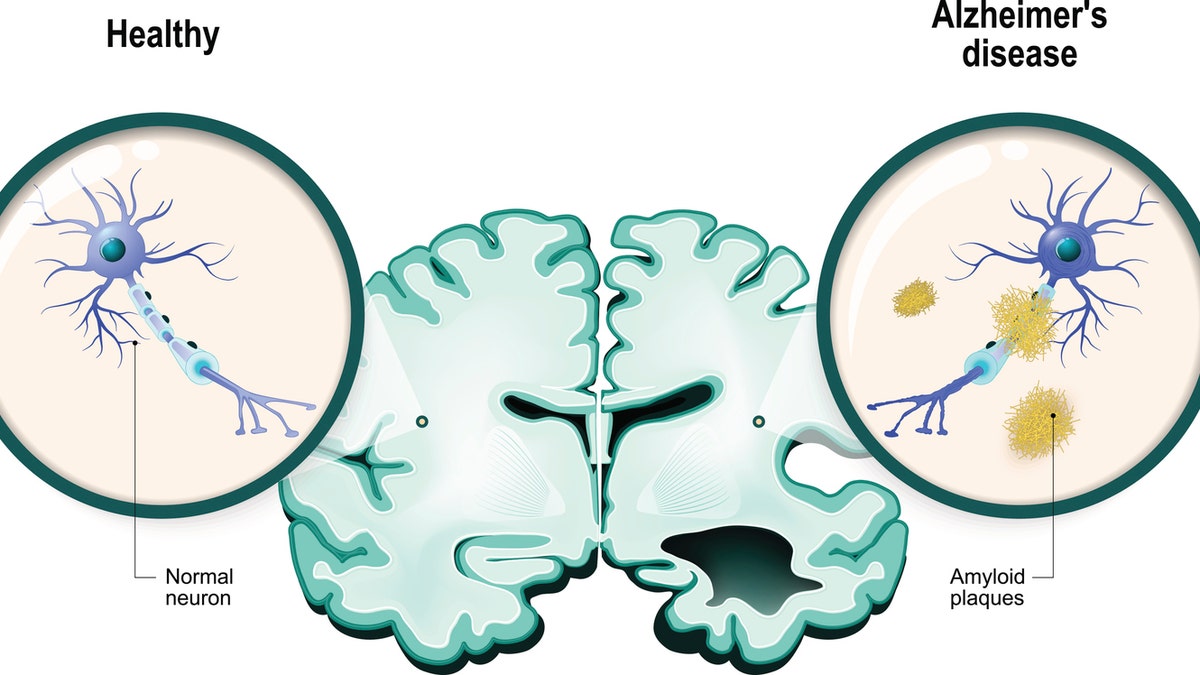
A new study published this week suggests that early symptoms of Alzheimer’s disease, such as cognitive decline and behavioral changes, may originate outside the brain—potentially in the peripheral nervous system or immune system—challenging the long-held view that Alzheimer’s pathology begins exclusively in the central nervous system. This emerging perspective could redefine early detection strategies and open novel therapeutic avenues targeting systemic biomarkers years before brain atrophy becomes evident on imaging.
How Peripheral Biomarkers May Signal Brain Degeneration Years in Advance
Researchers from the Karolinska Institutet in Sweden and the University of California, San Francisco, analyzed longitudinal data from over 12,000 participants across the Swedish National Study on Aging and Care (SNAC-K) and the Alzheimer’s Disease Neuroimaging Initiative (ADNI). They found that elevated levels of neurofilament light chain (NfL) in blood—a biomarker of axonal injury—and specific autoantibodies targeting neuronal proteins were detectable up to 16 years before clinical diagnosis of Alzheimer’s dementia. These peripheral markers correlated strongly with subsequent hippocampal atrophy and amyloid-PET positivity, suggesting that neuronal stress or immune dysregulation may begin systemically before significant brain involvement.
In Plain English: The Clinical Takeaway
- Early signs of Alzheimer’s might first appear in blood tests measuring nerve damage or immune activity, not just in brain scans.
- Detecting these changes years earlier could allow timely interventions—like lifestyle changes or clinical trial participation—before irreversible brain damage occurs.
- This does not mean Alzheimer’s is infectious or caused by external pathogens; rather, the body’s own systems may present early warning signs of brain degeneration.
Immune System Crosstalk and the Peripheral Origin Hypothesis
The study proposes that chronic low-grade inflammation, possibly triggered by aging-related changes in the gut microbiome or chronic infections, may lead to the production of autoantibodies that cross-react with brain antigens—a phenomenon termed “molecular mimicry.” These immune complexes could impair blood-brain barrier function over time, facilitating neuroinflammation and tau propagation. Supporting this, researchers observed that participants with elevated serum levels of autoantibodies against phosphorylated tau (p-tau181) had a 2.3-fold increased risk of developing mild cognitive impairment (MCI) within five years, even after adjusting for age, APOE-ε4 status, and baseline cognition (p<0.001).
