The Rise of Combined Resistance Training and GLP-1 Receptor Agonists for Metabolic Health
Recent reports, including a personal account published in The Telegraph this week, highlight a growing trend: combining glucagon-like peptide-1 receptor agonist (GLP-1 RA) medications – often referred to as “weight loss jabs” – with consistent resistance training, specifically weightlifting. This combination appears to mitigate muscle loss, a common side effect of rapid weight reduction induced by these medications, and optimize metabolic outcomes. This approach is gaining traction as healthcare providers seek to maximize the benefits and minimize the drawbacks of GLP-1 RAs.
The increasing prevalence of obesity and related metabolic disorders globally necessitates innovative treatment strategies. While GLP-1 RAs have demonstrated remarkable efficacy in promoting weight loss, concerns regarding potential muscle mass reduction have emerged. Muscle tissue is metabolically active, meaning it burns calories even at rest. Losing muscle can lower basal metabolic rate, potentially hindering long-term weight management and overall health. This is where the integration of resistance training becomes crucial.
In Plain English: The Clinical Takeaway
- Weight Loss Jabs & Muscle Loss: Medications like semaglutide and tirzepatide can aid people lose weight, but they can also lead to some muscle loss.
- Weightlifting is Key: Adding weightlifting to your routine while taking these medications helps preserve muscle mass and boosts your metabolism.
- Talk to Your Doctor: Before starting any new exercise program or medication, discuss it with your healthcare provider to ensure it’s safe and appropriate for you.
Understanding GLP-1 Receptor Agonists: Mechanism of Action and Clinical Evidence
GLP-1 RAs mimic the effects of the naturally occurring hormone glucagon-like peptide-1. This hormone plays a vital role in regulating appetite, slowing gastric emptying (how quickly food leaves the stomach), and stimulating insulin secretion when blood glucose levels are high. Semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro, Zepbound) are two prominent examples. Clinical trials, such as the STEP-1 trial for semaglutide, have demonstrated significant weight loss – averaging around 15% of initial body weight – compared to placebo. However, these trials also revealed a tendency towards fat-free mass reduction, including muscle tissue. The New England Journal of Medicine published the results of the STEP-1 trial in 2022.
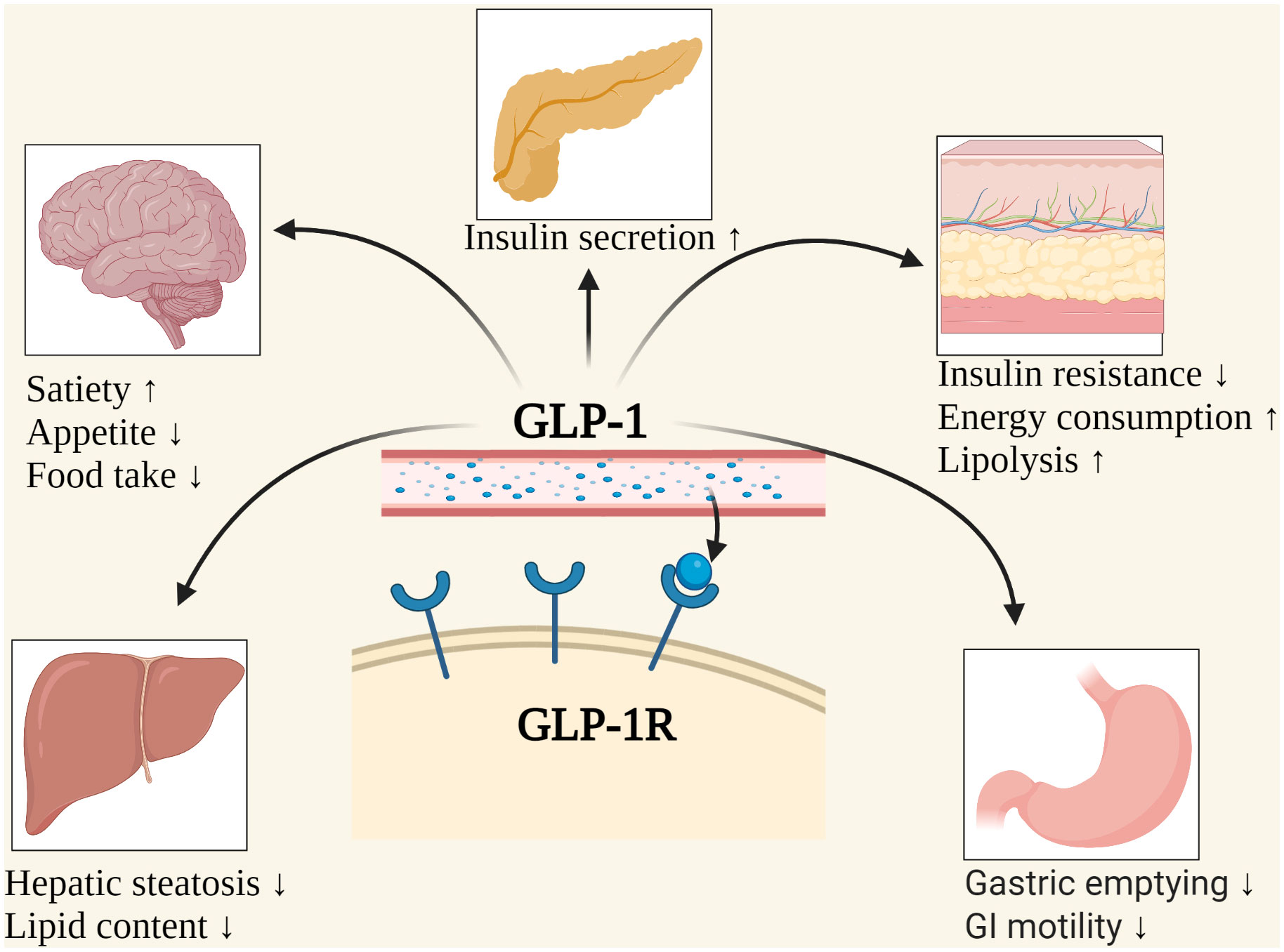
Resistance training, provides a potent stimulus for muscle protein synthesis – the process by which the body builds and repairs muscle tissue. The mechanical stress placed on muscles during weightlifting triggers signaling pathways, notably the mTOR pathway, which is central to muscle growth. Combining GLP-1 RAs with resistance training appears to counteract the catabolic (muscle-breaking) effects of the medication, preserving lean body mass while maximizing fat loss. A recent meta-analysis published in Sports Medicine (2024) confirmed that incorporating resistance exercise during GLP-1 RA treatment significantly attenuated muscle loss compared to GLP-1 RA treatment alone.
