Former NFL quarterback Tony Romo’s recent reflections on his father’s prostate cancer journey, in partnership with Bayer, highlight the importance of proactive screening, open communication with physicians, and available treatment options like darolutamide (NUBEQA). This story underscores the prevalence of prostate cancer – the most common cancer in American men – and the require for informed patient engagement.
Prostate cancer affects men globally, often presenting with subtle or no symptoms in its early stages. The emotional toll on families, as illustrated by the Romo’s experience, is significant. This article will delve into the clinical aspects of prostate cancer, the role of NUBEQA in advanced disease management, and the importance of understanding individual risk factors and treatment pathways. We will as well explore the broader public health implications and access to care.
In Plain English: The Clinical Takeaway
- Prostate cancer is common, especially with age. Regular checkups and discussing your risk with your doctor are crucial.
- New treatments are available. Medications like NUBEQA can aid manage advanced prostate cancer, but they arrive with potential side effects that need careful consideration.
- Open communication is key. Talking to your family and healthcare team about your concerns and treatment options is vital for navigating this journey.
Understanding Prostate Cancer: Epidemiology and Risk Factors
Prostate cancer is estimated to account for 29% of all cancer diagnoses in men in 2024, with approximately 299,010 new cases expected in the United States alone. Globally, incidence rates vary significantly, with higher rates observed in North America, Europe, Australia, and the Caribbean. This variation is likely due to a combination of genetic predisposition, lifestyle factors, and screening practices. The American Cancer Society estimates that about 1 in 8 men will be diagnosed with prostate cancer during their lifetime. Key risk factors include age (risk increases dramatically after age 50), family history of prostate cancer (particularly in first-degree relatives), and race/ethnicity (African American men have a higher risk of developing and dying from prostate cancer).
Darolutamide (NUBEQA): Mechanism of Action and Clinical Evidence
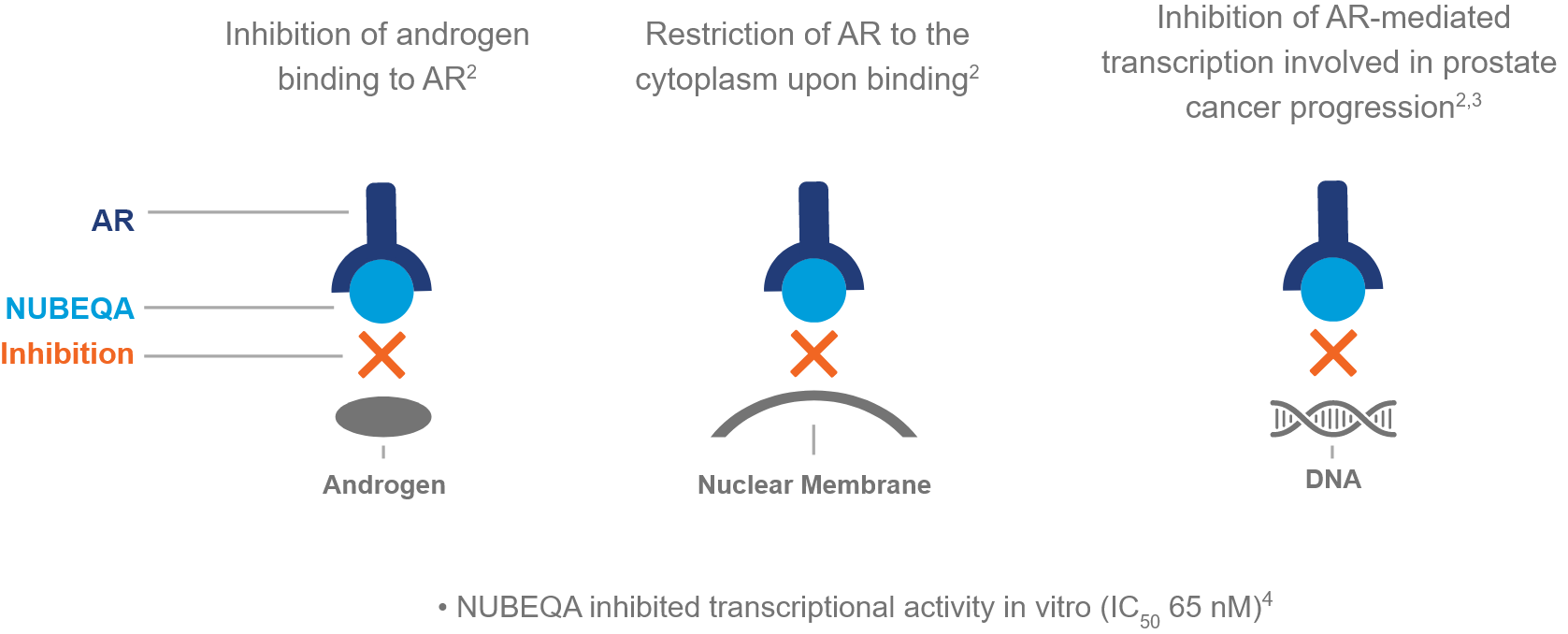
Darolutamide, marketed as NUBEQA by Bayer, is an androgen receptor inhibitor (ARI). Androgens, such as testosterone, fuel the growth of prostate cancer cells. NUBEQA works by binding to the androgen receptor, preventing testosterone from activating it and thus slowing or stopping cancer cell growth. This is a crucial mechanism, as even with androgen deprivation therapy (ADT) – a common initial treatment – some androgen activity can persist, driving disease progression.
