A new study offers a potential breakthrough in the early detection of Alzheimer’s disease (AD). Researchers have identified a protein, PPP2R5C, found in blood, that appears to decline in individuals years before the onset of clinical symptoms. This discovery, published in the journal Cell Reports Medicine, could pave the way for earlier diagnosis and intervention in a disease that currently lacks readily available, minimally invasive early detection methods.
Alzheimer’s disease, the most common form of dementia, affects millions worldwide. The hallmark of AD is the progressive deterioration of memory and cognitive function, driven by the accumulation of abnormal protein deposits in the brain. However, these pathological changes begin decades before symptoms manifest, highlighting the critical need for biomarkers that can identify the disease in its earliest stages – when interventions may be most effective. Current diagnostic tools, such as PET scans and cerebrospinal fluid analysis, are often expensive, invasive, and not widely accessible.
PPP2R5C: A Potential Early Indicator
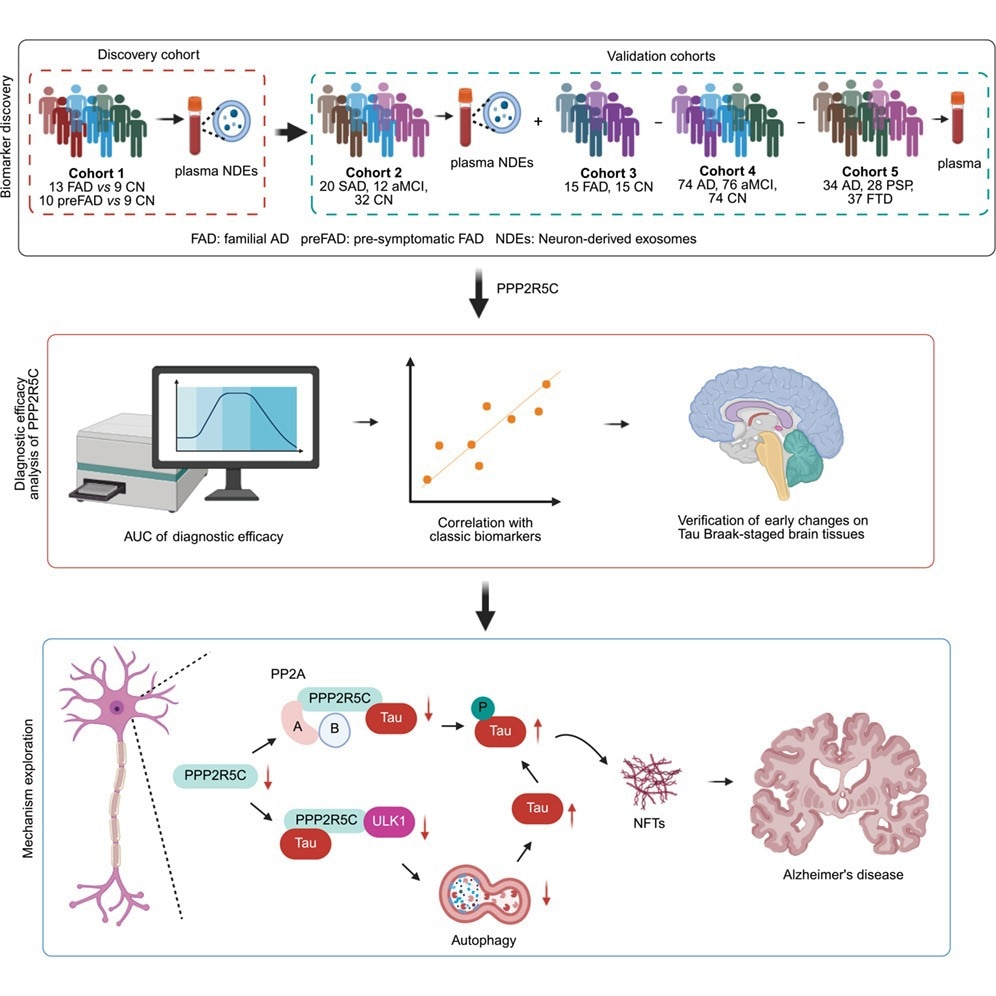
The research team focused on neuron-derived exosomes (NDEs) – tiny vesicles released by brain cells that circulate in the bloodstream. By analyzing NDEs from individuals with familial AD, presymptomatic familial AD, and healthy controls, they discovered that levels of a specific peptide from the protein PPP2R5C progressively decreased as the disease progressed. This initial finding was then validated in a larger cohort including individuals with amnestic mild cognitive impairment (aMCI) and sporadic AD, confirming the association between reduced PPP2R5C levels and early Alzheimer’s pathology. The study, published on February 19, 2026, suggests that measuring PPP2R5C in a simple blood test could offer a less burdensome way to screen for the disease. Read the study in PubMed.
How PPP2R5C Impacts Tau Regulation
The researchers also investigated the underlying mechanisms by which PPP2R5C might be linked to Alzheimer’s disease. A key feature of AD is the abnormal phosphorylation of the tau protein, leading to the formation of neurofibrillary tangles that disrupt brain function. The study revealed that PPP2R5C interacts with tau, reducing its levels and phosphorylation through activation of PP2A, an enzyme that regulates tau, and a process called autophagy – the cell’s way of clearing out damaged proteins. Specifically, the research points to the involvement of the ULK1 complex in this autophagolysosomal pathway. Lower PPP2R5C levels were correlated with increased phosphorylation of ULK1, suggesting a regulatory relationship.
Study Findings and Biomarker Performance
To assess the feasibility of using PPP2R5C as a biomarker, researchers analyzed total plasma PPP2R5C levels in several cohorts. They found that plasma PPP2R5C levels were approximately 61.3% lower in individuals with aMCI and 31.6% lower in those with AD compared to cognitively normal controls. The biomarker demonstrated a good ability to distinguish AD from controls, with an area under the receiver operating characteristic curve (AUROC) of 0.8494. However, its ability to differentiate between aMCI and AD was more limited (AUROC of 0.5931). Notably, PPP2R5C levels were positively associated with scores on the Mini-Mental State Examination (MMSE), a common cognitive assessment tool, and negatively correlated with levels of phosphorylated tau in the blood. Learn more about the study in Cell Reports Medicine.
Postmortem brain analyses further supported the findings, showing lower PPP2R5C levels in individuals with AD compared to cognitively normal individuals. Importantly, the reduction in PPP2R5C expression appeared to occur as early as Braak stage II, a relatively early stage of Alzheimer’s pathology characterized by limited neurofibrillary tangle formation.
Future Directions and Validation
While these findings are promising, researchers emphasize that PPP2R5C is not yet a definitive diagnostic marker for Alzheimer’s disease. Larger, longitudinal studies – following individuals over time – and studies that include more diverse populations are needed to validate these results. Standardization of the assay used to measure PPP2R5C will also be crucial before it can be implemented in routine clinical practice. Further research is needed to fully understand the complex interplay between PPP2R5C, tau regulation, and the development of Alzheimer’s disease.
The identification of PPP2R5C as a potential biomarker represents a significant step forward in the fight against Alzheimer’s disease. If validated in larger studies, a simple blood test based on PPP2R5C levels could revolutionize early detection, allowing for earlier intervention and potentially slowing the progression of this devastating disease.
Disclaimer: This article provides information for general knowledge and informational purposes only, and does not constitute medical advice. It is essential to consult with a qualified healthcare professional for any health concerns or before making any decisions related to your health or treatment.
What are your thoughts on this potential new biomarker? Share your comments below, and please share this article with anyone who might find it helpful.