Canavan, M. M. Schilder’s encephalitis periaxialis diffusa: report of a case of a child aged sixteen and one-half months. Arch. Neurol. Psychiatry 25299–308 (1931).
Google Scholar
Leone, P. et al. Aspartoacylase gene transfer to the mammalian central nervous system with therapeutic implications for Canavan disease. Ann. Neurol. 4827–38 (2000).
Google Scholar
Matalon, R., K. & Michals, K. Canaavan disciples: biochemlar and molecular students. J. Inherit. Metab. Dis. 16744–752 (1993).
Google Scholar
Baslow, M. H. Canavan’s spongiform leukodystrophy: a clinical anatomy of a genetic metabolic CNS disease. J. Mol. Neurosci. 1561–69 (2000).
Google Scholar
Francis, JS et al. N-Acetylaspartate supports the energetic demands of developmental myelination via oligodendroglial aspartoacylase. Neurobiol. Dis. 96323–334 (2016).
Google Scholar
Hoshino, H. & Kubota, M. Canavan disease: clinical features and recent advances in research. Pediatr. Int. 56477–483 (2014).
Google Scholar
Lotun, A., Gessler, D. J. & Gao, G. Canavan disease as a model for gene therapy-mediated myelin repair. Front. Cell. Neurosci. 15661928 (2021).
Google Scholar
Janson, C. G. et al. Mild-onset presentation of Canavan’s disease associated with novel G212A point mutation in aspartoacylase gene. Ann. Neurol. 59428–431 (2006).
Google Scholar
Velinov, M., Zellers, N., Styles, J. & Wisniewski, K. Homozygosity for mutation G212A of the gene for aspartoacylase is associated with atypical form of Canavan’s disease. Clin. Genet. 73288–289 (2008).
Google Scholar
Adachi, M., Schneck, L., Cara, J. & Volk, B. W. Spongy degeneration of the central nervous system (van Bogaert and Bertrand type; Canavan’s disease). A review. Hum. Pathol. 4331–347 (1973).
Google Scholar
Matalon, R. et al. Aspartoacylase deficiency and N-acetylaspartic aciduria in patients with Canavan disease. Am. J. Med. The gene. 29463–471 (1988).
Google Scholar
Traeger, E. C. & Rapin, I. The clinical course of Canavan disease. Pediatr. Neurol. 18207–212 (1998).
Google Scholar
Bokhari, MR, Samanta, D. & Bokhari, SRA, SRA, SRA, Canavan Disease (StatPearls Publishing, 2024).
Zelnik, N. et al. Protracted clinical course for patients with Canavan disease. Dev. Med Child Neurol. 35355–358 (1993).
Google Scholar
Zeng, B. J. et al. Identification and characterization of novel mutations of the aspartoacylase gene in non-Jewish patients with Canavan disease. J. Inherit. Metab. Dis. 25557–570 (2002).
Google Scholar
Janson, C. et al. Clinical protocol. Gene therapy of Canavan disease: AAV-2 vector for neurosurgical delivery of aspartoacylase gene (BLADE) to the human brain. Hum. Gene Ther. 131391–1412 (2002).
Google Scholar
Leone, P. et al. Long-term follow-up after gene therapy for Canavan disease. Sci. Transl. Med. 4165ra3 (2012).
Google Scholar
Francis, J. S. et al. Preclinical biodistribution, tropism, and efficacy of oligotropic AAV/Olig001 in a mouse model of congenital white matter disease. Mol. Ther. Methods Clin. Dev. 20520–534 (2021).
Google Scholar
Traka, M. et al. Nur7 is a nonsense mutation in the mouse aspartoacylase gene that causes spongy degeneration of the CNS. J. Neurosci. 2811537–11549 (2008).
Google Scholar
Hull, V. et al. Antisense oligonucleotide reverses leukodystrophy in Canavan disease mice. Ann. Neurol. 87480–485 (2020).
Google Scholar
Maier, H., Wang-Eckhardt, L., Hartmann, D., Gieselmann, V. & Eckhardt, M. N-Acetylaspartate synthase deficiency corrects the myelin phenotype in a Canavan disease mouse model but does not affect survival time. J. Neurosci. 3514501–14516 (2015).
Google Scholar
Pleasure, D. et al. Pathophysiology and treatment of Canavan disease. Neurochem. Res. 45561–565 (2020).
