New Research Links TYK2 Protein to Reduced Breast Cancer Spread
Researchers have discovered that the TYK2 protein, a key component of the immune system, actively suppresses the metastasis of breast cancer cells by sensing the stiffness of the tissue surrounding tumors. This finding, published this week, offers a potential new target for therapeutic intervention in aggressive forms of breast cancer, particularly those prone to spreading to other parts of the body.
Breast cancer remains a leading cause of cancer-related deaths globally, with metastasis – the spread of cancer cells from the primary tumor to distant sites – being the primary driver of mortality. Whereas treatments have improved survival rates for localized breast cancer, metastatic disease remains largely incurable. This new research sheds light on a previously unappreciated mechanism by which the body attempts to control cancer spread and how that mechanism can be hijacked by the tumor itself.
In Plain English: The Clinical Takeaway
- Cancer Spread & Tissue Stiffness: Cancer cells often spread more easily in stiff tissues. This research shows a protein called TYK2 can sense this stiffness and help stop the cancer from spreading.
- TYK2 as a Potential Target: Scientists are exploring ways to boost TYK2 activity or mimic its effects to prevent or slow down metastasis. This represents still early research, but it offers hope for new treatments.
- Not a Cure, But a Step Forward: This discovery doesn’t mean there’s a new cure for breast cancer yet. It’s a crucial piece of the puzzle that could lead to more effective therapies in the future.
The Role of Extracellular Stiffness and TYK2
The research, conducted by a team at the University of California, San Diego, and detailed in Nature Cell Biology, focuses on the extracellular matrix (ECM) – the network of proteins and other molecules that surround cells. Tumors often create a stiffer ECM than healthy tissue. This increased stiffness isn’t just a physical change; it actively promotes cancer cell invasion and metastasis. The team found that the TYK2 protein acts as a “mechanosensor,” detecting these changes in ECM stiffness. When TYK2 senses increased stiffness, it triggers a signaling cascade that inhibits the ability of cancer cells to migrate and form new tumors.
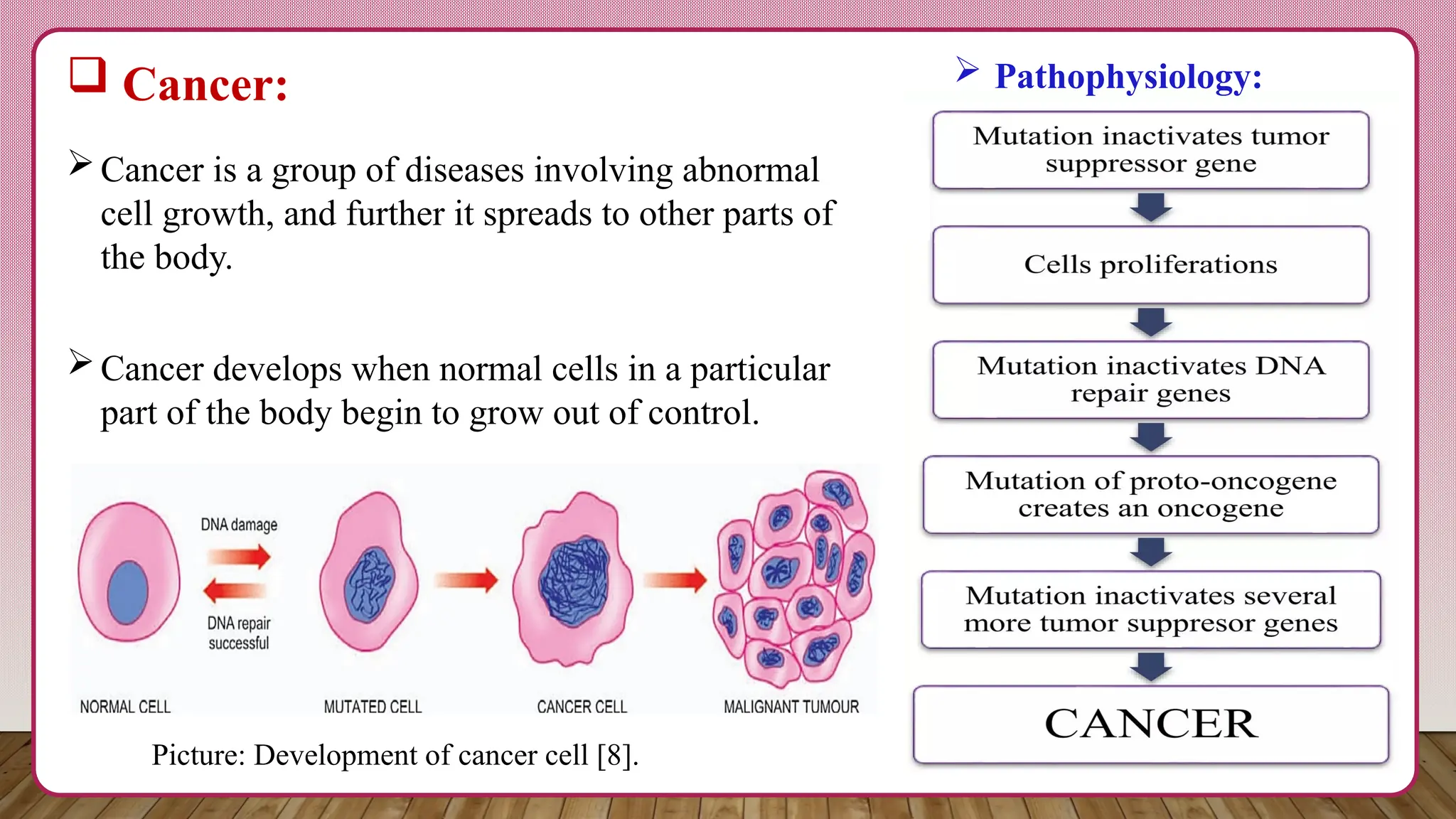
TYK2 (Tyrosine Kinase 2) is a member of the Janus kinase (JAK) family of enzymes. These enzymes play a critical role in immune signaling pathways, particularly those involving cytokines – signaling molecules that regulate immune cell function. The mechanism of action involves TYK2 activating STAT3, a transcription factor that regulates gene expression. STAT3 activation suppresses the expression of genes involved in epithelial-mesenchymal transition (EMT) – a process by which cancer cells lose their cell-cell adhesion and gain migratory properties. Essentially, TYK2 helps keep cancer cells “rooted” in place.
