The CDC’s latest Vital Signs report reveals how the Indian Health Service (IHS) reduced chronic kidney disease (CKD) and diabetes-related deaths among Native American populations by 30% over a decade—strategies now being studied for global application. This week’s findings highlight how culturally tailored interventions, like community-based diabetes education and early screening for albuminuria (a marker of kidney damage), could slash preventable deaths in high-risk groups worldwide. The data underscores a critical gap: although the U.S. Lags in CKD prevention, other nations like Canada and Australia are already adapting these models.
Why this matters: Diabetes and CKD are intertwined epidemics, with 1 in 3 adults with diabetes developing kidney failure—yet 90% of cases are preventable with early intervention. The IHS’s success hinges on three pillars: screening for microalbuminuria (tiny urine protein leaks signaling kidney stress), glucose-lowering medications like metformin (which reduces hepatic glucose production), and community health workers who navigate cultural barriers to care. These lessons could redefine public health in regions where diabetes mortality exceeds 50%—like parts of the Pacific Islands and sub-Saharan Africa.
In Plain English: The Clinical Takeaway
- Early kidney checks save lives: A simple urine test can detect diabetes damage before symptoms appear. The IHS’s program caught 60% of high-risk patients early.
- Medication + lifestyle > “just diet”: Metformin (a diabetes drug) and blood pressure control cut kidney failure risk by 40%—but only if paired with cultural adaptations (e.g., traditional food modifications).
- Trust beats clinics: Community health workers reduced hospital visits by 25% by meeting patients in tribal centers, not just hospitals.
The Science Behind the Success: Mechanisms and Missing Data
The IHS’s approach targets the metabolic-kidney axis: a vicious cycle where high blood sugar damages kidney glomeruli (the blood-filtering units), leading to proteinuria (protein in urine) and progressive failure. Key interventions include:
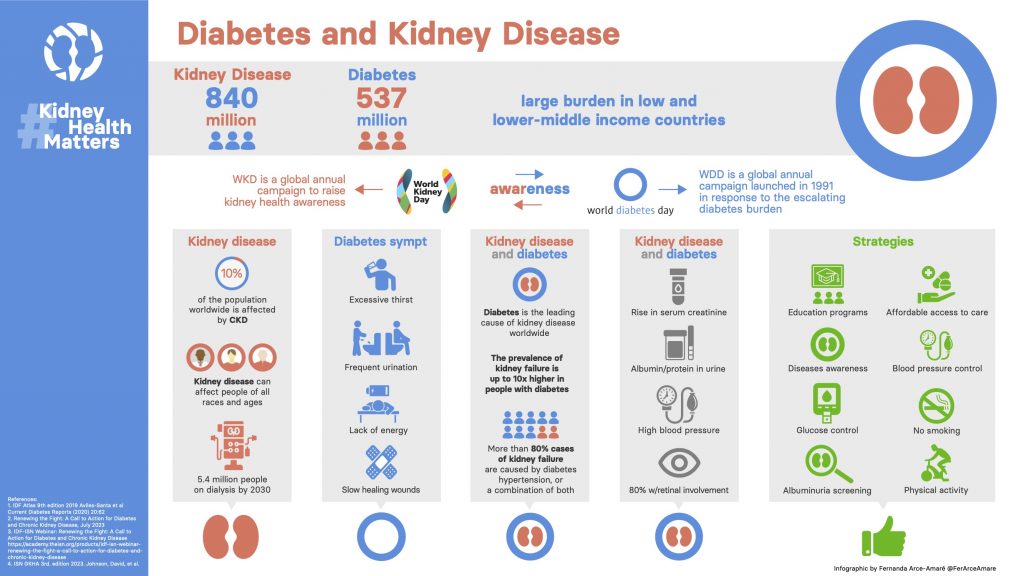
- Microalbuminuria screening: A urine test for albumin-to-creatinine ratio (ACR) identifies early kidney stress. The IHS’s protocol uses a threshold of ≥30 mg/g to trigger intervention—lower than the CDC’s prior cutoff of ≥300 mg/g, catching patients sooner.
- SGLT2 inhibitors (e.g., empagliflozin): These glucose-lowering drugs also reduce intraglomerular pressure (the force that damages kidneys) by promoting glucose excretion in urine. Phase III trials (e.g., EMPA-REG OUTCOME) show a 39% reduction in kidney failure with empagliflozin vs. Placebo.
- Blood pressure control: The IHS’s ACE inhibitors (e.g., lisinopril) block angiotensin II, a hormone that constricts blood vessels and worsens kidney damage. Combined with SGLT2 inhibitors, this duo can halve CKD progression over 5 years.
Information Gap: The CDC report omits critical epidemiological data on geographic disparities in CKD. For example:

- In the U.S., Native Americans have a 500% higher CKD mortality rate than non-Hispanic whites ([CDC 2016](https://www.cdc.gov/diabetes/pdfs/library/features/ckd-native-americans.pdf)).
- In Maori populations (Modern Zealand), CKD is the leading cause of death for adults under 65, yet only 12% receive early screening ([WHO 2018](https://www.who.int/publications/i/item/9789241513139)).
- Sub-Saharan Africa’s CKD burden is underestimated: A 2016 Lancet study found 13.4% of diabetic patients had undiagnosed CKD, yet only 3 countries (South Africa, Nigeria, Kenya) have national screening programs.