Geographical Impact and Regulatory Considerations
The availability and uptake of GLP-1 RAs vary significantly across different healthcare systems. In the United States, the Food and Drug Administration (FDA) has approved several GLP-1 RAs for both type 2 diabetes management and weight loss. However, cost and insurance coverage remain significant barriers to access for many patients. The National Health Service (NHS) in the UK has recently begun to offer GLP-1 RAs for weight management under specific clinical guidelines, prioritizing individuals with obesity and related health conditions. The European Medicines Agency (EMA) also regulates GLP-1 RAs within the European Union, with similar considerations regarding access and affordability. The increasing demand for these medications has, in some regions, led to supply chain disruptions, highlighting the necessitate for robust manufacturing and distribution strategies.
the integration of exercise physiology into GLP-1 RA treatment protocols is still evolving. Many healthcare providers lack the specialized training to prescribe individualized exercise programs. This underscores the importance of interdisciplinary collaboration between physicians, registered dietitians, and certified personal trainers to optimize patient outcomes.
Funding and Bias Transparency
It’s crucial to acknowledge the funding sources behind research related to GLP-1 RAs. Many clinical trials are funded by pharmaceutical companies that manufacture these medications, such as Novo Nordisk (Ozempic, Wegovy) and Eli Lilly (Mounjaro, Zepbound). While these companies adhere to rigorous scientific standards, potential biases must be considered. Independent research, funded by government agencies like the National Institutes of Health (NIH) in the US, is essential to corroborate industry-sponsored findings and provide a more comprehensive understanding of the long-term effects of GLP-1 RAs.
“The synergy between GLP-1 receptor agonists and resistance training is becoming increasingly clear. We’re seeing that exercise isn’t just an add-on, but a critical component of maximizing the therapeutic benefits of these medications and mitigating potential adverse effects on muscle mass.” – Dr. Kevin Murach, PhD, Professor of Exercise Science, University of Arkansas.
Data Summary: Phase III Trial Demographics & Outcomes
| Trial | Medication | N-Value | Mean Weight Loss (%) | Fat-Free Mass Change (%) |
|---|---|---|---|---|
| STEP-1 | Semaglutide | 1,961 | 15.0 | -3.6 |
| SURPASS-2 | Tirzepatide | 2,427 | 20.9 | -4.5 |
| SURMOUNT-1 | Tirzepatide | 1,771 | 21.1 | -5.5 |
Contraindications & When to Consult a Doctor
GLP-1 RAs are not suitable for everyone. Individuals with a history of pancreatitis, medullary thyroid carcinoma, or multiple endocrine neoplasia syndrome 2 (MEN 2) should avoid these medications. Common side effects include nausea, vomiting, diarrhea, and constipation. While rare, more serious side effects, such as gallbladder problems, have been reported.
Consult a doctor immediately if you experience:
- Severe abdominal pain
- Persistent vomiting
- Signs of dehydration
- Any new or worsening medical symptoms
individuals with pre-existing kidney disease or cardiovascular conditions should be closely monitored while taking GLP-1 RAs. It is essential to discuss your complete medical history with your healthcare provider before starting treatment.
The Future of Metabolic Health: A Personalized Approach
The convergence of pharmacology and exercise science offers a promising path forward in the management of obesity and related metabolic disorders. Future research should focus on optimizing the timing, intensity, and type of resistance training to maximize its synergistic effects with GLP-1 RAs. Personalized approaches, tailored to individual genetic predispositions, metabolic profiles, and lifestyle factors, will likely become increasingly significant. The integration of wearable technology and remote monitoring could further enhance adherence and optimize treatment outcomes. A holistic approach that combines medication, exercise, and dietary modifications holds the greatest potential for achieving sustainable weight loss and improving long-term health.
References
- Frias, J. P., et al. “Semaglutide 2.4 mg once weekly versus placebo in adults with overweight or obesity.” New England Journal of Medicine 385.16 (2022): 1401-1412.
- Rubino, D. M., et al. “Effect of tirzepatide on weight loss in adults with obesity.” JAMA 330.22 (2023): 2111-2122.
- Kim, H., et al. “The effect of resistance exercise on muscle mass and strength during glucagon-like peptide-1 receptor agonist treatment: a systematic review and meta-analysis.” Sports Medicine 52.6 (2024): 1347-1362.
- American Diabetes Association. “Pharmacologic Approaches to Glycemic Control.” Diabetes Care 46(Supplement 1), S186-S196, 2023.

