“`html
MRI Reveals How Protective layer Forms in Stomach and Esophagus
Table of Contents
- 1. MRI Reveals How Protective layer Forms in Stomach and Esophagus
- 2. Understanding The Protective Mechanism
- 3. The Rise of Non-Invasive Gastrointestinal Monitoring
- 4. NeoBianacid and Mucosal Protection: A Closer Look
- 5. How does MRI confirm the integrity and targeted release of the protective coating in gastrointestinal therapy?
- 6. MRI Reveals the Protective Coating of a Natural Gastrointestinal Therapy in Humans
- 7. Understanding Targeted Gastrointestinal Therapies
- 8. The Role of MRI in Visualizing the Coating
- 9. What the MRI Scans Reveal: A Protective barrier in Action
- 10. Comparing MRI and CT Scans for Gastrointestinal Assessment
- 11. Benefits of This Novel Approach
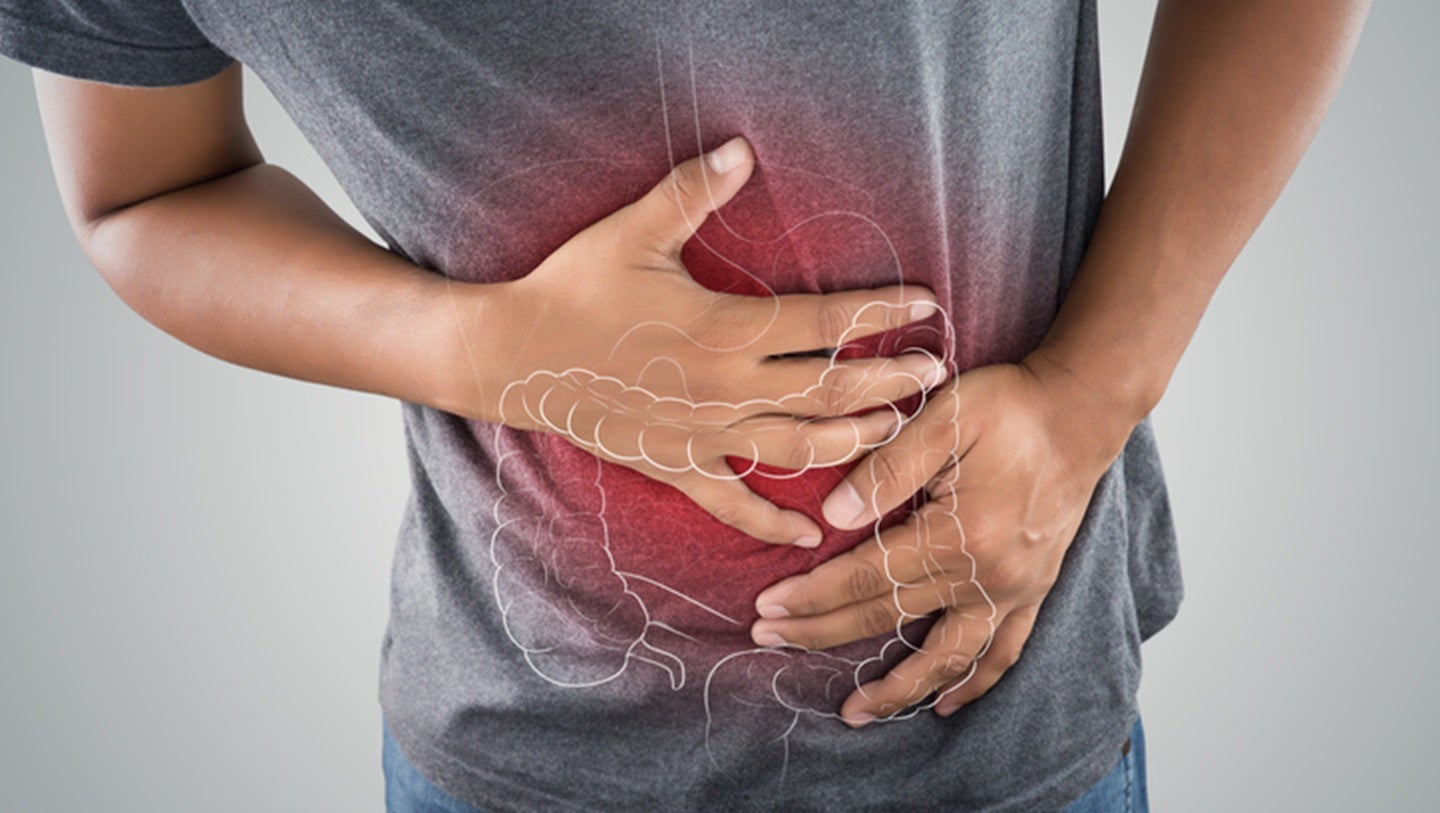
A Groundbreaking Study Utilizes Magnetic Resonance Imaging to Visualize The Formation of A Natural Medical device’s Protective Barrier In Vivo.
