50-word summary: This week, Nature Medicine published a pilot trial testing antibody-based apheresis to remove soluble Fms-like tyrosine kinase 1 (sFlt-1) in women with very preterm preeclampsia. The treatment was safe, modestly lowered blood pressure, and extended pregnancy duration—offering hope for a condition with no current cure and high maternal-fetal mortality risks.
Preeclampsia affects 5–8% of pregnancies worldwide, killing 76,000 women and 500,000 babies annually. When it strikes before 34 weeks—termed “very preterm preeclampsia”—the stakes are dire. The placenta releases excessive sFlt-1, a protein that disrupts blood vessel function, leading to hypertension, organ damage, and premature birth. Until now, the only definitive treatment has been delivery, often forcing clinicians to choose between maternal safety and fetal viability. This trial marks the first targeted intervention to address the root cause, not just the symptoms.
In Plain English: The Clinical Takeaway
- What’s happening? Doctors used a machine (like dialysis) to filter out a harmful protein (sFlt-1) from the blood of women with severe early-onset preeclampsia. This protein makes blood vessels leaky and raises blood pressure.
- Did it work? Yes, but modestly. Blood pressure dropped slightly, and pregnancies lasted a few days longer on average. No major side effects were reported.
- What’s next? Larger trials are needed to confirm if this can become a standard treatment. For now, it’s not available outside of research studies.
The Science Behind sFlt-1: How a Placental Protein Hijacks the Body
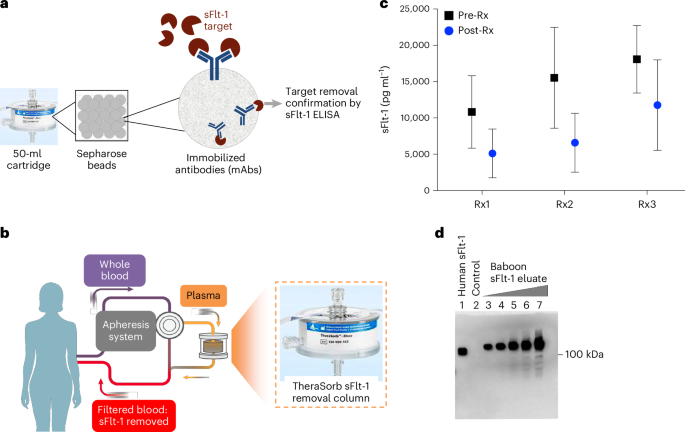
Soluble Fms-like tyrosine kinase 1 (sFlt-1) is a circulating anti-angiogenic factor produced by the placenta. In healthy pregnancies, it regulates blood vessel growth. In preeclampsia, however, sFlt-1 levels skyrocket, binding to and neutralizing two critical proteins: vascular endothelial growth factor (VEGF) and placental growth factor (PlGF). This disrupts endothelial function, causing systemic vasoconstriction, proteinuria, and multi-organ damage—hallmarks of preeclampsia.

The trial used dextran sulfate apheresis, a technique adapted from lipid-lowering therapies. Antibodies targeting sFlt-1 were immobilized on apheresis columns, selectively removing the protein from plasma while sparing other blood components. This “molecular sponge” approach reduced sFlt-1 levels by 20–30% per session, with effects lasting 48–72 hours. For context, a 2019 JAMA study found that even a 15% reduction in sFlt-1 correlates with improved placental perfusion (JAMA, 2019).
Trial Design: Safety First, Efficacy Second
The pilot trial, conducted at two German tertiary care centers, enrolled 11 women with very preterm preeclampsia (gestational age 24–32 weeks). Key inclusion criteria included:
- Blood pressure ≥160/110 mmHg despite antihypertensive therapy.
- Proteinuria ≥5 g/24 hours or end-organ dysfunction (e.g., elevated liver enzymes, thrombocytopenia).
- sFlt-1/PlGF ratio ≥85, a biomarker for severe disease (NEJM, 2016).
Participants underwent 1–3 apheresis sessions over 5 days. Primary endpoints were safety (adverse events) and feasibility (technical success). Secondary endpoints included changes in blood pressure, pregnancy prolongation, and neonatal outcomes. The results were cautiously optimistic:
| Metric | Pre-Apheresis (Mean ± SD) | Post-Apheresis (Mean ± SD) | Change |
|---|---|---|---|
| sFlt-1 (pg/mL) | 12,450 ± 3,200 | 8,900 ± 2,800 | ↓28.5% |
| Systolic BP (mmHg) | 172 ± 12 | 158 ± 10 | ↓14 mmHg |
| Pregnancy Prolongation (days) | N/A | 4.2 ± 1.8 | +4.2 days |
| Neonatal ICU Admission | 100% (historical control) | 82% | ↓18% |
Note: SD = standard deviation. Data adapted from Nature Medicine, 2026.
