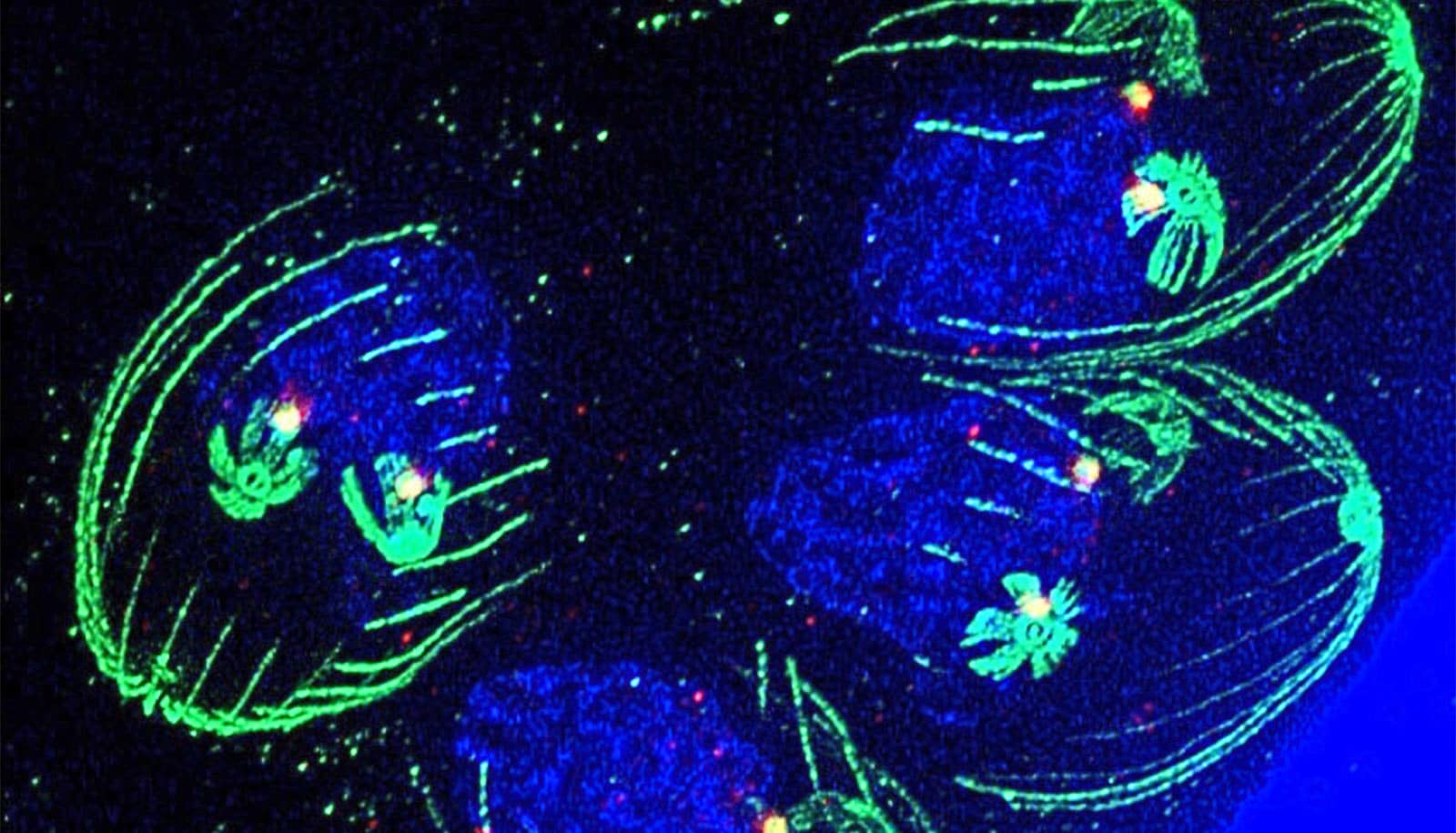
Researchers at USF Health have successfully mapped the cell cycle of Toxoplasma gondii using advanced fluorescent imaging. This breakthrough reveals a unique “forked” growth pattern in the parasite, offering critical new targets for developing drugs to treat chronic infections that currently remain incurable.
The implications of this discovery extend far beyond the microscope. For the estimated one-third of the global population carrying this latent infection, the transition from an acute, treatable phase to a chronic, dormant phase represents a significant medical blind spot. By visualizing the precise molecular mechanics of how this parasite replicates, we move closer to therapies that can eradicate the cysts hiding in the brain, rather than merely suppressing symptoms.
In Plain English: The Clinical Takeaway
- New Visibility: Scientists can now watch the parasite grow in real-time using glowing markers, something previously impossible due to its microscopic size.
- Unique Biology: Unlike human cells that divide in a straight line, this parasite branches out like a fork, allowing it to multiply rapidly and evade detection.
- Treatment Hope: Understanding this “forked” cycle identifies weak points where future medications could stop the parasite from forming permanent, untreatable cysts.
