Researchers are developing “universal” vaccines targeting conserved regions of respiratory pathogens, such as the influenza hemagglutinin stem. By inducing broad immunity against multiple strains of flu, COVID-19, and RSV, these candidates aim to replace seasonal shots with a single, long-term prophylactic solution to prevent future pandemics.
The current paradigm of respiratory vaccination is reactive. Every year, the WHO predicts which influenza strains will dominate, leading to a “mismatch” that can reduce vaccine efficacy. A universal approach shifts the strategy from chasing mutations to targeting the “Achilles heel” of the virus—parts of the protein structure that cannot mutate without killing the virus itself.
In Plain English: The Clinical Takeaway
- One Shot, Many Strains: Instead of a new flu shot every year, scientists are building a vaccine that recognizes the “core” of the virus, making it effective against almost all versions.
- Nasal Delivery: New research focuses on nasal sprays that stop the virus in the nose and throat (mucosal immunity) before it ever reaches the lungs.
- Not Yet Available: While results in mice and early human trials are promising, these are not yet available at your local pharmacy. they are still in the rigorous testing phase.
Targeting the Hemagglutinin Stem: The Molecular Strategy
To understand the breakthrough, we must appear at the mechanism of action—how the vaccine works at a cellular level. Most seasonal flu vaccines target the “head” of the hemagglutinin (HA) protein. The head is highly mutable, meaning it changes shape frequently, allowing the virus to evade our immune system.
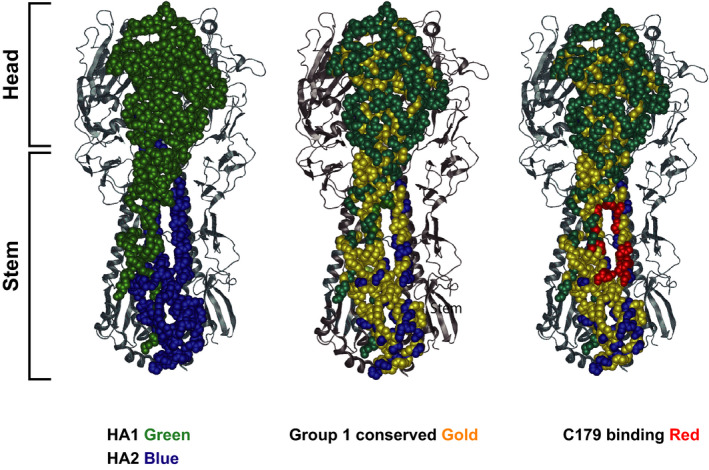
The new “universal” candidates target the HA stem. The stem is a highly conserved region, meaning it remains virtually identical across different strains. By focusing the immune response here, the vaccine creates antibodies that can neutralize a broad spectrum of viruses, including avian strains like H5N1 that pose a pandemic threat.
This approach utilizes “mosaic” antigens—engineered proteins that mimic multiple different strains simultaneously. This “trains” the B-cells in our immune system to recognize a wider variety of pathogens, effectively broadening the humoral response (the production of antibodies in the blood and mucus).
From Lab to Clinic: Navigating the Regulatory Pipeline
The transition from animal models to human application is where many “miracle” vaccines fail. Currently, several candidates are in Phase 1 and 2a clinical trials. These phases are designed to establish safety and a preliminary “proof of concept” regarding immunogenicity (the ability to provoke an immune response).
For these to reach the public, they must pass through the FDA (U.S. Food and Drug Administration) and EMA (European Medicines Agency) rigorous Phase 3 trials, involving thousands of participants to prove efficacy. In the UK, the NHS would evaluate the cost-effectiveness of a universal vaccine versus the annual cost of seasonal campaigns before widespread rollout.
Funding for this research is a hybrid of public and private interests. Significant portions of the “mosaic” and stem-based research are funded by the National Institutes of Health (NIH) and BARDA (Biomedical Advanced Research and Development Administration) in the US, ensuring that the primary goal is public health security rather than purely commercial profit.
| Vaccine Approach | Target Region | Current Stage | Primary Goal |
|---|---|---|---|
| Stem-based (HA) | Conserved HA Stem | Phase 1/2a | Broad-spectrum Flu protection |
| Mosaic Antigen | Multiple Strain Mimicry | Pre-clinical/Early Phase | Cross-strain immunity |
| Intranasal EV | Mucosal Membrane | Animal Models (Mice) | Prevention of upper respiratory entry |
“The goal is not just to prevent severe disease, but to achieve ‘sterilizing immunity’—stopping the virus at the point of entry in the nasal mucosa so that transmission is halted entirely.”
The Geo-Epidemiological Impact of Mucosal Immunity
The shift toward intranasal delivery is a game-changer for global health. Traditional intramuscular injections create systemic immunity (antibodies in the blood), but they often fail to prevent the virus from colonizing the upper respiratory tract. This is why some vaccinated individuals still spread the virus.
By stimulating the Nasal-Associated Lymphoid Tissue (NALT), these new vaccines create a frontline defense. This is particularly critical in high-density urban environments in Asia and South America, where respiratory pathogens spread rapidly. Reducing the “viral shed” (the amount of virus a person releases) could theoretically lower the R0—the basic reproduction number—of future outbreaks.
Yet, access remains a hurdle. While the US and EU may adopt these quickly, the cold-chain requirements (the need for ultra-low temperature storage) for some mRNA-based universal candidates could limit deployment in rural regions of the Global South unless thermostable versions are developed.
Contraindications & When to Consult a Doctor
While universal vaccines are still in development, the general principles of vaccination contraindications apply. Individuals with a history of severe allergic reactions (anaphylaxis) to vaccine components, such as egg proteins or polyethylene glycol (PEG), must consult an immunologist.

Patients with severely compromised immune systems (e.g., those undergoing chemotherapy or living with advanced HIV) should seek a physician’s guidance, as their ability to mount a response to a “broad” vaccine may differ from the general population.
Seek immediate medical attention if you experience shortness of breath, swelling of the throat, or a rapid heart rate following any vaccination. These are signs of a rare but serious systemic allergic reaction.
The Path Forward: A Future Without Seasonal Shots
We are not yet at the finish line, but the trajectory is clear. The convergence of mRNA technology, protein engineering, and mucosal delivery is moving us toward a “one-and-done” respiratory shield. While we must continue with seasonal boosters for now, the transition to a universal platform would drastically reduce the global burden of respiratory disease and provide a critical buffer against the next zoonotic leap.