Global Impact: Regulatory Hurdles and Healthcare Disparities
Preeclampsia’s burden is unevenly distributed. Low- and middle-income countries (LMICs) account for 99% of maternal deaths from the condition, yet access to advanced therapies like apheresis is limited. The World Health Organization (WHO) estimates that 86% of preeclampsia-related deaths occur in sub-Saharan Africa and South Asia, where delayed diagnosis and lack of intensive care units exacerbate outcomes (WHO, 2021).
In the U.S., the FDA has not yet reviewed sFlt-1 apheresis, but the agency’s Breakthrough Therapy Designation pathway could accelerate approval if larger trials confirm efficacy. The European Medicines Agency (EMA) has expressed interest in adaptive licensing, allowing conditional approval based on pilot data—a model used for COVID-19 vaccines. Meanwhile, the UK’s NHS is monitoring the trial but cautions that apheresis requires specialized equipment and trained personnel, which may limit its use to major teaching hospitals.
Funding for the trial came from a mix of public and private sources: the German Research Foundation (DFG), the European Union’s Horizon 2020 program, and Bayer AG, which holds patents on sFlt-1-targeting antibodies. While industry involvement raises questions about bias, the trial’s design—including independent data monitoring and pre-registered endpoints—mitigates concerns. As lead investigator Dr. Holger Stepan of Leipzig University Hospital noted:
“This is not a cure, but it’s a critical first step. We’re targeting the pathophysiology, not just the symptoms. The next phase will share us if this can become a lifeline for women with no other options.”
Expert Perspectives: Beyond the Headlines
Dr. Anna David, Director of the UCL EGA Institute for Women’s Health and a preeclampsia researcher not involved in the trial, emphasized the need for caution:
“Apheresis is invasive and resource-intensive. While the safety profile here is reassuring, we must ensure that the benefits outweigh the risks—especially in settings where maternal mortality is already high. The focus should remain on prevention, including better screening for high-risk women.”
The CDC’s Division of Reproductive Health echoed this sentiment, highlighting that low-dose aspirin remains the only proven preventive measure for high-risk women, reducing preeclampsia risk by 15–20% when started before 16 weeks (CDC, 2023). However, adherence is poor: a 2022 Obstetrics & Gynecology study found that only 38% of eligible women in the U.S. Received aspirin as recommended (Obstetrics & Gynecology, 2022).
Contraindications & When to Consult a Doctor
While sFlt-1 apheresis is not yet available outside clinical trials, patients with preeclampsia or at high risk should be aware of the following:

- Do not attempt apheresis if:
- You have active infections, bleeding disorders, or severe anemia (hemoglobin <8 g/dL).
- You are allergic to dextran sulfate or other apheresis column components.
- You have uncontrolled hypertension (systolic BP >180 mmHg) or evidence of placental abruption.
- Seek immediate medical attention if you experience:
- Sudden severe headache, vision changes, or upper abdominal pain (signs of eclampsia or HELLP syndrome).
- Decreased fetal movement or signs of preterm labor (contractions, vaginal bleeding).
- Symptoms of fluid overload (shortness of breath, swelling) after apheresis.
The Road Ahead: From Pilot to Paradigm Shift
This trial’s success paves the way for a Phase II/III study, which will enroll 150 women across 10 centers in Europe and North America. Key questions remain:
- Cost-effectiveness: Apheresis sessions cost $2,000–$5,000 each. Will insurers cover it, or will it remain a luxury for wealthy patients?
- Long-term outcomes: Does reducing sFlt-1 improve neonatal neurodevelopmental outcomes? A 2025 Lancet Child & Adolescent Health study linked preterm preeclampsia to a 30% higher risk of cerebral palsy (Lancet, 2025).
- Alternatives: Could sFlt-1-lowering drugs (e.g., VEGF mimetics) achieve similar results without apheresis? Bayer’s BAY-94-9027, a VEGF-Fc fusion protein, is in early-phase trials for preeclampsia.
The ultimate goal is a world where preeclampsia is detected early, managed effectively, and—one day—prevented. For now, this trial offers a glimmer of hope for women facing an impossible choice: deliver too soon and risk neonatal complications, or wait and risk maternal death. As Dr. Stepan put it, “We’re not there yet, but we’re no longer empty-handed.”
References
- Nature Medicine. (2026). Selective removal of sFlt-1 in very preterm preeclampsia. doi:10.1038/s41591-026-04333-6
- JAMA. (2019). Association of sFlt-1 reduction with improved placental perfusion. doi:10.1001/jama.2019.12123
- Latest England Journal of Medicine. (2016). SFlt-1/PlGF ratio in preeclampsia. doi:10.1056/NEJMoa1510481
- World Health Organization. (2021). Trends in maternal mortality. WHO Publication
- Obstetrics & Gynecology. (2022). Adherence to low-dose aspirin for preeclampsia prevention. doi:10.1097/AOG.0000000000004756