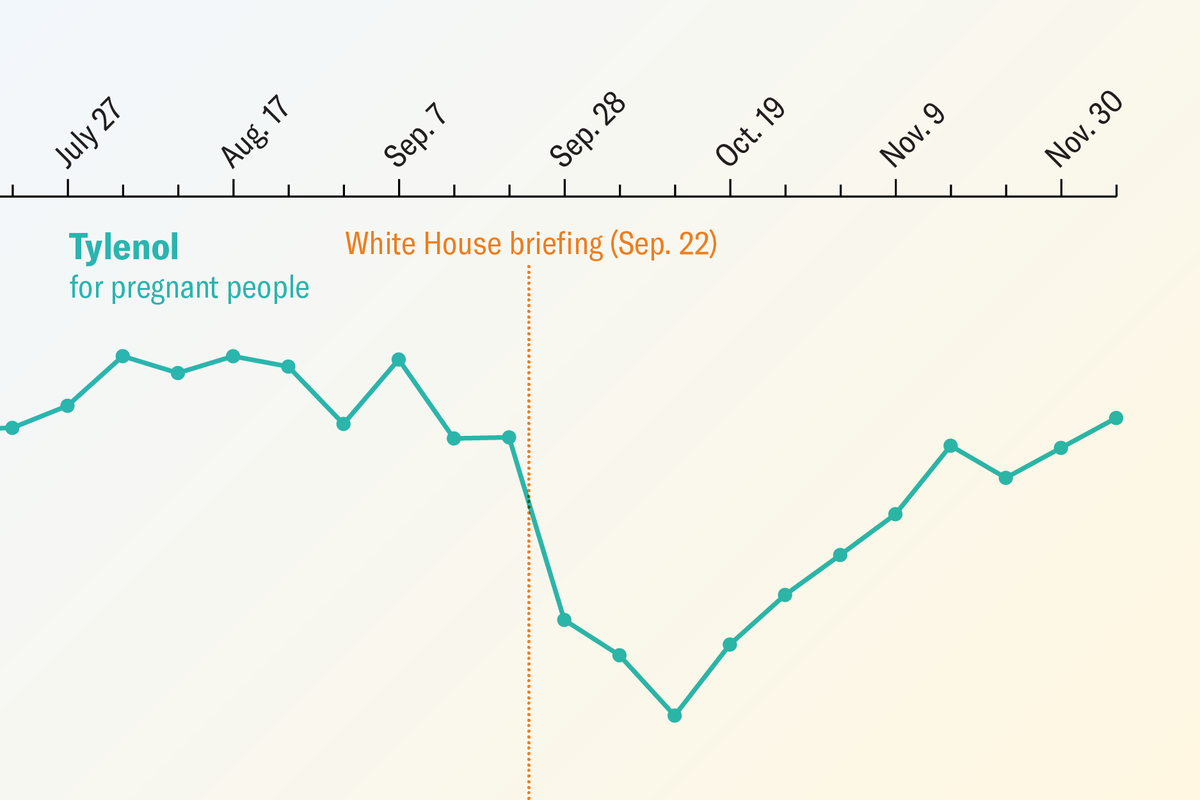
A recent analysis published in The Lancet reveals a notable shift in medication practices following unsubstantiated claims made by former President Donald Trump linking acetaminophen – the active ingredient in Tylenol – to an increased risk of autism. The study found a significant decrease in Tylenol orders for pregnant patients presenting at emergency rooms in the months after Trump’s public statements, alongside a concerning rise in prescriptions for an unproven autism treatment.
On September 22, 2025, President Trump held a White House briefing where he asserted a connection between acetaminophen use and autism, despite a lack of supporting evidence. He advised pregnant women to limit Tylenol use to situations where they couldn’t “tough it out.” During the same briefing, Trump and then-Secretary of Health and Human Services Robert F. Kennedy Jr. Promoted leucovorin (folinic acid) as a treatment for autism, again without scientific backing. This sequence of events appears to have had a measurable impact on healthcare decisions.
Researchers analyzing emergency department data observed a decrease of up to 20 percent in acetaminophen prescriptions for pregnant patients in the weeks following Trump’s announcement. Importantly, there was no corresponding change in acetaminophen orders for non-pregnant patients, suggesting the shift was directly related to the publicized claims. The analysis encompassed data from 88,857 pregnant patients and 853,216 nonpregnant patients. The study also documented a 71 percent increase in the number of children aged five to 17 prescribed leucovorin as outpatients.
Impact on Patient Care and Public Trust
The findings highlight the potential for political messaging to influence medical decisions, even in the face of established scientific consensus. “Directly after this press conference, thousands of women who would have had their fever or pain treated did not, based on false statements and mischaracterizations of data from the White House,” explained Dr. Jeremy Faust, an emergency physician at Brigham and Women’s Hospital and an assistant professor at Harvard Medical School, a co-author of the Lancet study.
Medical experts emphasize that acetaminophen remains the safest available pain reliever and fever reducer for use during pregnancy. Untreated fever during pregnancy carries significant risks, including miscarriage, birth defects, and premature birth. Research has even linked fevers during pregnancy to a potentially increased likelihood of autism in offspring, a point often overlooked in the recent discourse.
Existing Research Contradicts Claims
A large-scale study published in JAMA in 2024, following nearly 2.5 million children in Sweden from 1995 to 2019, found no evidence linking acetaminophen use during pregnancy to an increased risk of autism or attention deficit hyperactivity disorder (ADHD). The study accounted for genetic predispositions by comparing siblings, revealing that even within the same family, acetaminophen use during one pregnancy did not correlate with a higher risk of neurodevelopmental conditions in that child compared to siblings. “Within siblings where the mother took acetaminophen during one pregnancy but not during another, the siblings still shared the exact same autism and ADHD risk,” stated Brian Lee, an epidemiologist at Drexel University and senior author of the JAMA study. Read more about the JAMA study here.
The Lancet study did not determine whether pregnant patients opted for alternative medications or simply went without pain or fever relief. Nonsteroidal anti-inflammatory drugs (NSAIDs) like ibuprofen and aspirin are generally not recommended during pregnancy due to potential risks, including low amniotic fluid and birth defects. Opioids also carry risks of addiction and withdrawal symptoms for both mother and baby.
Leucovorin: An Unproven Treatment
The promotion of leucovorin as an autism treatment by Trump and his administration was also met with skepticism from the medical community. Lee noted that studies supporting leucovorin’s effectiveness are “provisional at best.” In fact, the largest study on the medication, involving only 77 children, was retracted in January due to errors in its data analysis.
“Words matter, and patients and providers want to be able to trust the guidance coming from our governmental institutions,” said Jennifer Braverman, a maternal and fetal medicine physician at University of Colorado Medicine, who was not involved in the Lancet study. She added that the former president’s announcement likely led some pregnant women to receive less safe alternatives or simply endure pain and fever without treatment, a situation she described as “remarkably dismissive of women’s pain and suffering.”
This incident underscores the critical importance of evidence-based medical advice and the potential consequences of disseminating misinformation, particularly when it concerns vulnerable populations like pregnant women and children. The long-term effects of this shift in medication practices remain to be seen, but the study serves as a stark reminder of the need for clear, accurate, and trustworthy health information.
Disclaimer: This article provides informational content and should not be considered medical advice. Always consult with a qualified healthcare professional for any health concerns or before making any decisions related to your health or treatment.
What are your thoughts on the influence of political statements on healthcare decisions? Share your perspective in the comments below.