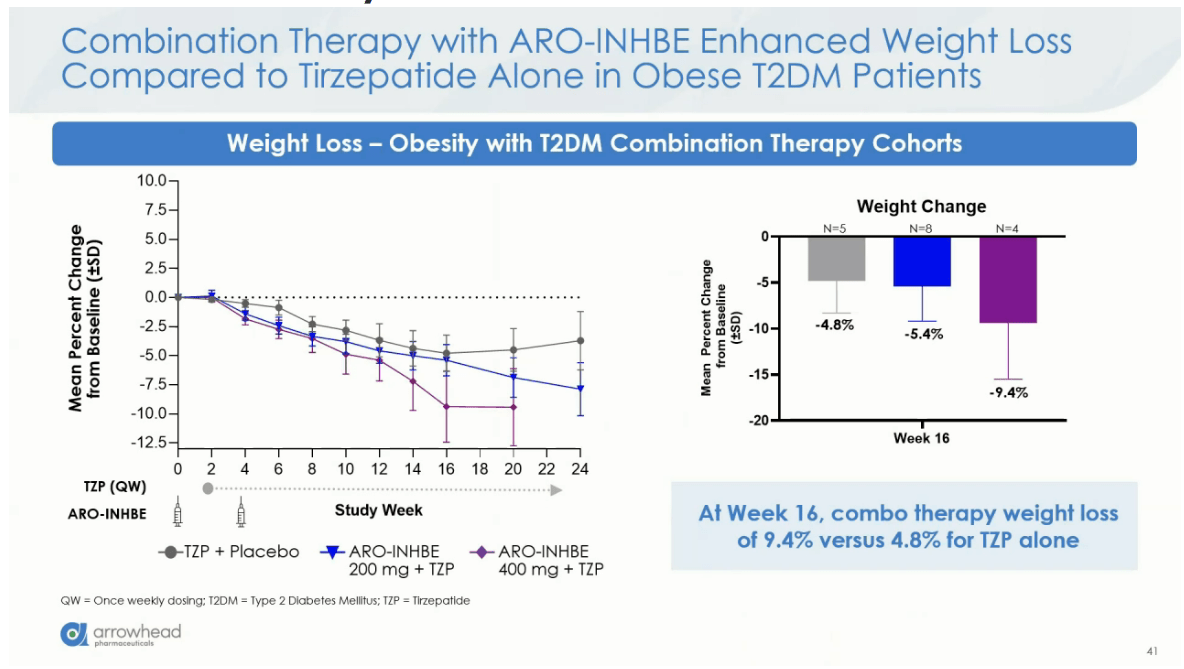
Arrowhead Pharmaceuticals’ Q2 2026 results highlight ARO-INHBE, an RNAi therapeutic demonstrating significant fat reduction (-23.2%) in obese patients with diabetes. This targeted gene-silencing approach offers a potential alternative to GLP-1 agonists by modulating the activin B pathway to improve body composition and metabolic health without solely relying on appetite suppression.
The current landscape of obesity treatment is dominated by GLP-1 (glucagon-like peptide-1) receptor agonists, which have revolutionized weight loss but often result in a concerning loss of lean muscle mass—a condition known as sarcopenia. The data released this week suggests a paradigm shift. By targeting the INHBE gene, Arrowhead is moving beyond caloric restriction and toward the biological reprogramming of how the body stores and utilizes fat.
In Plain English: The Clinical Takeaway
- Not a Diet Pill: Unlike traditional weight loss drugs that make you feel full, this treatment “turns off” a specific gene that tells your body to store fat.
- Focus on Quality: The goal is to reduce actual fat mass while protecting the muscle you need for mobility and strength.
- Metabolic Reset: It is specifically designed for those struggling with the intersection of obesity and Type 2 Diabetes, aiming to improve how the body handles insulin.
The Molecular Machinery: How RNAi Silences Fat Storage
To understand ARO-INHBE, one must understand the mechanism of action—the specific biochemical process through which a drug produces its effect. ARO-INHBE utilizes RNA interference (RNAi), a biological process where small RNA molecules inhibit gene expression by neutralizing specific messenger RNA (mRNA) molecules. In simpler terms, it acts as “genetic scissors” that prevent the body from producing activin B, a protein encoded by the INHBE gene.
