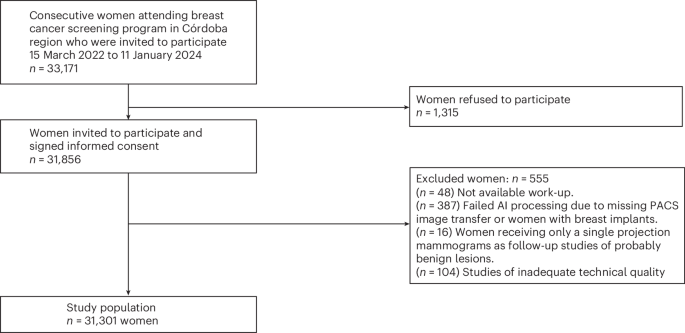
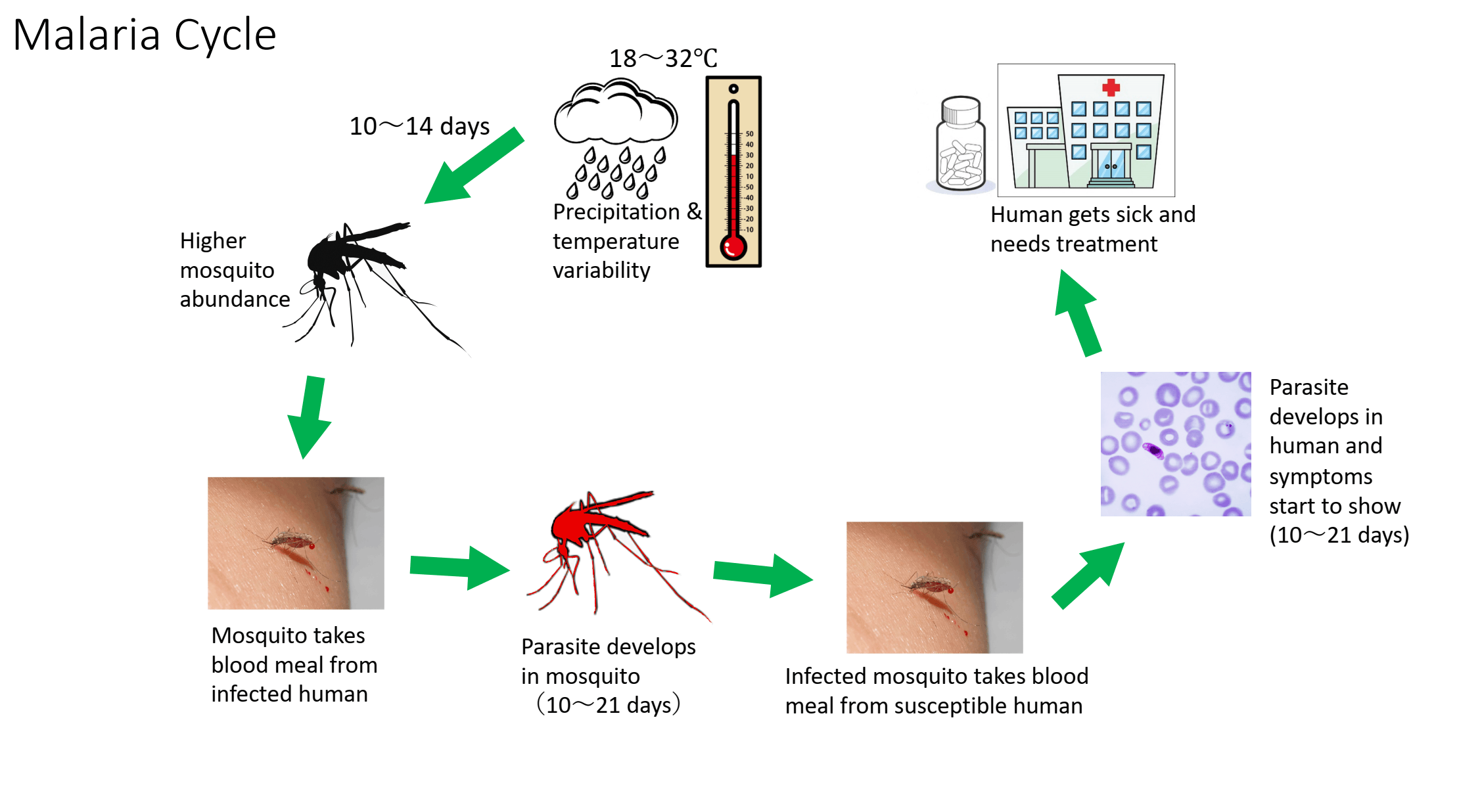
Central Bank of Russia Exchange Rates: May 9, 2026 (USD, EUR, CNY)
Moscow is currently a city of contradictions. On one street, the air is thick with the scent of celebratory blooms and the rhythmic thrum of military parades preparing for May ... Read More