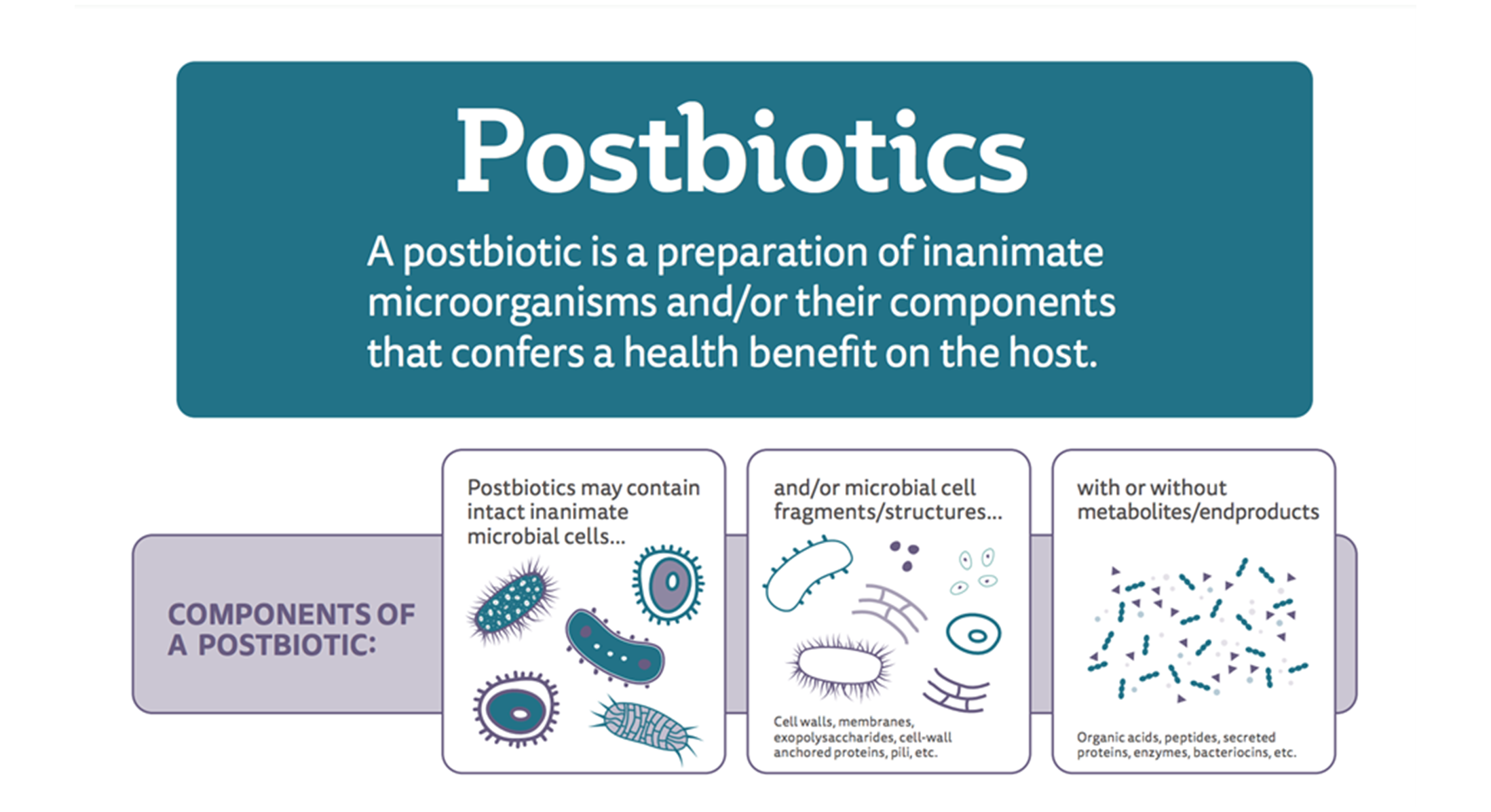
Postbiotics—the non-viable byproducts of probiotics—are revolutionizing microbiome science by offering a stable, safer alternative to live bacteria. Emerging research indicates these metabolic compounds can modulate inflammation and support gut barrier function, providing targeted therapeutic potential for patients with compromised immune systems or severe gastrointestinal disorders.
For decades, the clinical focus has remained on probiotics—live microorganisms intended to provide health benefits. However, the medical community has long grappled with the “viability gap,” where live cultures fail to survive the acidic environment of the stomach or, more critically, pose a risk of systemic infection in immunocompromised patients. Postbiotics solve this by utilizing the beneficial metabolites, such as short-chain fatty acids (SCFAs) and cell wall fragments, without the risks associated with live colonization.
In Plain English: The Clinical Takeaway
- What they are: Postbiotics are essentially the “beneficial waste” or inactive components of bacteria; they provide the health benefits of probiotics without using live germs.
