Recent advancements in whole-genome sequencing (WGS) are making significant strides in the clinical management of solid tumors. A comprehensive study involving 935 patients, conducted between January 2021 and November 2022, has highlighted the real-world utility of WGS in identifying actionable biomarkers that can guide treatment decisions for various cancer types. The findings suggest that WGS could play a pivotal role in optimizing personalized cancer therapy.
The mean age of the patients included in the study was 60.9 years, with 54% being female. The predominant cancer types among the cohort were non-small cell lung cancer (NSCLC) at 23%, cancers of unknown primary (CUP) at 16%, and soft tissue sarcomas at 10%. The successful application of WGS in this population was underlined by the feasibility of obtaining viable tumor samples, which were assessed through cryosection analysis. This meticulous preparation allowed for the extraction of DNA from tumor cells while ensuring sufficient quality and quantity for accurate sequencing.
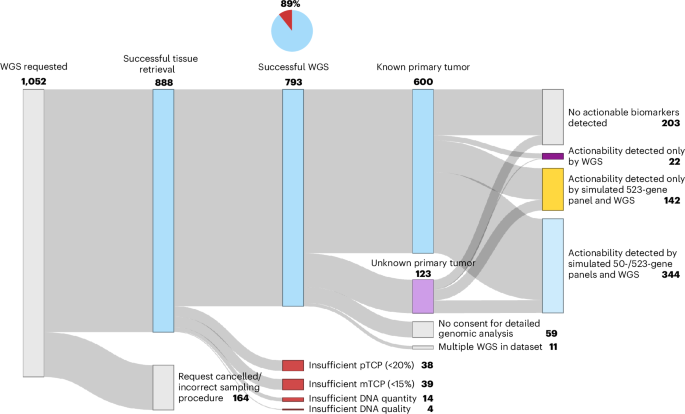
Of the 888 samples deemed suitable for WGS, diagnostic reports were generated for 89%, demonstrating a high success rate. Notably, surgical biopsy samples yielded a success rate of 91%, compared to 57% for cytology samples. The average turnaround time for results was six working days, indicating a swift process for integrating genomic insights into clinical practice.
Clinical Actionability of WGS Findings
The study further delved into the clinical actionability of the biomarkers identified through WGS. A significant 73% of patients had at least one potentially actionable biomarker, suggesting that WGS could guide treatment pathways effectively. This includes biomarkers associated with both standard-of-care and experimental treatments, providing a broad spectrum of options for clinicians.
When simulating coverage from commonly used gene panels, the study revealed that only 49% of patients would have been identified as harboring actionable biomarkers using a smaller 50-gene panel, while a more comprehensive 523-gene panel increased this figure to 70%. WGS identified additional actionable biomarkers in about 8% of cases, demonstrating its superiority in detecting clinically relevant alterations compared to panel-based sequencing.
Impact on Treatment Decisions
The implications of these findings are particularly pronounced for patients with CUP, where WGS significantly aids in establishing an initial cancer diagnosis. In this cohort, 63% were able to receive definitive tissue-of-origin diagnoses, which is crucial for guiding appropriate treatment strategies. Among these patients, 73% had at least one actionable biomarker, with 69% linked to experimental treatments, showcasing the potential of WGS to not only clarify diagnoses but also to expand therapeutic possibilities.
In patients with known primary tumors, 73% had actionable biomarkers, with an average of 1.3 biomarkers per patient. Notably, patients with high tumor mutational burden (≥10 mutations per megabase) were identified, which is often associated with better responses to immunotherapies. This understanding allows clinicians to tailor treatments based on genetic profiles, optimizing patient outcomes.
Survival Outcomes and Future Directions
Survival analysis revealed that patients with at least one actionable biomarker who received biomarker-informed therapy post-WGS had improved overall survival (OS), with a median increase from 309 to 405 days. What we have is a promising indication of the potential impact of genomic-guided treatment decisions on patient longevity. Notably, the benefit was most pronounced in patients without prior systemic therapy, suggesting that early genomic insights could substantially alter treatment trajectories.
As WGS continues to evolve, its integration into routine clinical practice promises to revolutionize the approach to cancer treatment. The ability to identify actionable biomarkers not only enhances the precision of therapeutic strategies but also fosters a more personalized approach to oncology. Moving forward, continuous research and validation of WGS applications will be essential in establishing standardized protocols for its apply in diverse clinical settings.
For patients and healthcare providers, these advancements underscore the importance of genomic testing in facilitating informed treatment decisions. As WGS technology becomes more accessible, its role in diagnosing and managing cancers is likely to grow, ultimately impacting patient care and treatment outcomes.
the real-world utility of whole-genome sequencing in solid cancers is becoming increasingly evident. As healthcare systems adapt to incorporate these genomic insights into clinical workflows, the focus will remain on enhancing patient outcomes through personalized medicine.
This article is intended for informational purposes only and should not be considered medical advice. Always consult with a healthcare professional regarding medical decisions.