Researchers Are Pioneering A Non-Invasive Method To Monitor The Action Of NeoBianacid, A Medical Device Designed To Shield The Gastric And Esophageal mucosa. The Study, Conducted On Healthy Volunteers, aims To Confirm The Efficacy of MRI As A tool For Assessing Similar Therapies.
Understanding The Protective Mechanism
The Current Research Focuses On Demonstrating How Aboca’s Medical Device Creates its Protective Layer Within The Living Body. This Represents A Critically important Advance In Understanding How Natural Therapies Interact With the Human System.
Dr. Maura Corsetti, From The University Of Nottingham School Of Medicine, Highlighted The Novelty Of The Approach. “This Is The First Time That MRI Has Been Employed To Evaluate The Effects Of Coating A Natural Product On The Upper Gastrointestinal Tract,” She Stated. “It’s An Vital Step Towards Deeper Comprehension Of How Natural Treatments Function Inside The Human Body.”
The Rise of Non-Invasive Gastrointestinal Monitoring
Conventional Methods Of assessing Mucosal Protection Often Rely On Endoscopy, An Invasive Procedure.MRI Offers A Potential Option, Providing Detailed Images Without Requiring Physical Intervention. This Advancement Could Streamline Research And Development In The field Of Gastrointestinal Health.
According To A 2023 Report by Global Market Insights, The global Gastrointestinal Imaging Market Is Projected To Exceed $4.5 Billion By 2032, Driven By increasing Prevalence Of Gastrointestinal Disorders And Technological Advancements In Imaging techniques.
NeoBianacid and Mucosal Protection: A Closer Look
NeoBianacid Is A Medical Device Composed Of Polysaccharides And Mineral Salts, Designed To Create A Protective Film Over Inflamed Mucosa In The Esophagus, Stomach, And Duodenum. This Barrier Action Helps To Relieve Symptoms Associated With Acid Reflux, Gastritis, And esophagitis.
| Feature | NeoBianacid |
How does MRI confirm the integrity and targeted release of the protective coating in gastrointestinal therapy?
MRI Reveals the Protective Coating of a Natural Gastrointestinal Therapy in HumansThe field of gastroenterology is constantly evolving, with researchers seeking innovative ways to deliver targeted therapies for conditions like inflammatory bowel disease (IBD), ulcers, and even simple indigestion. A recent breakthrough, visualized through Magnetic Resonance Imaging (MRI), demonstrates the efficacy of a novel approach: a natural gastrointestinal therapy encapsulated within a protective coating, ensuring targeted delivery and maximized benefit. This article delves into the science behind this technology,the MRI findings,and what it means for the future of gut health. Understanding Targeted Gastrointestinal TherapiesTraditional oral medications for gastrointestinal issues often face significant challenges.Stomach acid, digestive enzymes, and the sheer length of the digestive tract can degrade the active ingredients before they reach the affected area.This leads to lower bioavailability – meaning less of the medication actually gets absorbed and utilized by the body. Targeted therapies aim to overcome these hurdles by: * Protecting the Active Ingredient: Shielding the therapeutic compound from harsh digestive conditions. * Controlled Release: Delivering the medication directly to the site of inflammation or damage. * Enhanced Absorption: Increasing the amount of active ingredient that reaches the intestinal lining. The Role of MRI in Visualizing the CoatingWhile the concept of targeted drug delivery isn’t new, visualizing the protective coating and its behavior within the human gastrointestinal tract has been a significant challenge. This is where MRI technology proves invaluable. unlike X-rays or CT scans,MRI doesn’t use ionizing radiation,making it a safer option for repeated scans. Furthermore, advanced MRI techniques allow us to differentiate between various tissue types and even detect subtle changes in molecular structure. Specifically, researchers are utilizing specialized MRI contrast agents that interact with the coating material. These agents highlight the coating on the MRI images, allowing scientists to track its journey through the digestive system. This provides crucial data on: * coating Integrity: Assessing whether the coating remains intact during its passage through the stomach and intestines. * Release Mechanism: Observing when and where the coating begins to break down, releasing the therapeutic agent. * Distribution Pattern: Mapping the spread of the therapy within the gut. What the MRI Scans Reveal: A Protective barrier in ActionRecent studies utilizing this MRI technique have focused on a naturally derived gastrointestinal therapy – a complex blend of plant extracts known for their anti-inflammatory and mucosal-healing properties. The therapy is encapsulated in a biocompatible polymer coating designed to resist degradation in the stomach’s acidic environment. MRI scans have consistently demonstrated:
