Wegovy (semaglutide) typically induces a modest initial weight reduction in the first month, often ranging from 1% to 4% of total body weight. Although individual results vary, the medication functions by mimicking GLP-1 hormones to suppress appetite, necessitating a structured caloric deficit and exercise to ensure sustainable fat loss.
The global surge in the adoption of GLP-1 receptor agonists represents a paradigm shift in metabolic medicine. We are moving away from the outdated “willpower” narrative of weight loss and toward a clinical understanding of obesity as a chronic endocrine disorder. For patients beginning this journey, the first 30 days are less about the number on the scale and more about the physiological adaptation to a medication that fundamentally alters the communication between the gut and the brain.
In Plain English: The Clinical Takeaway
- The “Hunger Switch”: Wegovy tells your brain you are full and slows down how fast your stomach empties, making you sense satisfied with much smaller portions.
- Not a Magic Bullet: While the drug suppresses appetite, the quality of weight lost (fat vs. Muscle) depends entirely on your protein intake and resistance training.
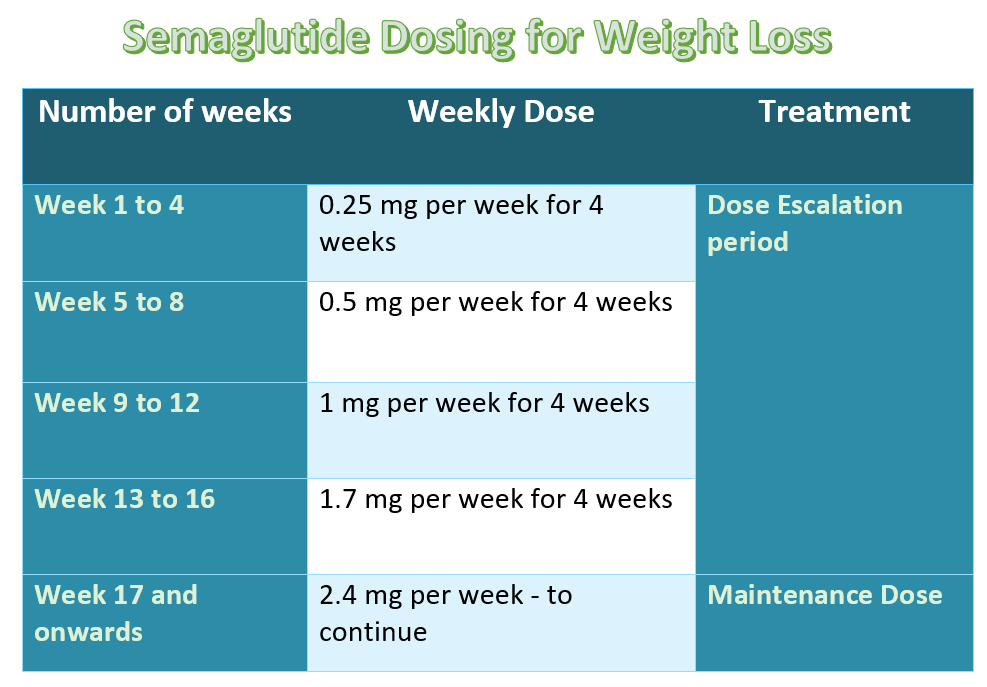
- The Titration Phase: Your first month is usually a “loading dose” to let your body adjust, meaning the most dramatic weight loss typically happens in months three through six, not month one.
The Molecular Mechanism: How Semaglutide Rewires Satiety
Wegovy is a GLP-1 (glucagon-like peptide-1) receptor agonist. In simple terms, it is a synthetic version of a hormone your body naturally produces after eating. Its primary mechanism of action—the specific biological process by which the drug produces its effect—is twofold: it targets the hypothalamus in the brain to decrease appetite and slows gastric emptying, which keeps food in the stomach longer.
This creates a state of prolonged satiety. However, the metabolic impact extends beyond hunger. Semaglutide improves insulin sensitivity, which helps the body manage blood glucose more efficiently. When paired with a calorie-restricted diet, the body is forced to mobilize stored adipose tissue (body fat) for energy. Without the accompanying diet and exercise mentioned in the patient’s query, the efficacy of the drug is significantly diminished, as the medication manages the urge to eat, but the patient must still manage the choice of what to eat.