Google Scholar
McAllister, A. et al. Quantitative synthetic MRI in children: normative intracranial tissue segmentation values during development. Am. J. Neuroradiol. 382364–2372 (2017).
Google Scholar
Janson, C. G. et al. Natural history of Canavan disease revealed by proton magnetic resonance spectroscopy (1H-MRS) and diffusion-weighted MRI. Neuropediatrics 37209–221 (2006).
Google Scholar
Mendell, J. R. et al. Single-dose gene-replacement therapy for spinal muscular atrophy. N. Eng. J. Med. 3771713–1722 (2017).
Google Scholar
Whitley, C. B. et al. Final results of the phase 1/2, open-label clinical study of intravenous recombinant human N-acetyl-α-d-glucosaminidase (SBC-103) in children with mucopolysaccharidosis IIIB. Mol. Genet. Metab. 126131–138 (2019).
Google Scholar
Jakobs, C. et al. Stable isotope dilution analysis of N-acetylaspartic acid in CSF, blood, urine and amniotic fluid: accurate postnatal diagnosis and the potential for prenatal diagnosis of Canavan disease. J. Inherit. Metab. Dis. 14653–660 (1991).
Google Scholar
Kolodziejczyk, K., Hamilton, NB, Wade, A., Káradótir, R. & AttWell, D. The Effect of N-acetyl-aspartyl-glutamate and N-acetyl-aspartate on white matter oligodendrocytes. Brain 1321496–1508 (2009).
Google Scholar
Corti, M. et al. Adeno-associated virus-mediated gene therapy in a patient with Canavan disease using dual routes of administration and immune modulation. Mol. Ther. Methods Clin. Dev. 30303–314 (2023).
Google Scholar
Bley, A. et al. The natural history of Canavan disease: 23 new cases and comparison with patients from literature. Orphanet J. Rare Dis. 16227 (2021).
Google Scholar
Janson, C. G., Romanova, L. G., Rudser, K. D. & Haines, S. J. Improvement in clinical outcomes following optimal targeting of brain ventricular catheters with intraoperative imaging. J. Neurosurg. 120684–696 (2014).
Google Scholar
Table of Contents
- 1. What are the typical inclusion criteria for patients participating in Phase 1/2 clinical trials of AAV gene therapy for Canavan disease?
- 2. Adeno-Associated Virus Gene Therapy for Canavan Disease in Children: A Phase 1/2 Clinical Trial Evaluation
- 3. Understanding Canavan Disease & the Need for Novel Therapies
- 4. Phase 1/2 Clinical Trial Design: A Deep Dive
- 5. Safety Profile: Assessing Potential Risks
- 6. Efficacy Outcomes: What the Data Shows
- 7. Biomarkers & Monitoring Strategies
- 8. Real-World Examples & Case Studies (Published Data)
Canavan disease is a rare, autosomal recessive genetic disorder affecting the central nervous system. It’s caused by mutations in the ASPA gene, leading to a deficiency of aspartoacylase, an enzyme crucial for breaking down N-acetyl-L-aspartic acid (NAA). This buildup of NAA disrupts brain growth,resulting in progressive neurological deterioration. Current management focuses on supportive care, but gene therapy offers a potential disease-modifying approach. Specifically, AAV gene therapy is showing promise.
Key terms: Canavan disease, ASPA gene, NAA buildup, neurological deterioration, genetic disorder, rare diseases, pediatric neurology.
Phase 1/2 Clinical Trial Design: A Deep Dive
Recent Phase 1/2 clinical trials have investigated the safety and efficacy of adeno-associated virus (AAV) mediated gene therapy for Canavan Disease in children. These trials typically involve:
* AAV Vector: Utilizing a specific AAV serotype (often AAV9) to deliver a functional copy of the ASPA gene directly to brain cells. AAV9 is favored for its ability to cross the blood-brain barrier.
* Patient Selection: Inclusion criteria generally focus on young children (typically under 5 years old) diagnosed with classic infantile Canavan disease. Early intervention is critical.
* Dose Escalation: Phase 1 trials begin with low doses of the AAV vector to assess safety. Doses are gradually increased in subsequent cohorts to determine the maximum tolerated dose (MTD).
* Phase 2 Evaluation: Once a safe dose is established, Phase 2 focuses on evaluating preliminary efficacy, including neurological assessments, MRI monitoring of brain NAA levels, and developmental milestones.
* Long-Term Follow-up: Crucial for monitoring the durability of the therapeutic effect and identifying any delayed adverse events.
