors that disrupt key signaling pathways, promoting immune evasion and tumorigenesis.
Implications of Microbial Metabolites
Short-chain fatty acids (SCFAs), produced during the fermentation of dietary fibers by beneficial gut bacteria, play a protective role against cancer. SCFAs like acetate, propionate, and butyrate support epithelial health, reinforce barrier functions, and regulate immune responses. Butyrate, in particular, has demonstrated anti-tumor properties by promoting apoptosis in cancerous cells.
Conversely, dysbiosis alters bile acid metabolism, increasing the formation of secondary bile acids that can damage DNA and activate oncogenic pathways, further contributing to a pro-cancer environment.
Future Directions in Cancer Research
Recent advancements in metagenomics and metabolomics have opened avenues for identifying cancer-linked bacteria and metabolic signatures. These tools may facilitate early diagnosis and personalized treatment strategies. The composition of the gut microbiome has been linked to the efficacy of chemotherapy and immunotherapy, highlighting its potential impact on treatment outcomes.
Ongoing research is exploring various microbiome-targeted interventions, including probiotics, prebiotics, dietary fiber enrichment, and fecal microbiota transplantation (FMT), to restore microbial balance and enhance therapeutic efficacy.
As understanding of the gut microbiome’s role in gastrointestinal cancers continues to evolve, large-scale longitudinal studies will be essential to establish causal relationships and translate findings into clinical practice. Further exploration of host-microbe interactions will be crucial in developing comprehensive strategies for cancer prevention and treatment.
For more insights on this topic and related health matters, feel free to share your thoughts and comments below.
Disclaimer: This article is for informational purposes only and does not constitute medical advice.