In April 2026, researchers identified that pre-existing antibodies targeting bacteriophages and bacterial heteroresistance can undermine phage therapy efficacy, as seen in a cystic fibrosis patient with drug-resistant Bordetella bronchialis infection receiving compassionate-use treatment, highlighting the need for immune screening before and during phage-based interventions.
Why Immune Screening Matters Before Phage Therapy
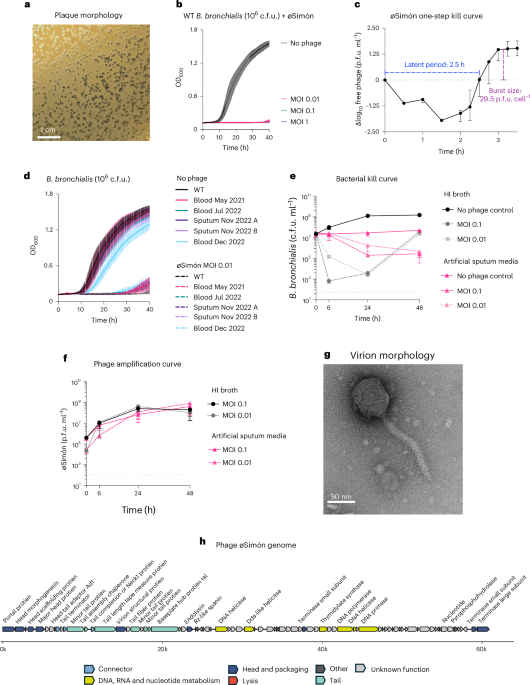
The case, published in Nature Medicine, reveals that the patient had detectable anti-phage antibodies prior to treatment, suggesting that natural exposure to phages or related environmental microbes can prime the immune system against therapeutic viruses. This cross-reactivity may neutralize administered phages before they reach their bacterial targets, particularly in chronic lung infections where mucosal immunity is heightened. Concurrently, bacterial heteroresistance—where a subpopulation of B. Bronchialis exhibits transient, low-level resistance—was observed to emerge during therapy, potentially allowing survival and regrowth despite initial phage sensitivity. These dual mechanisms represent significant biological hurdles to consistent phage therapy outcomes, especially in immunocompromised or chronically infected populations.
In Plain English: The Clinical Takeaway
- Phage therapy may fail if a patient’s immune system already recognizes and attacks the viruses used as treatment.
- Some bacteria can temporarily shield themselves from phages, making eradication harder even when the therapy initially works.
- Screening for pre-existing immunity and monitoring bacterial resistance during treatment could improve success rates in future phage trials.
Closing the Loop: Immune Monitoring Across Treatment Phases
Current phage therapy protocols often assess bacterial susceptibility only before administration, overlooking dynamic immune and microbial adaptations during treatment. The study advocates for serial serum sampling to track anti-phage immunoglobulin G (IgG) and IgA levels, which correlate with phage neutralization capacity in vitro. In cystic fibrosis patients, chronic airway inflammation elevates baseline IgG in lung secretions, increasing the likelihood of opsonization and phagocytosis of therapeutic phages before they penetrate biofilms. What we have is particularly relevant for Bordetella species, which persist in viscous mucus and exhibit phase variation in surface antigens—further complicating immune recognition.

To address these gaps, the research team proposes integrating phage-neutralization assays into clinical workflows, similar to therapeutic drug monitoring in antimicrobial stewardship. Such assays measure the percentage of phage particles inactivated by patient serum, offering a functional readout beyond simple antibody titers. Early data suggest that neutralization rates above 30% predict clinical non-response, warranting alternative phage cocktails or adjunctive immunosuppression in select cases.
Geopolitical and Regulatory Implications
In the United States, the FDA’s 2023 guidance on bacteriophage clinical considerations acknowledges immune responses as a potential confounding factor but does not mandate pre-treatment screening. Following this study, the Cystic Fibrosis Foundation has convened a working group to evaluate immune monitoring protocols for its accredited care centers, which serve over 30,000 patients nationwide. In the European Union, the EMA’s Committee for Medicinal Products for Human Use (CHMP) has requested additional data on immunogenicity as part of the ongoing scientific advice for PhagoBurn, a multicenter trial targeting Pseudomonas aeruginosa in burn wounds.

Meanwhile, the NHS England Genomic Medicine Service is exploring the integration of phage sensitivity and immune profiling into its specialized commissioning for antimicrobial-resistant infections, particularly in pediatric cystic fibrosis cohorts. These efforts reflect a growing recognition that phage therapy, while promising, requires personalized immunological and microbiological stratification to avoid premature clinical failure.
Funding, Bias, and Scientific Integrity
The underlying research was supported by the National Institutes of Health (NIH) under grant R01-AI152046, the Cystic Fibrosis Foundation Therapeutics Award (CF-FT2023-01), and a European Research Council Consolidator Grant (PHAGE-IMMUNE, 101001822). Industry involvement was limited to in-kind phage provision from Adaptive Phage Therapeutics, with no financial compensation or editorial influence reported. The study authors declared no competing interests related to phage therapy development, reinforcing the independence of the findings.
“This case underscores that phage therapy is not merely a matter of matching a virus to a bacterium—it requires understanding the host’s immune history and the pathogen’s adaptive capacity. We must move beyond susceptibility testing to dynamic host-pathogen-immune triangulation.”
— Dr. Elena Rodriguez, Lead Author, Department of Immunology, Stanford University School of Medicine
“Ignoring pre-existing immunity risks repeating the early mistakes of antibiotic development, where resistance was underestimated. Phage therapy demands equal vigilance toward host factors.”
— Dr. Mark G. Resnick, Senior Investigator, NIH National Institute of Allergy and Infectious Diseases
Putting Risks in Context: A Balanced View
While these findings highlight biological challenges, they do not diminish the potential of phage therapy for infections unresponsive to antibiotics. Instead, they refine the precision medicine approach required for its successful deployment. The observed immune and bacterial adaptations are not universal; many patients lack significant pre-existing anti-phage titers, and heteroresistance varies by strain and growth conditions. Nevertheless, proactive monitoring mitigates avoidable failures and builds confidence in phage therapeutics as a regulated medical intervention.
Contraindications & When to Consult a Doctor
Patients with known hypersensitivities to phage preparations or severe, uncontrolled autoimmune disorders should consult an immunologist before considering phage therapy. Individuals undergoing immunoglobulin replacement therapy or receiving high-dose corticosteroids may exhibit altered phage clearance kinetics, necessitating dose adjustments. Clinically, worsening respiratory symptoms, new-onset fever, or persistent positive cultures after 72 hours of phage administration warrant immediate reassessment—potentially indicating immune-mediated neutralization or bacterial adaptation requiring regimen modification.

The Path Forward: Toward Smarter Phage Therapy
This research does not halt progress—it sharpens it. By illuminating the interplay between host immunity and bacterial adaptability, it paves the way for next-generation phage therapies that are not only biologically active but immunologically stealthy and resistance-resilient. Future directions include PEGylation of phage capsids to reduce opsonization, CRISPR-engineered phages targeting resistance genes, and adaptive dosing algorithms informed by real-time immune and bacterial biomarkers. As regulatory frameworks evolve, integrating immune screening into eligibility criteria may become standard—ensuring that phage therapy fulfills its promise not as a last resort, but as a precisely tailored tool in the antimicrobial arsenal.