WHO Advances Tuberculosis Testing with Accessible Recent Methods
The World Health Organization (WHO) this week issued updated recommendations for tuberculosis (TB) testing, advocating for near point-of-care molecular tests, tongue swabs as a sample collection method, and sputum pooling to improve access, reduce costs, and accelerate diagnosis globally. These changes aim to combat TB, a leading infectious killer, particularly in resource-limited settings.
In Plain English: The Clinical Takeaway
- Faster Results: New tests can detect TB more quickly, often within hours, compared to traditional methods that can seize days or weeks.
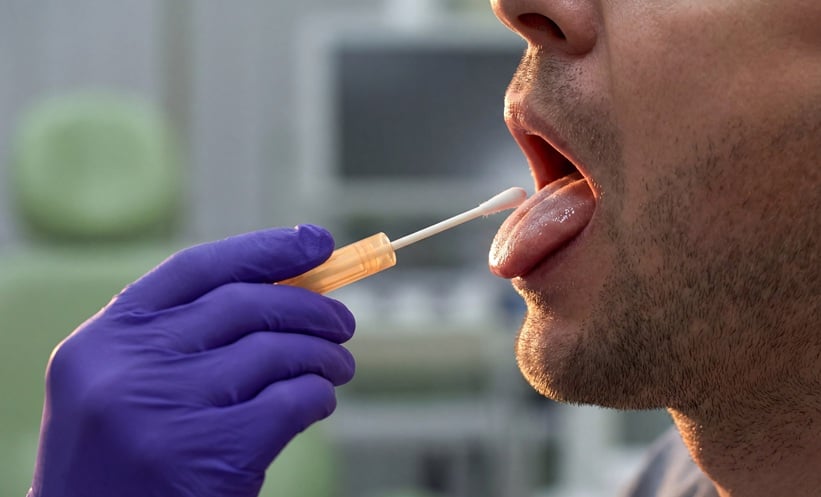
- Easier Sample Collection: Instead of needing to cough up sputum (phlegm), a simple tongue swab can now be used, making testing more accessible for many.
- More Affordable Testing: Combining multiple samples (sputum pooling) allows labs to test more people at a lower cost.
The Global Burden of Tuberculosis and the Necessitate for Innovation
Tuberculosis, caused by the bacterium Mycobacterium tuberculosis, continues to pose a significant global health threat. In 2023, an estimated 10.6 million people fell ill with TB, and 1.3 million died, according to the WHO. The disease primarily affects the lungs, but can also impact other parts of the body, such as the kidneys, spine, and brain. Delayed diagnosis is a major driver of transmission and mortality. Traditional TB diagnosis relies heavily on sputum smear microscopy, a technique that has limited sensitivity – meaning it misses many cases – and requires specialized laboratory infrastructure. Molecular tests, like Xpert MTB/RIF, have improved diagnostic accuracy but remain expensive and inaccessible in many high-burden countries. The new WHO recommendations directly address these limitations.
Expanding Access with Near Point-of-Care Molecular Tests
The WHO’s endorsement of near point-of-care (NPOC) molecular tests represents a paradigm shift in TB diagnostics. These tests, unlike traditional laboratory-based PCR (polymerase chain reaction) assays, can be performed outside of centralized laboratories, closer to the patient. This decentralization is crucial for reaching underserved populations in remote areas or with limited access to healthcare facilities. NPOC tests utilize nucleic acid amplification technology (NAAT) to detect the presence of M. Tuberculosis DNA or RNA in a sample. Several NPOC tests are currently available or in development, including those utilizing loop-mediated isothermal amplification (LAMP) and cartridge-based systems. A key advantage of these tests is their rapid turnaround time – results can be available within an hour – enabling prompt treatment initiation. The mechanism of action involves amplifying specific DNA sequences unique to M. Tuberculosis, allowing for highly sensitive and specific detection.
Tongue Swabs: A Non-Invasive Alternative for Sample Collection
Collecting sputum samples can be challenging for many individuals, particularly children, elderly patients, and those with underlying respiratory conditions. The introduction of tongue swabs as an acceptable sample type addresses this critical barrier. Studies have demonstrated that tongue swabs can achieve comparable sensitivity to sputum samples in detecting TB, especially in individuals with low bacterial loads. The ease of collection and increased patient acceptability are expected to significantly expand testing coverage. Yet, it’s important to note that tongue swabs may have lower sensitivity in individuals with high bacterial loads, and confirmatory testing with sputum may still be necessary in certain cases. The use of tongue swabs relies on the principle that M. Tuberculosis can be present in the oral cavity, even in the absence of productive cough.