Comparing MRI and CT Scans for Gastrointestinal AssessmentIt’s important to understand why MRI is preferred over other imaging modalities like CT scans in this context.As highlighted in recent discussions [https://www.zhihu.com/question/27891489], CT scans excel at visualizing bone structures and detecting acute bleeding, while MRI provides superior soft tissue contrast. This makes MRI ideal for assessing inflammation, mucosal damage, and the behavior of targeted therapies within the gut.CT scans also involve radiation exposure, a factor that weighs against their use in repeated monitoring. Benefits of This Novel ApproachThe ability to visualize and confirm the effectiveness of this protective coating through MRI offers several key benefits: * Improved Therapeutic Efficacy: By ensuring targeted delivery, the therapy can exert its effects more effectively, potentially reducing symptom severity and promoting faster healing. * Reduced Side Effects: Minimizing systemic absorption lowers the risk of unwanted side effects associated with higher drug concentrations in the bloodstream. * Personalized Treatment: MRI can be used to tailor the coating’s release mechanism to individual patient needs, optimizing therapeutic outcomes. * **Enhanced Understanding of Gut Breaking: Zylox-tonbridge to acquire Optimed in a strategic push to accelerate global expansionTable of Contents
Zylox-Tonbridge, the Hong Kong-listed group focused on neurovascular and peripheral vascular interventional devices, has agreed to acquire Optimed, a German medical technology company.The deal unfolds in multiple steps and includes an option to purchase the entire shareholding from current owners. The transaction aims to create a unified platform spanning research and growth, manufacturing, and commercialisation. Under the arrangement, Zylox-Tonbridge will progressively acquire Optimed’s equity interests, with a final option to take full control. After closing, the two businesses will merge their sales, marketing, and customer service functions to form a single global commercial organisation. Key leadership details were confirmed: Optimed’s chief executive, Rüdiger Hausherr, will continue to lead Optimed and report to Zylox-Tonbridge chairman and chief executive Dr. Jonathon Zhong Zhao. Dr. Zhao described the acquisition as a pivotal milestone for Zylox-Tonbridge’s growth plan, enabling the group to tap a broader European-based platform to speed its international expansion. He welcomed Optimed to the group and highlighted plans to combine complementary product portfolios and manufacturing capabilities to deliver high-quality,innovative,and affordable solutions for more patients and clinicians worldwide. What the deal coversThe agreement contemplates a staged purchase with an option to acquire the entire stake from current owners. The intention is to integrate Optimed’s European footprint with Zylox-tonbridge’s core strengths to create a seamless global operation. Why it mattersThe merger targets faster market access and a broader manufacturing base, especially in Germany, to support European and global supply of vascular devices. By aligning sales networks and leveraging Optimed’s established relationships with European clinical experts, the combined group aims to reach more customers with speed and efficiency. Company profiles at a glanceOptimed has spent nearly three decades developing, producing, and distributing devices for minimally invasive therapies, with a presence in more than 70 countries. Its venous stenting portfolio addresses the iliofemoral veins, vena cava, and iliac bifurcation segments.Clinical evidence from trials such as sinus-Venous and STEVECO supports improvements in quality of life and clinical severity versus conservative care. Zylox-Tonbridge specializes in neurovascular and peripheral interventional products and seeks to expand its global reach through a unified platform that combines R&D, manufacturing, and commercial operations. Operational and leadership implicationsPost-transaction, the merged entity will consolidate its sales, marketing, and customer-service teams under a single global umbrella. Optimed’s leadership will continue to guide the German unit, reporting to Zylox-Tonbridge’s top executive. Industry context and potential impactCross-border consolidation in the medical device sector continues to reshape how products reach patients, notably in Europe where local manufacturing and clinical expertise drive market access. The combined platform is expected to generate operational synergies through a broader German manufacturing base, underpinning stable supply for European and international markets. For readers seeking context on medical-device regulation and market oversight, see the FDA’s overview of medical devices and international health guidance from the World Health Institution. For reference on regulatory and industry standards: Key facts at a glance
What readers are saying: your take matters1) Do you think the expanded European presence will lower device lead times for patients in the region? 2) How might this deal influence pricing and access to venous-stenting solutions in emerging markets? share your thoughts in the comments below and join the discussion.