Decoding the “Forked” Cell Cycle Mechanism
The core of this advancement lies in the adaptation of fluorescent imaging technology. Historically, studying Toxoplasma gondii was akin to navigating a dark room; the organism is too little for standard observation, and its protein markers differ significantly from human cells. The research team, led by investigators at the USF Health Morsani College of Medicine, overcame this by identifying a specific protein, PCNA1, located in the parasite’s nucleus.
By attaching a neon green fluorescent tag to PCNA1, the team created a visual roadmap of the parasite’s life cycle. The data revealed a deviation from the standard eukaryotic cell cycle. While human cells typically grow, copy DNA, and split sequentially, Toxoplasma exhibits a overlapping, branching structure. As described by the research team, the cycle begins with a single “handle” before branching into multiple “prongs.” This allows up to three cell cycle phases to occur simultaneously.
This biological efficiency explains the parasite’s virulence. The ability to overlap growth phases enables rapid multiplication during the acute stage, overwhelming the host’s initial immune response before the organism retreats into a dormant state. This dormancy, characterized by the formation of tissue cysts, is the primary barrier to a cure. Current antiparasitic drugs, such as pyrimethamine and sulfadiazine, target the active replication phase but are ineffective against the dormant bradyzoites within the cysts.
Public Health Implications and Regulatory Pathways
The epidemiological burden of toxoplasmosis is often underestimated in public discourse. While often asymptomatic in healthy adults, the stakes are critically high for specific demographics. The Centers for Disease Control and Prevention (CDC) notes that congenital toxoplasmosis can lead to severe neurological sequelae, including hydrocephalus and chorioretinitis, if transmission occurs during pregnancy.
From a regulatory perspective, this research aligns with the FDA and EMA’s increasing focus on “neglected tropical diseases” and antimicrobial resistance. The current standard of care relies on older antifolate drugs that carry significant toxicity profiles, including bone marrow suppression and hypersensitivity reactions. A therapy targeting the specific “forked” replication mechanism identified in this study could offer a safer pharmacokinetic profile.
“Toxoplasmosis remains a significant public health concern, particularly for immunocompromised individuals and pregnant women. Advances in understanding the fundamental biology of the parasite are essential for developing the next generation of targeted therapeutics that can penetrate the blood-brain barrier.” — Consensus Statement, Centers for Disease Control and Prevention (CDC)
the geographic distribution of the parasite varies, with higher seroprevalence rates observed in Latin America and parts of Continental Europe compared to North America and Asia. This geo-epidemiological variance suggests that environmental factors and food safety protocols play a massive role in transmission. Any new treatment developed from this imaging data would need to be accessible globally, not just in high-income healthcare systems.
Clinical Staging and Therapeutic Targets
The distinction between the acute and chronic stages of toxoplasmosis is vital for clinical management. The acute stage involves tachyzoites, the rapidly dividing form that causes initial infection symptoms. The chronic stage involves bradyzoites, which form cysts primarily in neural and muscle tissue. The fluorescent imaging study specifically targets the transition point between these stages.
By mapping the PCNA1 protein shifts, researchers can now identify the exact molecular window where the parasite commits to cyst formation. Interrupting this process is the “holy grail” of toxoplasmosis research. If a drug can block the branching mechanism during the cell cycle, it could prevent the establishment of chronic latency entirely.
| Feature | Acute Stage (Tachyzoites) | Chronic Stage (Bradyzoites) |
|---|---|---|
| Replication Rate | Rapid (Overlapping cell cycles) | Slow / Dormant |
| Location | Bloodstream and various tissues | Encysted in brain and muscle tissue |
| Current Treatment Efficacy | High (Pyrimethamine/Sulfadiazine) | None (No known cure for cysts) |
| Immune Evasion | High multiplication overwhelms immunity | Hides within cysts, evading detection |
Contraindications & When to Consult a Doctor
While this research offers hope for future pharmaceuticals, current management relies on existing protocols. Patients should be aware of the following clinical guidelines:
- Pregnancy: Pregnant women should avoid changing cat litter and consuming undercooked meat. If exposure is suspected, immediate serological testing is required. Do not wait for symptoms.
- Immunocompromised Status: Individuals with HIV/AIDS or those undergoing chemotherapy are at risk for reactivation of latent cysts, leading to toxoplasmic encephalitis. Any new neurological symptom warrants immediate MRI and consultation.
- Medication Toxicity: Current treatments can cause folate deficiency. Patients on long-term suppressive therapy require regular blood count monitoring to prevent bone marrow toxicity.
The path from fluorescent imaging to a market-ready drug is long, often spanning a decade of clinical trials. However, the ability to visualize the enemy’s strategy is the first decisive victory in this conflict. As the research team continues to test compounds against these newly mapped growth stages, the medical community remains cautiously optimistic that the “forked” path of Toxoplasma may finally meet its dead end.
References
- Centers for Disease Control and Prevention. Toxoplasmosis.
- Batra, M., et al. “Fluorescent imaging reveals cell cycle dynamics in Toxoplasma gondii.” mBio.
- World Health Organization. Toxoplasmosis Fact Sheet.
- National Library of Medicine. PubMed Database.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider for diagnosis and treatment.