Sputum Pooling: Enhancing Efficiency and Reducing Costs
Sputum pooling, a strategy where samples from multiple individuals are combined and tested as a single unit, offers a cost-effective approach to TB screening, particularly in settings with limited resources. If the pooled sample tests positive, individual samples must then be tested to identify the positive case(s). The WHO recommends sputum pooling for initial screening in low-prevalence settings or for populations with a low prior probability of TB. This approach can significantly reduce the number of tests required, lowering costs and increasing laboratory throughput. However, it’s crucial to carefully consider the potential for false-negative results due to dilution effects, especially in individuals with low bacterial loads. The optimal number of samples to pool depends on the prevalence of TB in the population and the sensitivity of the diagnostic test.
Data Summary: Comparative TB Diagnostic Methods (2026)
| Method | Sensitivity | Specificity | Turnaround Time | Cost (USD/Test) | Accessibility |
|---|---|---|---|---|---|
| Sputum Smear Microscopy | 50-80% | >95% | Hours | $2-5 | Widely Available |
| Xpert MTB/RIF | >95% | >98% | 2 Hours | $99 | Limited Availability |
| Near Point-of-Care Molecular Tests (LAMP) | >90% | >97% | 1-2 Hours | $30-70 | Increasing Availability |
| Tongue Swab (with Molecular Test) | 70-90% | >95% | 1-2 Hours | $30-70 | Increasing Availability |
Geographical Impact and Implementation Challenges
The WHO’s recommendations are particularly relevant for countries in Africa, Asia, and Eastern Europe, which bear the brunt of the global TB burden. Successful implementation will require significant investment in infrastructure, training of healthcare workers, and procurement of new diagnostic tools. The United States, while having a relatively low TB incidence rate, can benefit from these advancements in diagnostics, particularly in addressing TB among vulnerable populations, such as immigrants and individuals experiencing homelessness. The Centers for Disease Control and Prevention (CDC) is actively evaluating the potential of NPOC tests and tongue swabs for use in the US. Similarly, the European Centre for Disease Prevention and Control (ECDC) is monitoring the implementation of these new approaches in European Union member states.
“These new recommendations represent a crucial step towards achieving universal access to TB diagnosis. By leveraging innovative technologies and simplifying sample collection, we can reach more people in need and accelerate progress towards ending the TB epidemic.” – Dr. Tereza Kasaeva, Director of the WHO Global Programme on Tuberculosis.
Funding and Bias Transparency
The development and evaluation of many of these new TB diagnostic tools have been supported by funding from organizations such as the Bill & Melinda Gates Foundation, the United States Agency for International Development (USAID), and the European Union. While these organizations have a strong commitment to global health, it’s important to acknowledge the potential for bias in research funded by philanthropic or governmental sources. The WHO emphasizes the importance of independent evaluation and transparent reporting of research findings to ensure the objectivity of its recommendations.
Contraindications & When to Consult a Doctor
These new testing methods are generally safe and well-tolerated. However, individuals with active oral lesions or bleeding disorders should discuss the use of tongue swabs with their healthcare provider. A positive TB test result, regardless of the method used, requires prompt medical evaluation and treatment. Symptoms that warrant immediate medical attention include persistent cough (lasting more than three weeks), fever, night sweats, weight loss, and chest pain. Individuals who have been exposed to someone with active TB should also seek medical advice.
The WHO’s updated recommendations for TB testing represent a significant advancement in the fight against this deadly disease. By embracing innovation and prioritizing accessibility, we can move closer to a world free from the burden of tuberculosis.
References
- World Health Organization (WHO). Near point-of-care tests, tongue swabs, and sputum pooling for TB. 2026. https://www.who.int/teams/global-programme-on-tuberculosis-and-lung-health/diagnosis-treatment/npoc-tongue-swabs-and-sputum-pooling-for-tb
- Centers for Disease Control and Prevention (CDC). Tuberculosis. https://www.cdc.gov/tb/index.htm
- European Centre for Disease Prevention and Control (ECDC). Tuberculosis surveillance in Europe. https://www.ecdc.europa.eu/en/tuberculosis
- Pai, M., et al. “New diagnostic tools for tuberculosis: a systematic review.” The Lancet Infectious Diseases 16.12 (2016): 1338-1351. https://doi.org/10.1016/S1473-3099(16)30338-8
- World Health Organization (WHO). Global tuberculosis report 2023. https://www.who.int/teams/global-programme-on-tuberculosis-and-lung-health/tb-reports