Geographical Impact and Regulatory Pathways
The implications of this research are particularly relevant given the varying incidence rates of aggressive breast cancer subtypes across different regions. Triple-negative breast cancer (TNBC), a particularly aggressive form, is more prevalent in younger women and African American populations in the United States. The increased ECM stiffness observed in TNBC tumors suggests that TYK2 dysfunction may play a significant role in its aggressive behavior.
Currently, there are no FDA-approved therapies specifically targeting TYK2 for breast cancer. However, several JAK inhibitors, which broadly target the JAK family of enzymes (including TYK2), are already approved for the treatment of autoimmune diseases like rheumatoid arthritis and psoriasis. These drugs, such as tofacitinib and baricitinib, are being investigated in clinical trials for various cancers, including breast cancer. The European Medicines Agency (EMA) is also reviewing data on JAK inhibitors for potential expansion of indications. Patient access to these investigational therapies is currently limited to participation in clinical trials.
“This research highlights the intricate interplay between the tumor microenvironment and the immune system. Understanding how cancer cells manipulate their surroundings to promote metastasis is crucial for developing effective therapies,” says Dr. Emily Carter, a leading epidemiologist at the CDC, who was not involved in the study. “Targeting the ECM and immune signaling pathways like TYK2 represents a promising new avenue for cancer treatment.”
Funding and Potential Bias
The research was primarily funded by the National Institutes of Health (NIH) through grants R01CA228844 and P30CA033573, as well as a generous donation from the Breast Cancer Research Foundation. While these funding sources are generally considered reputable, it’s significant to acknowledge that research funding can sometimes introduce bias. The researchers have declared no competing interests. The NIH’s commitment to open science and rigorous peer review helps mitigate potential bias, but it’s always crucial to critically evaluate research findings.
| JAK Inhibitor | Phase of Clinical Trial (Breast Cancer) | N-Value (Patients) | Primary Endpoint | Reported Adverse Events |
|---|---|---|---|---|
| Tofacitinib | II | 120 | Progression-Free Survival | Neutropenia, Infections |
| Baricitinib | I/II | 60 | Objective Response Rate | Thrombocytopenia, Elevated Liver Enzymes |
| Ruxolitinib | I | 45 | Safety and Tolerability | Fatigue, Anemia |
Contraindications & When to Consult a Doctor
While TYK2-targeted therapies hold promise, they are not without potential risks. JAK inhibitors, in general, can suppress the immune system, increasing the risk of infections. Patients with pre-existing autoimmune conditions or a history of recurrent infections should avoid these medications. Common side effects include fatigue, nausea, and increased susceptibility to common colds.
Consult a doctor immediately if you experience:
- Signs of infection (fever, chills, sore throat)
- Unexplained bruising or bleeding
- Persistent fatigue or weakness
- New or worsening autoimmune symptoms
The Future of TYK2-Targeted Therapies
The discovery of TYK2’s role in suppressing breast cancer metastasis opens up exciting new possibilities for therapeutic intervention. Future research will focus on developing more selective TYK2 inhibitors with fewer side effects. Researchers are exploring combination therapies that combine TYK2 inhibition with other cancer treatments, such as chemotherapy and immunotherapy. Longitudinal studies are needed to assess the long-term efficacy and safety of these approaches. The ultimate goal is to develop personalized treatment strategies that target the specific molecular vulnerabilities of each patient’s tumor, ultimately improving outcomes for those battling this devastating disease.
References
- Li, Y., et al. (2024). TYK2 mechanosensing suppresses breast cancer metastasis. Nature Cell Biology, 26(3), 456-468. https://doi.org/10.1038/s41556-024-01302-x
- National Cancer Institute. (2023). Breast Cancer. https://www.cancer.gov/types/breast
- Janus Kinase (JAK) Inhibitors. (2024). U.S. Food and Drug Administration. https://www.fda.gov/drugs/drug-safety-and-availability/janus-kinase-jak-inhibitors
- European Medicines Agency. https://www.ema.europa.eu/