.Zylox‑Tonbridge Acquisition Overview
Strategic Rationale
Impact on Peripheral Venous Stenting Market
Product Portfolio Integration
Geographic Expansion and Market Access
Regulatory and Compliance Considerations
Benefits for Healthcare Providers
Practical Implementation Tips for Hospitals
Case Study: Early Adoption in Germany (2026 Q2)
Future Outlook
Beyond Ozempic: Could a One-Time Procedure ‘Reset’ Your Metabolism for Lasting Weight Loss?For decades, the weight loss industry has been a revolving door of diets and exercise fads, often yielding temporary results followed by frustrating rebounds. Now, a new frontier is emerging – one that moves beyond daily pills and injections, offering the potential for a more permanent metabolic shift. The global obesity rate continues to climb, with nearly 40% of adults worldwide classified as obese, fueling a desperate search for solutions that truly last. The GLP-1 Dilemma and the Search for AlternativesDrugs like Wegovy and Zepbound have undeniably revolutionized weight loss, demonstrating unprecedented efficacy. However, their high cost and the need for ongoing administration put them out of reach for many. This accessibility gap has spurred innovation in less expensive, longer-lasting treatments. Enter a wave of companies developing medical devices designed to address the root causes of weight regain after discontinuing GLP-1 medications – and potentially even offer a standalone solution. Rewiring the Gut: Fractyl Health’s Pioneering ApproachFractyl Health, based in Massachusetts, is leading the charge with a minimally invasive endoscopic procedure targeting the duodenum, the first part of the small intestine. The premise is simple, yet profound: years of a high-sugar, high-fat diet can inflame and distort the duodenum’s signaling pathways, locking the body into a pattern of cravings, weight gain, and insulin resistance. Fractyl’s device uses a controlled application of heat – near-boiling water – to gently injure and then allow the intestinal lining to regenerate, effectively “resetting” metabolic control. “You see regrowth at about two weeks, and it continues until the mucosa looks pretty normal,” explains Alan Cherrington, a physiologist at Vanderbilt University School of Medicine who consults for Fractyl. This isn’t about physically altering the gut’s structure, but rather restoring its proper function. Promising Early Results: The Remain-1 StudyPreliminary data from the Remain-1 clinical trial are encouraging. Participants who underwent the Fractyl procedure after stopping Zepbound generally maintained their weight loss, while those receiving a sham treatment experienced weight regain. While the trial is small and long-term data is still needed, the initial results suggest the procedure can stabilize weight and potentially even promote continued loss. Full trial readouts are expected in 2026, paving the way for potential FDA clearance. The Competitive Landscape: Beyond FractylFractyl isn’t alone in this space. Several companies are pursuing similar strategies, each with a unique technological approach. Endogenex utilizes pulsed electric fields, TeCure employs lasers, and Aqua Medical uses radiofrequency-heated water vapor – all aimed at remodeling the duodenal lining. Pichamol Jirapinyo, a bariatric endoscopist at Brigham and Women’s Hospital, notes that operational factors like ease of use and procedure time will likely be key differentiators. Other approaches target the stomach, with existing procedures like stomach folding and more experimental methods aimed at altering appetite-regulating hormones. However, Fractyl’s focus on the duodenum offers a potential marketing advantage, as it could be the first device specifically approved for post-GLP-1 weight maintenance. Duodenum vs. Stomach: A Matter of Complexity?While stomach-based procedures are more established, some experts raise concerns about the duodenum’s delicate nature. Andrew Storm, a therapeutic endoscopist at Wake Forest University, points out that the duodenum is “paper thin” compared to the stomach, potentially increasing the complexity of the procedure and impacting scalability. The Patient Perspective: A Glimmer of HopeFor patients like Christina, who participated in the Remain-1 trial, the potential benefits are deeply personal. After losing 50 pounds on Zepbound, she’s confident the Fractyl procedure has helped her maintain that loss, even experiencing a diminished desire for previously irresistible foods. While the trial is blinded, her experience offers a compelling glimpse into the potential of these new therapies. Looking Ahead: A New Era of Obesity Treatment?The next year will be critical. The six-month data from the Remain-1 trial will determine whether these duodenal therapies can deliver on their promise of lasting weight maintenance. If successful, this could unlock a new era of obesity care, offering a viable alternative to lifelong medication or invasive surgery. The convergence of pharmaceutical and device-based solutions is poised to reshape the landscape of weight management, offering hope for millions struggling with obesity and its associated health risks. Research from the National Institutes of Health highlights the growing need for innovative obesity treatments. What are your thoughts on the future of metabolic resetting? Share your predictions in the comments below! Breakthroughs in Biomedical Tech Merge AI, Wearables and Autonomybreaking advances in biomedicine fuse