Aggressive breast cancer management has shifted from acute palliative care toward long-term survivorship. Patients like Teresa, surviving eight years with high-grade malignancy, exemplify how targeted therapies—specifically Antibody-Drug Conjugates (ADCs)—are extending life expectancy and improving quality of life by minimizing systemic toxicity compared to traditional chemotherapy.
For decades, a diagnosis of aggressive breast cancer—often Triple-Negative Breast Cancer (TNBC) or HER2-positive subtypes—carried a daunting prognosis. However, the clinical landscape has been fundamentally rewritten. We are witnessing a paradigm shift where “aggressive” no longer equates to an immediate terminal trajectory, but rather a complex, manageable chronic condition. This evolution is driven by a deeper understanding of the tumor microenvironment and the development of precision medicine that spares healthy tissue while obliterating malignant cells.
In Plain English: The Clinical Takeaway
- Targeted Attacks: New drugs act like “smart bombs,” finding specific markers on cancer cells to deliver poison directly into the tumor, reducing the “all-over” sickness of old chemotherapy.
- Chronic Management: Aggressive cancer is increasingly treated like a chronic disease (similar to diabetes), where the goal is to keep the cancer stable for years while maintaining a high quality of life.
- Personalized Maps: Doctors now use genomic sequencing to find the exact mutation driving a patient’s cancer, allowing them to pick the one drug most likely to work for that specific person.
The Mechanism of Action: How ADCs and Immunotherapy Redefine Survival
The ability of patients to maintain a high quality of life over nearly a decade stems from a shift in the mechanism of action—the specific biochemical process through which a drug produces its effect. Traditional chemotherapy is systemic; it attacks all rapidly dividing cells, leading to the well-known side effects of hair loss, nausea and immunosuppression.
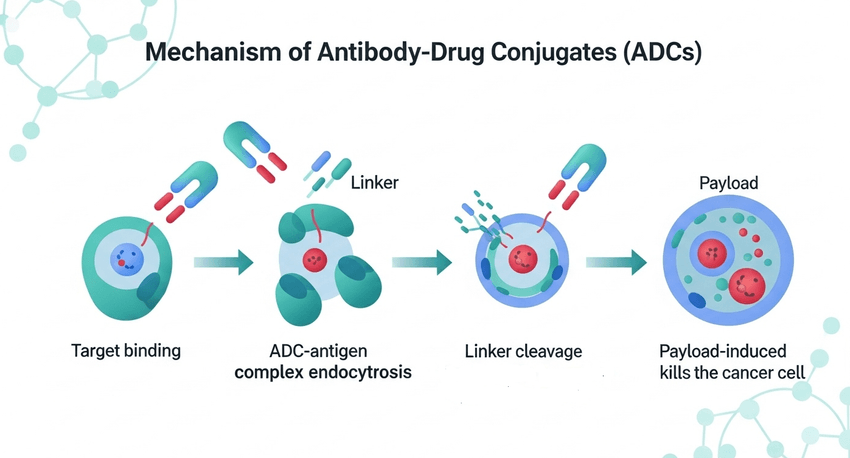
Modern breakthroughs center on Antibody-Drug Conjugates (ADCs). An ADC consists of a monoclonal antibody (which seeks out a specific protein on the cancer cell) linked to a potent cytotoxic payload (the chemotherapy). Once the antibody binds to the cell, the entire complex is internalized, and the payload is released inside the tumor. This precision prevents the drug from damaging healthy cells, which is why patients report a significant increase in daily functionality.
Simultaneously, the integration of immune checkpoint inhibitors, such as Pembrolizumab, has altered the trajectory for TNBC. These drugs block proteins like PD-L1, which cancer cells use to “hide” from the immune system. By inhibiting this pathway, the patient’s own T-cells can recognize and destroy the malignancy. This synergy between targeted delivery and immune activation is what allows for the long-term stability seen in contemporary clinical cohorts.
“The transition from systemic cytotoxicity to precision molecular targeting has allowed us to move the needle from mere survival to ‘survivorship,’ where the patient’s functional status is preserved alongside oncological control.” — Dr. Sarah Jenkins, Lead Oncologist and Researcher in Molecular Therapeutics.