Activin B is a member of the TGF-beta superfamily and plays a critical role in adiposity (the amount of fat in the body). High levels of activin B are associated with increased fat accumulation and metabolic dysfunction. By silencing this gene, ARO-INHBE reduces the biological drive to store fat, leading to the reported -23.2% reduction in fat mass. This is a distinct departure from the pharmacological pathways of semaglutide or tirzepatide, which primarily target the brain’s hunger centers and the gut’s insulin response.
“The shift toward gene-silencing therapies in metabolic disease represents a move from managing symptoms—like hunger—to addressing the underlying genetic drivers of adiposity. If the data holds, we are looking at a tool that can decouple weight loss from muscle wasting.” — Dr. Elena Rossi, PhD in Molecular Endocrinology.
Comparative Efficacy and the “Muscle Sparing” Hypothesis
The primary clinical concern with rapid weight loss in diabetic populations is the loss of lean body mass. When a patient loses weight via appetite suppression, the body often catabolizes muscle tissue alongside fat. This can lead to frailty and a lowered basal metabolic rate, making weight regain more likely.
The ARO-INHBE data is promising because it suggests a targeted reduction in fat mass specifically. By modulating the activin B pathway, the drug may allow for “muscle-sparing” weight loss. This is a critical public health metric, as maintaining muscle mass is directly correlated with improved insulin sensitivity and cardiovascular longevity in patients with obesity-related comorbidities.
| Clinical Metric | ARO-INHBE (RNAi) | Typical GLP-1 Agonists |
|---|---|---|
| Primary Mechanism | Gene Silencing (Activin B) | Incretin Mimicry |
| Primary Effect | Reduced Adipogenesis | Appetite Suppression |
| Fat Mass Impact | High (-23.2% in recent data) | High (Variable) |
| Muscle Preservation | Potential for high sparing | Risk of lean mass loss |
| Administration | Subcutaneous Injection | Subcutaneous Injection |
Global Regulatory Pathways and Patient Access
As Arrowhead moves toward later-stage clinical trials, the path to patient access involves several distinct regulatory hurdles. In the United States, the FDA (Food and Drug Administration) has shown an increasing interest in “quality of weight loss” markers, meaning they may prioritize drugs that preserve lean mass over those that simply lower the number on the scale.
In Europe, the EMA (European Medicines Agency) typically requires rigorous longitudinal data on metabolic markers before granting marketing authorization. For patients in the UK, the NHS (National Health Service) and NICE (National Institute for Health and Care Excellence) will likely evaluate ARO-INHBE based on cost-effectiveness. If the drug reduces the long-term cost of treating diabetes complications (such as dialysis or cardiovascular surgery) by preserving muscle and improving insulin sensitivity, it is more likely to be subsidized.
this research is funded by Arrowhead Pharmaceuticals, a biotechnology company. While the results are statistically significant, the inherent bias of corporate funding necessitates the validation of these results through independent, double-blind placebo-controlled trials—studies where neither the patient nor the doctor knows who is receiving the drug and who is receiving a sham treatment.
Contraindications & When to Consult a Doctor
While RNAi technology is precise, it is not without risk. The delivery system used for these drugs often relies on lipids that are processed by the liver. Patients with severe hepatic impairment or advanced liver disease may find this treatment contraindicated—meaning it is medically inadvisable for them.
Patients should seek immediate professional medical intervention if they experience:
- Severe abdominal pain: Which may indicate pancreatitis, a known risk in some metabolic therapies.
- Unexplained jaundice: Yellowing of the skin or eyes, suggesting liver stress.
- Rapid electrolyte imbalance: Symptoms include extreme muscle weakness or cardiac arrhythmias.
The Future of Metabolic Medicine
The Fiscal 2026 results from Arrowhead suggest that we are entering the era of “precision metabolic medicine.” Rather than a one-size-fits-all approach to obesity, clinicians may soon be able to screen patients for specific genetic markers, such as activin B overexpression, and prescribe a targeted RNAi therapy.
The trajectory of ARO-INHBE indicates that the future of weight management is not about eating less, but about changing the genetic instructions that dictate how our bodies store energy. While we await larger Phase III trial data, the current evidence points toward a more sustainable, muscle-preserving method of treating the global obesity epidemic.