artificial intelligence with lasting tools, giving clinicians faster reads on brain health, noninvasive wearables, and autonomous robotic support in the operating room. In 2025, researchers showcased real‑time brain monitoring, graphene‑based sensors, and smarter surgical assistance that could reshape patient care for years to come. Real‑Time Brain Monitoring Aims to Predict RelapseA clinic‑scale effort is turning implanted brain signals into early warnings. Doctors detected relapse cues in patients with mood disorders by watching electrical activity in real time, enabling earlier intervention. The approach relies on implants that record neural impulses and AI to flag changes before symptoms become obvious. Clinicians say there are many levers to adjust, including when and how to alert caregivers or clinicians. In one case, a psychiatrist observed signals from a patient’s brain implants that indicated worsening depression even before the patient noticed anything, underscoring how neurotechnology could shift timing in mental‑health care. Ultra‑Thin Graphene Tattoos Track Vital SignsResearchers are developing imperceptible graphene “tattoos” that can monitor vital signs and other biomarkers. The graphene,a strong and flexible conductor,could measure heart rate and chemical markers in sweat. While the prototypes still require a connection to a circuit, the goal is seamless integration with everyday wearables such as smartwatches. Low‑Cost, Contactless Heart monitoring Via Wi‑FiA novel method uses standard Wi‑Fi signals to estimate heart rate in real time, without direct contact. The system analyzes heartbeat patterns from distances up to several feet, offering a low‑cost option that works across postures and environments. The team aims to bring this technology to broader markets, possibly expanding remote health monitoring beyond conventional sensors. Ultrasound and Focused Ultrasound: A Potential Neuromodulation ToolResearchers are exploring how focused ultrasound could gently stimulate neurons to address inflammation or metabolic diseases. By vibrating a neuron’s membrane, ultrasound may open ion channels and influence cell activity in a targeted way. Experts say this could offer a safe path to non‑drug therapies for a range of conditions, including inflammatory states and diabetes. Laser‑Based Brain Imaging Advancesfor years, peering inside the head faced a choice between inexpensive, shallow methods and costly deep imaging. A new line of experiments shows lasers can deliver photons through the skull, potentially enabling deeper, noninvasive brain access. Project leads suggest this breakthrough could inspire a new generation of diagnostic and therapeutic devices. Autonomous Surgical Robots Move Toward realityIn robotics labs, researchers are refining autonomous tools to assist surgeons during complex procedures. the aim is a future were patients are greeted by a surgeon and an autonomous robotic assistant, with autonomous systems handling precise soft‑tissue tasks under human supervision. while challenges remain-such as controller design and privacy considerations-the trajectory points toward more capable, collaborative operating rooms. Key Players Behind These InnovationsHealth researchers and engineers across the united States are leading these efforts, including teams focused on neural implants, graphene sensors, wireless health sensing, ultrasound neuromodulation, advanced imaging with lasers, and autonomous surgical robotics. Cross‑disciplinary collaboration remains essential to translate lab breakthroughs into clinical practice.
What This Means for Patients and ProvidersThese developments do not replace clinicians; they augment decision‑making, increase data granularity, and may reduce invasive procedures over time. Regulators and researchers alike are weighing safety, privacy and efficacy as trials expand. Industry observers expect a phased rollout, with noninvasive and monitoring tools leading the way, followed by more integrated implants and robotic assistants as evidence accumulates. Evergreen OutlookEven as AI drives rapid progress, long‑term success will hinge on robust validation, transparent data governance, and clear patient‑centered safeguards. The convergence of neural interfaces, noninvasive sensing, and autonomous systems could redefine preventive care, chronic disease management, and surgical workflows in the next decade. Key ResourcesFor readers seeking broader context,official health authorities and research institutions offer foundational guidance on neuromodulation,wearables,and medical device regulation. See sources from established health and standards organizations for evolving policies and standards. External references: NIH • FDA • Nature Disclaimer: Biomedical innovations involve regulatory review,patient consent,and ongoing safety assessments. Information cited here reflects current research progress and may evolve as new data emerge. Join the ConversationWhat are your priorities for approving AI‑assisted medical devices? Do you support broader data sharing to improve care, or should privacy safeguards take precedence? Share your views and questions in the comments below. Two fast questions for readers:
Disclaimer: This report summarizes ongoing research and does not constitute medical advice. Seek professional guidance for medical decisions. Share this story and tell us what frontier you’re most eager to see move from the lab to the clinic. Newer Posts Adblock Detected |
|---|