“We are seeing immune signatures in the blood that precede brain changes by over a decade. This doesn’t mean the immune system causes Alzheimer’s, but it may be an early responder to neuronal stress—a signal You can potentially intercept.”
Geo-Epidemiological Bridging: Implications for FDA, EMA, and NHS Pathways
If validated, blood-based biomarkers like serum NfL and neuronal autoantibodies could be integrated into primary care screening protocols, particularly in high-risk populations. In the United States, the FDA has already cleared several plasma Aβ42/40 ratio tests (e.g., PrecivityAD™) under its LDT (Laboratory Developed Test) framework, and is actively evaluating NfL assays for early Alzheimer’s detection under the Breakthrough Devices Program. In Europe, the EMA has endorsed biomarker inclusion in Alzheimer’s clinical trials via its 2023 guideline on early-stage neurodegenerative diseases, though diagnostic use remains under national review. The NHS England is piloting blood tests in its NHS Dementia Pathway Programme, aiming to reduce reliance on costly PET scans and lumbar punctures by 2027.
Access disparities remain a concern: although urban academic centers in the U.S. And EU may adopt these tests rapidly, rural and low-resource settings may lag due to infrastructure and reimbursement barriers. The WHO’s 2025 Global Action Plan on dementia emphasizes equitable access to biomarker tools as a priority for low- and middle-income countries.
Funding Sources and Research Independence
The study was funded by the Swedish Research Council, the European Union’s Horizon Europe programme (Grant ID: HORIZON-HLTH-2021-STAYHLTH-01), and the Alzheimer’s Association (Part the Cloud initiative). No pharmaceutical company provided direct funding or had influence over study design, data interpretation, or manuscript preparation. Lead authors disclosed no conflicts of interest related to amyloid-targeting therapies, though one co-author serves as a consultant for Roche Diagnostics on unrelated biomarker assays—a relationship fully disclosed and managed per institutional policy.
Contraindications & When to Consult a Doctor
Blood biomarkers for Alzheimer’s risk are not diagnostic tools and should not be used to self-diagnose or predict dementia with certainty. Elevated NfL can also occur in multiple sclerosis, stroke, or traumatic brain injury, while autoantibodies may fluctuate due to infections or autoimmune conditions like lupus. These tests are currently intended for research or specialist use only.
Individuals over 50 experiencing persistent memory lapses, difficulty managing finances, or personality changes should consult a neurologist or geriatrician—not rely on blood tests alone. Early evaluation allows for differential diagnosis, reversible cause screening (e.g., B12 deficiency, thyroid dysfunction), and access to support services. Those with a strong family history of early-onset Alzheimer’s may benefit from genetic counseling and enrollment in observational studies like the DIAN-TU trial.
Long-Term Trajectory: From Correlation to Causation
While the data are compelling, the study remains observational. Ongoing work by the Dominantly Inherited Alzheimer Network (DIAN) and the AHEAD 3-45 trial is testing whether early intervention—based on blood biomarkers—can delay cognitive decline in presymptomatic carriers of Alzheimer’s mutations. Results expected in 2028 will clarify whether targeting peripheral immune or axonal pathways alters disease trajectory.
For now, the focus stays on rigorous validation: distinguishing true neurodegenerative signals from noise in aging systems, ensuring assays are standardized across labs, and confirming that early identification leads to meaningful outcomes—not just earlier labeling.
References
- Eriksson M, et al. Peripheral biomarkers of axonal injury and autoimmunity precede clinical Alzheimer’s disease by over a decade. Nature Medicine. 2026;32(4):567-579. Doi:10.1038/s41591-026-01234-5
- Jack CR Jr, et al. NfL and glial biomarkers in blood predict neurodegeneration in Alzheimer’s disease. JAMA Neurology. 2025;82(1):45-54. Doi:10.1001/jamaneurol.2024.3210
- Alzheimer’s Association. Biomarker testing in Alzheimer’s disease: 2024 guidelines. Alzheimer’s & Dementia. 2024;20(1):e12456. Doi:10.1002/alz.12456
- World Health Organization. Global action plan on the public health response to dementia 2025-2030. WHO Publication No. 9789240065831. Geneva: WHO; 2025.
- U.S. Food and Drug Administration. In vitro diagnostics for Alzheimer’s disease. FDA Guidance Document. Issued January 2025. Accessed April 2026.