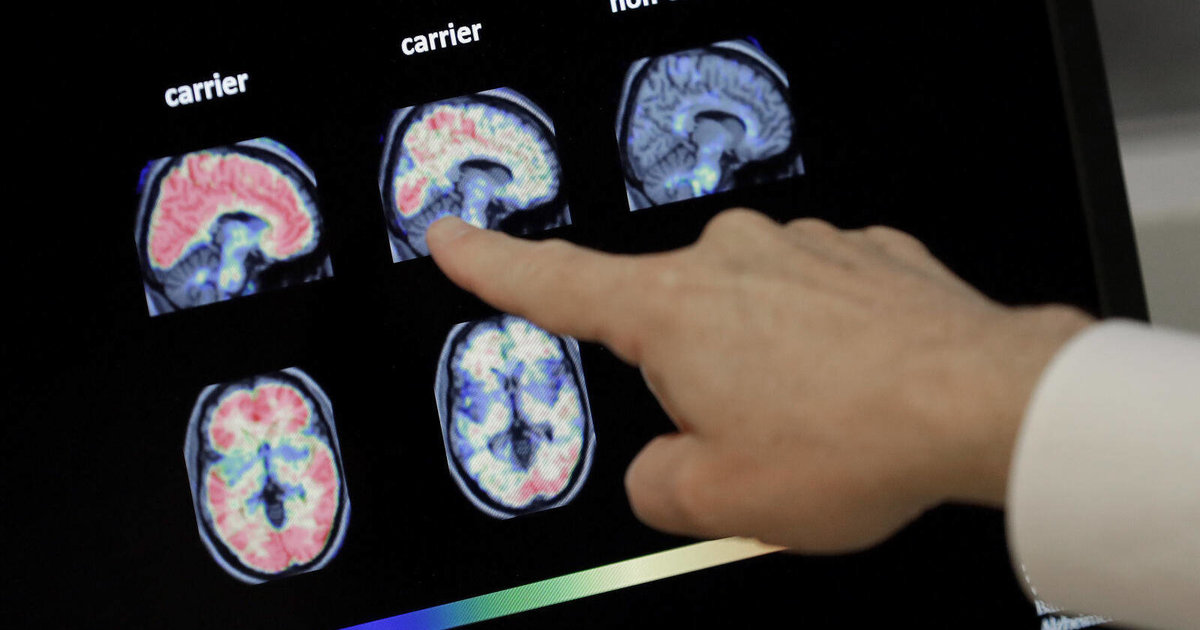
The announcement caused a stir at the annual Alzheimer’s Disease Clinical Trials conference held this year in San Francisco. On Tuesday, November 29, the results of a clinical trial in patients with early-stage Alzheimer’s disease treated with a monoclonal antibody called lecanemab were presented to an audience of attentive clinicians. For the first time, a 25% slowing of cognitive decline was observed in participants who received the treatment compared to the placebo group. These data were published on November 29 in the scientific journal NEJM.