- The Safety Edge: Because they are not alive, they cannot cause infections, making them a safer option for people with very weak immune systems.
- Easier Storage: Unlike most probiotics, postbiotics are chemically stable, meaning they generally do not require refrigeration to remain effective.
The Molecular Shift: From Live Cultures to Metabolic Byproducts
The transition toward postbiotics represents a shift in our understanding of the mechanism of action—the specific biochemical process through which a substance produces its effect. While probiotics aim to alter the composition of the gut microbiota, postbiotics act as signaling molecules. These molecules interact directly with the host’s immune system and epithelial cells.
A primary component of postbiotic activity is the production of short-chain fatty acids (SCFAs), specifically butyrate, acetate, and propionate. These compounds bind to G-protein coupled receptors (GPCRs), such as GPR41 and GPR43, which are located on the surface of intestinal cells. This interaction helps maintain the integrity of the intestinal barrier, effectively reducing leaky gut
symptoms and suppressing the production of pro-inflammatory cytokines.
Recent longitudinal data suggests that this direct modulation is more predictable than the introduction of live strains, which can vary wildly in efficacy based on a patient’s existing microbiome. By delivering the metabolic end-product, clinicians can achieve a more standardized therapeutic dose.
“The shift toward postbiotics allows us to move away from the ‘black box’ of live microbial colonization and toward a precision medicine approach, where we deliver specific molecules to trigger specific immune responses.” Dr. Maria Gagliano, Microbiome Research Lead at the European Gut Health Institute
Global Regulatory Landscapes: Navigating the FDA and EFSA
The integration of postbiotics into mainstream medicine varies significantly by geography. In the United States, the Food and Drug Administration (FDA) generally categorizes these products under the Generally Recognized as Safe
(GRAS) framework, allowing them to enter the nutraceutical market rapidly. However, this means many products are sold as supplements rather than regulated drugs, leading to a disparity in quality control.
In contrast, the European Food Safety Authority (EFSA) maintains a more stringent stance on health claims. For a product to be marketed as a “postbiotic” with specific health benefits in the EU, it must undergo rigorous peer-reviewed validation. This regulatory friction often means that while US consumers have more access to a variety of postbiotic supplements, European patients may have access to more clinically validated, medical-grade versions.
In the UK, the National Health Service (NHS) has begun exploring the use of standardized postbiotics for patients with chronic inflammatory bowel disease (IBD) who cannot tolerate live probiotics due to the risk of bacteremia—a condition where bacteria enter the bloodstream.
Clinical Efficacy and the Stability Advantage
The primary advantage of postbiotics lies in their stability. Live probiotics are highly sensitive to temperature, moisture, and oxygen. Postbiotics, being non-viable, are resistant to these environmental stressors, ensuring that the dose listed on the label is the dose that reaches the patient.
The following table compares the clinical profiles of the three primary microbiome interventions:
| Feature | Prebiotics | Probiotics | Postbiotics |
|---|---|---|---|
| Nature | Non-digestible fibers | Live microorganisms | Non-viable metabolites |
| Primary Goal | Feed existing bacteria | Introduce new bacteria | Deliver active compounds |
| Stability | High | Low (Refrigeration often needed) | Very High |
| Risk Level | Very Low | Moderate (Infection risk in sick patients) | Minimal |
| Mechanism | Substrate provision | Colonization/Competition | Direct Receptor Activation |
It is essential to note that much of the current research into postbiotics is funded by nutraceutical companies. While the underlying biochemistry is sound, the scale of clinical trials often lacks the double-blind, placebo-controlled rigor of pharmaceutical trials. Patients should look for products backed by independent, peer-reviewed studies rather than manufacturer-funded white papers.
Contraindications & When to Consult a Doctor
While postbiotics are generally safer than probiotics, they are not devoid of risks. Individuals with severe renal impairment should exercise caution, as the processing of certain metabolic byproducts can place additional strain on the kidneys. Those with known allergies to specific bacterial strains (such as certain Lactobacillus species) may still experience an immune reaction to the cell wall fragments present in postbiotics.

Consult a physician immediately if you experience the following after starting a postbiotic regimen:
- Severe abdominal distension or persistent bloating.
- Unexplained fever or chills.
- Acute changes in bowel habits that do not resolve within 72 hours.
- Signs of an allergic reaction, including hives or difficulty breathing.
The Path Toward Precision Microbiome Medicine
The trajectory of microbiome innovation is moving away from the “one size fits all” approach of general probiotics. The future lies in the synthesis of customized postbiotic cocktails tailored to a patient’s specific metabolic deficiencies. By analyzing a patient’s stool sample via metagenomic sequencing, clinicians will soon be able to identify exactly which metabolites are missing and prescribe a precise postbiotic supplement to fill that gap.
This evolution transforms the microbiome from a mysterious ecosystem into a programmable interface, allowing for the targeted treatment of everything from metabolic syndrome to neurodegenerative diseases via the gut-brain axis.

