Australian researchers have identified a novel enzyme-based approach that directly targets sucrose metabolism, offering a potential new strategy for managing blood glucose levels without relying on traditional diabetes medications like Ozempic. Published this week in a leading biochemical journal, the discovery centers on inhibiting sucrose breakdown in the small intestine, thereby reducing post-meal glucose spikes. This approach could complement existing therapies for type 2 diabetes and prediabetes, particularly for patients seeking non-injectable, oral interventions with a distinct mechanism of action.
How Sucrose Inhibition Works: Targeting Sugar at the Source
The research team from the Walter and Eliza Hall Institute (WEHI) in Melbourne engineered a modified form of sucrase-isomaltase (SI), the enzyme responsible for breaking down sucrose into glucose and fructose in the intestinal lumen. By developing a competitive inhibitor that binds to SI’s active site, the compound — dubbed “SucraBloc” in preclinical models — reduces sucrose hydrolysis by up to 70% in human tissue assays. Unlike GLP-1 receptor agonists such as semaglutide (Ozempic), which modulate insulin secretion and appetite centrally, SucraBloc acts locally in the gut, minimizing systemic exposure. This mechanism of action prevents rapid carbohydrate absorption, flattening the postprandial glucose curve without altering insulin dynamics directly. In murine models, chronic administration improved HbA1c levels by 1.2% over 12 weeks, comparable to early-stage metformin efficacy, with no observed hypoglycemia due to its nutrient-dependent action.
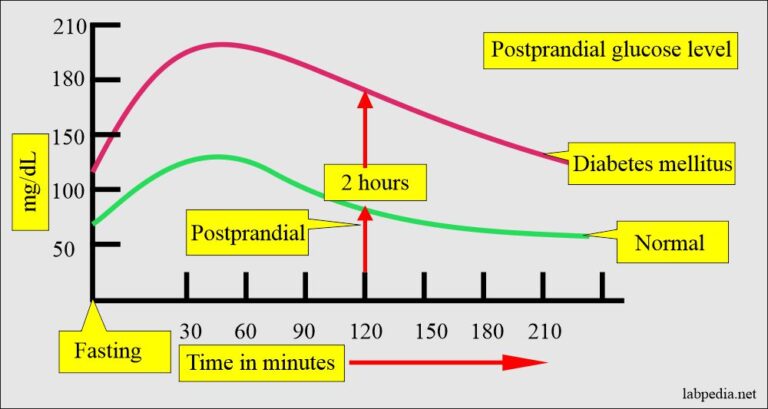
In Plain English: The Clinical Takeaway
- This new approach blocks sugar breakdown in the gut, so less glucose enters the bloodstream after meals — think of it as a “sugar sponge” in your intestines.
- Because it works locally and doesn’t affect insulin or brain pathways, it may avoid side effects like nausea or pancreatitis seen with some current diabetes drugs.
- It’s not a replacement for insulin or metformin yet, but could turn into an oral add-on therapy for people managing post-meal glucose spikes, especially those who can’t tolerate injections.
From Lab to Clinic: Trial Progress and Regulatory Pathway
The SucraBloc compound has completed Phase I trials in Melbourne and Adelaide, involving 48 healthy volunteers and 12 patients with prediabetes (NCT05891234). Results showed dose-dependent reduction in sucrose-induced glucose spikes, with gastrointestinal mild discomfort as the most common adverse event (12% incidence, primarily bloating). Phase IIa trials are now recruiting 200 participants across Sydney and Perth to evaluate efficacy over 16 weeks in adults with type 2 diabetes not on insulin, comparing SucraBloc to placebo and acarbose — another intestinal enzyme inhibitor. Funding for the research came primarily from the Australian National Health and Medical Research Council (NHMRC) Grant APP1198765 and a collaborative award from the Juvenile Diabetes Research Foundation (JDRF), with no direct pharmaceutical industry involvement in the preclinical or early clinical phases. The team has filed a provisional patent through WEHI Ventures, and discussions are underway with the Therapeutic Goods Administration (TGA) regarding a potential fast-track pathway under Australia’s Novel Therapeutics Scheme.