The approval of NUBEQA was based on the Phase III ARAMIS trial, a randomized, double-blind, placebo-controlled study involving 1,509 men with non-metastatic castration-resistant prostate cancer (nmCRPC). The trial demonstrated a statistically significant improvement in metastasis-free survival (MFS) with NUBEQA compared to placebo (40.4 months vs. 16.2 months, hazard ratio 0.41). Subsequent analyses have shown benefits across various subgroups, including patients with different Gleason scores and PSA levels. The ARAMIS trial also showed a statistically significant improvement in overall survival, although this benefit emerged later in the study.
Geographical Access and Regulatory Landscape
NUBEQA is currently approved in the United States by the Food and Drug Administration (FDA), in the European Union by the European Medicines Agency (EMA), and in several other countries worldwide. However, access to NUBEQA can vary significantly depending on national healthcare systems and reimbursement policies. In the US, Medicare and most private insurance plans typically cover NUBEQA, but patients may be subject to co-pays and deductibles. In Europe, reimbursement decisions are made at the national level, leading to variations in access across different countries. The National Health Service (NHS) in the UK has also approved NUBEQA, but access may be subject to clinical guidelines and budget constraints.
Funding and Bias Transparency
The ARAMIS trial, which formed the basis for NUBEQA’s approval, was funded by Bayer. It is important to acknowledge this funding source, as it may introduce potential bias. However, the ARAMIS trial was rigorously designed and conducted, and the results were published in a peer-reviewed journal (The New England Journal of Medicine). The FDA and EMA conducted independent reviews of the data before granting approval.
“The ARAMIS trial represents a significant advance in the treatment of non-metastatic castration-resistant prostate cancer. Darolutamide has demonstrated a clear benefit in terms of metastasis-free survival and overall survival, and it is now an important option for patients with this disease.” – Dr. Karim Fizazi, Principal Investigator of the ARAMIS trial, Gustave Roussy Cancer Campus, Villejuif, France.
Data Visualization: ARAMIS Trial Demographics and Outcomes
| Characteristic | NUBEQA (n=752) | Placebo (n=757) |
|---|---|---|
| Median Age (years) | 68 | 68 |
| Metastasis-Free Survival (months) | 40.4 | 16.2 |
| Hazard Ratio (MFS) | 0.41 | – |
| Overall Survival (months) | 29.4 | 24.3 |
| Hazard Ratio (OS) | 0.59 | – |
Contraindications & When to Consult a Doctor
NUBEQA is contraindicated in females and children. It should not be used in individuals with known hypersensitivity to darolutamide or any of its excipients. Common side effects include fatigue, pain, diarrhea, nausea, cough, and rash. More serious side effects, such as heart failure and seizures, have been reported in clinical trials. Patients should immediately consult a doctor if they experience any of these symptoms. Individuals with pre-existing heart conditions, high blood pressure, or a history of seizures should discuss the risks and benefits of NUBEQA with their healthcare provider before starting treatment. NUBEQA can interact with other medications, so it is crucial to inform your doctor about all the medicines you are taking.
The Romo family’s openness about their experience with prostate cancer serves as a powerful reminder of the importance of early detection and proactive management. While NUBEQA represents a significant advancement in treatment, it is not a cure. Ongoing research is focused on developing even more effective therapies and improving the quality of life for men with prostate cancer. The future of prostate cancer treatment lies in personalized medicine, tailoring treatment strategies to the individual characteristics of each patient’s disease.
References
- American Cancer Society. Prostate Cancer Key Statistics.
- National Cancer Institute. Prostate Cancer Stat Facts.
- Fizazi K, et al. Darolutamide in Non-Metastatic, Castration-Resistant Prostate Cancer. N Engl J Med. 2019;380(23):2222-2233.
- FDA. NUBEQA (darolutamide).
- National Institute of Diabetes and Digestive and Kidney Diseases. Prostate Cancer.