Analyzing the Evidence: Clinical Efficacy and the STEP Trials
To understand expected weight loss, we must look at the STEP (Semaglutide Treatment Effect in People with obesity) clinical trials. In the landmark STEP 1 trial, which involved 1,961 participants, those receiving 2.4 mg of semaglutide without diabetes lost an average of 14.9% of their initial body weight over 68 weeks, compared to 2.4% in the placebo group.
It is critical to note that this weight loss was not linear. The first month is characterized by a “titration” period where the dose is gradually increased (starting at 0.25 mg) to minimize gastrointestinal side effects. A patient should not be discouraged if the first month’s loss is modest. The biological “heavy lifting” begins once the patient reaches the maintenance dose.
| Metric | Placebo Group | Wegovy (2.4mg) Group | Clinical Significance |
|---|---|---|---|
| Average Weight Loss (%) | ~2.4% | ~14.9% | Highly Significant (p < 0.001) |
| Blood Pressure Reduction | Minimal | Significant decrease in Systolic/Diastolic | Reduced Cardiovascular Risk |
| Common Side Effects | Low | Nausea, Diarrhea, Vomiting | Dose-dependent; usually transient |
The research underlying these findings was funded by Novo Nordisk, the manufacturer of Wegovy. While the trials were double-blind and placebo-controlled—meaning neither the patients nor the researchers knew who received the drug—the funding source necessitates a rigorous look at long-term data regarding weight regain after cessation of the drug.
Global Regulatory Landscapes and Patient Access
The availability of semaglutide varies significantly by region. In the United States, the FDA has approved it for chronic weight management in adults with a BMI of 30 or higher (or 27 with weight-related comorbidities). In Europe, the EMA has followed similar guidelines, though reimbursement through national health systems like the NHS in the UK remains highly restrictive, often reserved for patients with severe comorbidities.
In South Korea, where the “Doctor Now” platform operates, the MFDS (Ministry of Food and Drug Safety) has approved the drug, but the market has seen a surge in “off-label” prescriptions for cosmetic weight loss. This creates a public health risk, as the drug is being administered to individuals who do not meet the clinical criteria for obesity, potentially exposing them to risks without a corresponding medical benefit.
“The therapeutic potential of GLP-1 agonists is transformative, but we must guard against the ‘lifestyle drug’ narrative. These are potent metabolic modifiers that require strict clinical supervision to avoid lean muscle wasting and gallbladder complications.” — Dr. Anika Sharma, Lead Epidemiologist in Metabolic Health.
The Muscle Paradox: Integrating Nutrition and Exercise
A critical “information gap” in most patient discussions is the distinction between weight loss and fat loss. Rapid weight loss induced by semaglutide can lead to a significant loss of lean muscle mass. This is a dangerous trade-off; muscle is metabolically active tissue that helps maintain your basal metabolic rate (BMR).
If a patient focuses solely on the scale, they may lose 5kg in a month, but a substantial portion could be muscle. To prevent this, evidence-based integration of high-protein intake (approximately 1.2g to 1.5g of protein per kg of body weight) and resistance training is mandatory. This ensures that the weight lost is predominantly adipose tissue, preventing the “yo-yo” effect where the metabolism crashes, leading to rapid weight regain after the medication is stopped.
Contraindications & When to Consult a Doctor
Wegovy is not safe for everyone. It is strictly contraindicated—meaning it must not be used—in patients with a personal or family history of Medullary Thyroid Carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2), due to observed risks in rodent studies.
Patients should seek immediate medical intervention if they experience:
- Severe abdominal pain: This could indicate acute pancreatitis, a rare but serious inflammation of the pancreas.
- Persistent vomiting: This can lead to severe dehydration and electrolyte imbalance.
- Severe upper right abdominal pain: This may signal the formation of gallstones, a known side effect of rapid weight loss.
As we move further into 2026, the medical consensus remains clear: Wegovy is a powerful tool, but it is a supplement to, not a replacement for, fundamental metabolic health practices. The goal of treatment should be “healthspan” extension, not merely a lower number on a scale.
References
- PubMed: STEP 1 Trial – Once-Weekly Semaglutide in Adults with Overweight or Obesity
- The Novel England Journal of Medicine: GLP-1 Receptor Agonists and Cardiovascular Outcomes
- The Lancet: Long-term Efficacy and Safety of Semaglutide in Weight Management
- World Health Organization: Global Report on Obesity and Metabolic Syndrome

