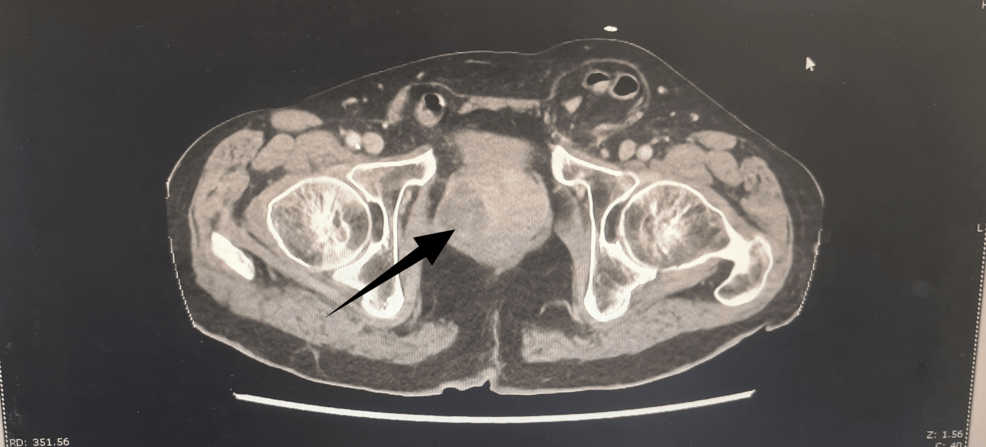
Dual Urinary Tract Pathology in a Frail Elderly Man
A recent clinical report highlights the diagnostic complexity of dual urinary tract pathologies—simultaneous, distinct disorders of the urinary system—in frail elderly patients. This case emphasizes the necessity of comprehensive geriatric ... Read More












/https://i.s3.glbimg.com/v1/AUTH_59edd422c0c84a879bd37670ae4f538a/internal_photos/bs/2026/A/4/xoiEohRsuNWx588AOLMg/captura-de-tela-2026-04-30-203104.png)