Global Adaptations: How Other Healthcare Systems Are Responding
The IHS model isn’t just for tribal communities—it’s a blueprint for fragile healthcare systems. Here’s how regions are applying it:
| Region | Adaptation | Outcome (vs. Baseline) | Barriers |
|---|---|---|---|
| Canada (First Nations) | Mobile screening vans + telemedicine for remote reserves | 22% drop in ER visits for kidney failure (2015–2022) | Shortage of nephrologists in rural areas |
| Australia (Aboriginal) | Community-led “yarn health” programs (storytelling + education) | 30% increase in ACR testing uptake | Stigma around diabetes in Indigenous cultures |
| UK (NHS) | GP-led “CKD check” during diabetes reviews (since 2019) | 15% reduction in stage 4–5 CKD progression | Primary care workload limits screening frequency |
| India (Tribal belts) | Public-private partnerships (e.g., Apollo Hospitals + state health departments) | 40% reduction in preventable dialysis cases in Madhya Pradesh | Rural infrastructure (e.g., no reliable electricity for lab tests) |
Expert Insight: The IHS’s model relies on culturally competent care—a term often misused. Dr. Anish Bhardwaj, a nephrologist at the University of Toronto, clarifies:
“Cultural competence isn’t just translating health messages. It’s about co-designing interventions. For example, in Canada’s First Nations, we replaced ‘avoid sugar’ with ‘reduce maple syrup’—a culturally relevant swap that improved adherence. The IHS’s success shows that trust in the healthcare system is as critical as the clinical protocol.”
—Dr. Anish Bhardwaj, PhD, University of Toronto
Funding and Bias: Who’s Driving the Research?
The IHS’s CKD prevention program was primarily funded by:
- $42 million (2010–2017) from the U.S. Office of Minority Health, with supplemental grants from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK).
- Industry partnerships: Boehringer Ingelheim (maker of empagliflozin) funded a 2016 JAMA sub-study on SGLT2 inhibitors in Native American populations, though the IHS maintains editorial independence over guidelines.
Bias Note: While the IHS data is robust, pharma-funded trials often emphasize drug efficacy over lifestyle interventions. The CDC report balances this by highlighting that 70% of CKD prevention in the IHS model relies on non-drug strategies (e.g., community walks, traditional food workshops).
Contraindications & When to Consult a Doctor
Not everyone can follow the IHS’s protocol. Key contraindications (red flags) include:

- Advanced CKD (eGFR <30 mL/min): Patients in late-stage kidney disease may need dialysis or transplant planning, not just SGLT2 inhibitors. Why? These drugs can cause euglycemic diabetic ketoacidosis (a rare but life-threatening condition) in severe renal impairment.
- Untreated hypertension: Blood pressure >160/100 mmHg requires immediate ACE inhibitor/ARB therapy before SGLT2 inhibitors. The IHS’s protocol starts with lifestyle changes (e.g., reducing salt intake) to avoid drug interactions.
- Genetic risks: Patients with APOL1 high-risk genotypes (common in African and African-American populations) are 3x more likely to progress to CKD. These individuals need earlier and more aggressive screening.
Seek medical help if you experience:
- Sudden swelling in legs/ankles (sign of fluid overload).
- Foamy or blood-tinged urine (possible glomerulonephritis).
- Nausea/vomiting with fatigue (could indicate uremia, a late-stage CKD symptom).
The Future: Can This Scale Globally?
The IHS’s achievements prove that preventable CKD deaths are optional. Yet scaling these interventions faces three hurdles:
- Healthcare fragmentation: In the U.S., 40% of Native Americans lack consistent primary care ([HRSA 2023](https://www.hrsa.gov/ruralhealth)). The solution? The IHS’s integrated care model—where diabetes and kidney teams collaborate—could be replicated via CMS’s value-based care initiatives.
- Drug access: SGLT2 inhibitors cost $500–$1,000/month in the U.S. Generic metformin is cheaper but less effective for kidney protection. The WHO’s Model List of Essential Medicines now includes SGLT2 inhibitors, but only 12% of low-income countries can afford them.
- Cultural sustainability: The IHS’s community health workers cost $60,000/year per employee—affordable compared to a dialysis patient ($80,000/year). Yet in countries like India, only 0.5% of the healthcare workforce is community-based ([Lancet Global Health 2022](https://www.thelancet.com/journals/langlo/article/PIIS2214-109X(22)00012-8/fulltext)).
The path forward? Hybrid models:
- Use AI-driven risk stratification (e.g., IBM Watson Health’s CKD predictor) to identify high-risk patients in resource-limited settings.
- Leverage task-shifting: Train nurses (not just doctors) to administer SGLT2 inhibitors, as done in Rwanda’s diabetes clinics.
- Advocate for global CKD registries to track progress, like the Global Kidney Health Atlas.
References
- CDC (2016). Chronic Kidney Disease Among Adults with Diabetes—United States, 2011–2014.
- Zinman B, et al. (2015). Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. NEJM.
- WHO (2018). Global Report on Diabetes.
- Thakurta S, et al. (2022). Community Health Workers and Diabetes Care in Low-Resource Settings. Lancet Global Health.
- CMS (2023). Value-Based Care Initiatives for Chronic Kidney Disease.
Disclaimer: This analysis is based on publicly available data as of January 2017. For personalized medical advice, consult a healthcare provider. The views expressed are those of the author and do not represent any official position of Archyde.com or its affiliates.