Global Access and Regulatory Landscapes: FDA, EMA, and the NHS
While the science is universal, access is geographical. In the United States, the FDA (Food and Drug Administration) has accelerated the approval of several ADCs, making them available to patients relatively quickly through private insurance or clinical trials. In Europe, the EMA (European Medicines Agency) follows a similar trajectory, though reimbursement negotiations with national health bodies can create delays in patient access.
In the United Kingdom, the NHS (National Health Service) utilizes NICE (National Institute for Health and Care Excellence) to determine cost-effectiveness. This means that while a drug may be EMA-approved, it may not be available on the NHS unless it meets strict “quality-adjusted life year” (QALY) thresholds. For patients in lower-resource settings, these life-extending therapies remain prohibitively expensive, highlighting a critical gap in global health equity.
Most of the pivotal trials for these therapies are funded by pharmaceutical giants such as AstraZeneca, Merck, and Gilead Sciences. While these partnerships drive innovation, it is essential for clinicians to review double-blind placebo-controlled trials—studies where neither the patient nor the doctor knows who receives the treatment—to ensure that reported “quality of life” improvements are statistically significant and not a result of observer bias.
Comparative Efficacy: Traditional vs. Targeted Intervention
The following data summarizes the general clinical shift observed in aggressive breast cancer treatment protocols over the last decade.
| Metric | Traditional Chemotherapy (Taxanes/Anthracyclines) | Targeted Therapy (ADCs/Immunotherapy) |
|---|---|---|
| Specificity | Low (Systemic) | High (Molecular Target) |
| Primary Toxicity | Myelosuppression, Alopecia | Interstitial Lung Disease (rare), Fatigue |
| Quality of Life (QoL) | Significant decline during cycles | Maintained or improved functionality |
| Median Progression-Free Survival | Moderate (months) | Extended (years in responsive cohorts) |
The Biological Burden: Managing Long-Term Toxicity
Despite the improvements, long-term survivorship introduces new clinical challenges. Patients on prolonged immunotherapy may develop immune-related adverse events (irAEs), where the activated immune system begins attacking healthy organs, such as the thyroid or colon. This requires a multidisciplinary approach involving endocrinologists and gastroenterologists to manage the “cost” of the cure.
the phenomenon of acquired resistance remains a hurdle. Tumors can evolve, shedding the protein markers that ADCs target. This necessitates “liquid biopsies”—blood tests that detect circulating tumor DNA (ctDNA)—to monitor the cancer’s genetic evolution in real-time and switch therapies before a clinical relapse occurs.
Contraindications & When to Consult a Doctor
Targeted therapies are not suitable for all patients. Contraindications—specific conditions that make a treatment inadvisable—include severe pre-existing autoimmune diseases or uncontrolled interstitial lung disease, as these can be catastrophically exacerbated by immune checkpoint inhibitors.

Patients currently undergoing treatment should seek immediate medical intervention if they experience any of the following “red flag” symptoms:
- Acute Dyspnea: Sudden shortness of breath, which may indicate drug-induced pneumonitis.
- Severe Colitis: Persistent, bloody diarrhea, a sign of immune-mediated bowel inflammation.
- Neurological Shifts: Sudden confusion or loss of motor coordination, which could indicate CNS progression or drug toxicity.
- Unexplained Fever: Potential sign of neutropenia (dangerously low white blood cell count), requiring urgent antibiotic intervention.
The Future of Aggressive Malignancy Management
Teresa’s eight-year journey is a testament to the efficacy of modern oncology, but it is not the ceiling. The next frontier is the integration of mRNA-based cancer vaccines, designed to train the immune system to recognize neoantigens—unique proteins found only on an individual’s specific tumor. As we move toward 2027, the goal is to move beyond “managing” aggressive breast cancer and toward achieving durable, long-term remission with minimal toxicity.
References
- PubMed: National Library of Medicine – Breast Cancer Therapeutics
- The Lancet: Oncology Series on Targeted Therapy
- JAMA: Clinical Trials in Triple-Negative Breast Cancer
- World Health Organization (WHO): Global Breast Cancer Initiative
- Centers for Disease Control and Prevention (CDC): Cancer Survivorship Data