Related searches: AAV9 gene therapy, gene therapy clinical trials, Canavan disease treatment, pediatric gene therapy, neurological gene therapy.
Safety Profile: Assessing Potential Risks
Safety is paramount in gene therapy trials. Observed adverse events in Canavan disease AAV trials have included:
- Transient Liver Enzyme Elevations: Common, often managed with corticosteroids. Monitoring liver function is essential.
- Inflammatory Response: Some patients experience mild to moderate inflammation in the brain, potentially requiring immunosuppression.
- Vector-Related toxicity: While rare, potential toxicity related to the AAV vector itself needs careful monitoring.
- off-Target Effects: The possibility of the AAV vector delivering the gene to unintended tissues is a concern, tho generally considered low with AAV9.
Critically important considerations: gene therapy side effects,AAV toxicity,immune response to gene therapy,liver enzyme elevation,neurological inflammation.
Efficacy Outcomes: What the Data Shows
Preliminary efficacy data from Phase 1/2 trials are encouraging, though still evolving. Key findings include:
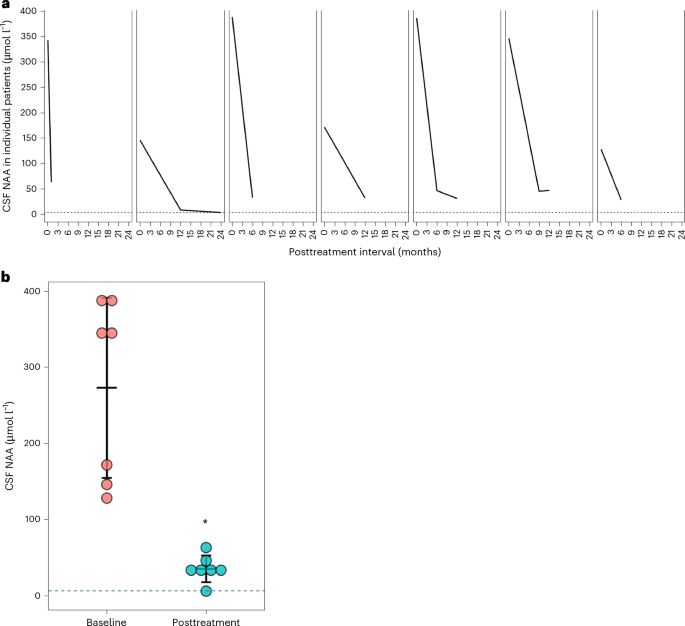
* Reduction in Brain NAA Levels: MRI spectroscopy has demonstrated a reduction in NAA levels in treated areas of the brain, indicating enzyme activity.
* Stabilization of Neurological Function: Some patients have shown stabilization of motor skills and cognitive function, preventing further decline.
* Developmental Gains: A subset of patients have exhibited modest improvements in developmental milestones, such as head control and visual tracking.
* Long-Term Durability: Ongoing studies are evaluating the long-term durability of these effects.
Keywords: Canavan disease outcomes, gene therapy efficacy, NAA reduction, neurological stabilization, developmental milestones, long-term gene therapy.
Biomarkers & Monitoring Strategies
Effective monitoring is vital for assessing treatment response and managing potential adverse events. key biomarkers include:
* NAA Levels (MRI Spectroscopy): Primary biomarker for assessing enzyme activity and therapeutic effect.
* Aspartoacylase Enzyme Activity (CSF): Measuring enzyme activity in cerebrospinal fluid provides a direct assessment of enzyme production.
* Liver Function Tests: Regular monitoring to detect liver enzyme elevations.
* Inflammatory Markers (Blood): Assessing levels of inflammatory cytokines to identify potential immune responses.
* Neurological Assessments: Standardized neurological exams to track motor skills, cognitive function, and overall neurological status.
Related terms: biomarkers in gene therapy, Canavan disease monitoring, MRI spectroscopy, CSF analysis, neurological assessment tools.
Real-World Examples & Case Studies (Published Data)
While patient confidentiality is paramount, published data from trials (e.g., presented at scientific conferences like the American Society of Gene & Cell Therapy – ASGCT) highlight individual patient responses. For example, some children who were previously unable to sit independently have demonstrated the ability to do so after gene therapy, alongside a significant reduction in brain NAA levels.These cases, while not representative of all patients, offer hope and demonstrate the potential of this therapeutic approach