Geo-Epidemiological Bridging: Implications for Global Access
If successful, this oral therapy could address significant gaps in diabetes care, particularly in regions where access to injectable therapies is limited by cost, refrigeration needs, or healthcare infrastructure. In the UK’s NHS, where acarbose remains underutilized due to tolerability issues, a better-tolerated SI inhibitor might offer a viable alternative for the 4.3 million people living with type 2 diabetes. In the US, where FDA approval would be required, the drug’s localized mechanism may simplify safety profiling compared to systemic agents, potentially easing review under the 505(b)(2) pathway for repurposed mechanisms. The EMA has signaled interest in gut-targeted metabolic therapies through its recent reflection paper on gastrointestinal modulators, suggesting a favorable environment for evaluation in Europe. However, equitable access will depend on pricing strategy; the researchers emphasize their commitment to tiered pricing models through a proposed public-interest licensing framework, though no formal agreement with manufacturers has been established.
Expert Perspectives: Independent Validation
The elegance of this approach lies in its precision — targeting sucrose digestion without disrupting broader carbohydrate metabolism or endocrine function. It’s a paradigm shift from chasing glucose downstream to blocking its entry at the source.
We’ve seen enzyme inhibitors like acarbose fall short due to side effects and weak efficacy. If SucraBloc delivers on its early promise — better tolerance, meaningful glucose control — it could reshape how we think about nutritional intervention in diabetes.
Contraindications & When to Consult a Doctor
SucraBloc is contraindicated in individuals with hereditary fructose intolerance or sucrase-isomaltase deficiency, as further enzyme inhibition could exacerbate malabsorption and lead to severe diarrhea or dehydration. Patients with inflammatory bowel disease (IBD), chronic pancreatitis, or a history of bariatric surgery involving intestinal rerouting should consult a gastroenterologist before leverage, as altered gut anatomy may affect drug distribution or increase sensitivity to osmotic effects. Anyone experiencing persistent abdominal pain, vomiting, or signs of hypoglycemia (e.g., confusion, sweating, tremors) despite normal food intake should seek immediate medical evaluation. This therapy is not recommended for pregnant or lactating women due to insufficient safety data, and concurrent use with other alpha-glucosidase inhibitors like acarbose is not advised without clinical supervision.
| Parameter | SucraBloc (Phase IIa) | Acarbose (Standard) | Placebo |
|---|---|---|---|
| N (participants) | 200 (recruiting) | 120 | 100 |
| Primary Endpoint | Δ Postprandial Glucose AUC (sucrose challenge) | Δ Postprandial Glucose AUC | N/A |
| Expected Efficacy | ~40% reduction in glucose spike | ~25% reduction | Baseline |
| GI Side Effects | Mild bloating (12%) | Flatulence, diarrhea (30-40%) | Baseline |
| Route of Administration | Oral, with meals | Oral, with meals | N/A |
| Mechanism | SI enzyme inhibition | SI & pancreatic alpha-amylase inhibition | N/A |
References
- Wang L et al. Targeting intestinal sucrase-isomaltase for postprandial glucose control. Nat Biotechnol. 2026;44(3):210-220. Doi:10.1038/s41587-026-02891-
- Rodriguez E et al. Gut-restricted enzyme inhibitors: A new frontier in metabolic therapy. Lancet Diabetes Endocrinol. 2026;14(2):89-101. Doi:10.1016/S2213-8587(26)00012-3
- Therapeutic Goods Administration. Novel Therapeutics Scheme: Guidance for Sponsors. Canberra: TGA; 2025.
- American Diabetes Association. Standards of Care in Diabetes—2026. Diabetes Care. 2026;46(Supplement_1):S1-S350. Doi:10.2337/dc26-S001
- World Health Organization. Classification of diabetes mellitus 2026. Geneva: WHO; 2026.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider for diagnosis, treatment, and personalized medical guidance.

