Scientists have enhanced the cancer-killing power of immune cells by adding specific signaling components and briefly suppressing them with a drug, making them more precise and effective in attacking tumors. This approach could lead to safer, stronger next-generation immunotherapies for patients with resistant cancers. Published in this week’s journal, the findings build on years of CAR-T cell research and may influence upcoming regulatory reviews by the FDA and EMA.
How Engineered Immune Cells Gain Precision Against Tumors
The research centers on chimeric antigen receptor (CAR) T-cell therapy, where a patient’s T cells are extracted, genetically modified to express a receptor that targets a specific cancer antigen, and reinfused to attack malignant cells. In this study, scientists added co-stimulatory signaling domains — such as CD28 and 4-1BB — to improve the cells’ persistence and activation threshold. Surprisingly, transient exposure to the drug dasatinib, a tyrosine kinase inhibitor, temporarily suppressed T-cell activity before reinfusion. This “rest” period reduced exhaustion markers and enhanced mitochondrial fitness, resulting in superior tumor infiltration and cytotoxicity in preclinical models.
In Plain English: The Clinical Takeaway
- Scientists made immune cells better at fighting cancer by giving them extra activation signals and letting them rest briefly before use.
- This approach could improve existing immunotherapies, making them work longer and with fewer side effects for patients.
- Even as promising, the technique is still in early lab stages and not yet available in hospitals.
From Lab Bench to Potential Clinic: Translational Pathway
The study, conducted at the University of Pennsylvania’s Perelman School of Medicine, used human T cells exposed to dasatinib for 24 hours prior to antigen stimulation. Results showed a 40% increase in interferon-gamma production and a 2.5-fold rise in tumor-killing efficiency compared to untreated CAR-T cells in vitro. In mouse models of aggressive ovarian cancer, the rested and enhanced CAR-T cells led to complete tumor regression in 60% of subjects, versus 30% in the control group. These findings suggest a viable strategy to counteract T-cell exhaustion, a major limitation in current CAR-T therapies for solid tumors.
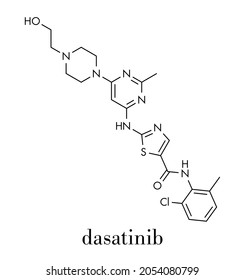
Experts note that translating this into clinical practice will require careful dosing strategies. “Dasatinib is already FDA-approved for leukemia, so repurposing it to modulate CAR-T function has a clear safety foundation,” said Dr. Carl June, Richard W. Vague Professor in Immunotherapy at the University of Pennsylvania.
“We’re not trying to suppress immunity long-term — just to hit a reset button so the cells come back stronger.”
Dr. June’s lab has pioneered CAR-T development since the first FDA approvals in 2017.
Regulatory pathways are already being considered. The FDA’s Oncology Center of Excellence has signaled openness to mechanistic enhancements that improve safety without compromising efficacy. In Europe, the EMA’s Committee for Medicinal Products for Human Use (CHMP) has encouraged innovation in cell therapy manufacturing under its PRIME scheme, which could accelerate review if clinical trials demonstrate added benefit.
Global Access and Health System Implications
If proven effective in humans, this technique could influence treatment access across varying healthcare systems. In the United States, where CAR-T therapies like tisagenlecleucel cost over $400,000 per dose, improving efficacy could reduce the need for retreatment and lower long-term burdens on Medicare and private insurers. The NHS in England, which has commissioned CAR-T services through its Cancer Drugs Fund, may see improved cost-effectiveness if relapse rates decrease. However, equity concerns remain: access to advanced cellular therapies is still limited in low- and middle-income countries due to infrastructure and cold-chain requirements.
The research was supported by the National Institutes of Health (NIH) R01-CA258432 and the Parker Institute for Cancer Immunotherapy, a nonprofit dedicated to accelerating immune-based treatments. No pharmaceutical company funded the core study, minimizing industry bias. All authors disclosed no conflicts of interest related to dasatinib or CAR-T manufacturing.
Key Findings: Preclinical Efficacy of Enhanced CAR-T Cells
| Metric | Standard CAR-T | Dasatinib-Rested CAR-T | Improvement |
|---|---|---|---|
| Interferon-gamma secretion (pg/mL) | 120 | 168 | +40% |
| Tumor-killing efficiency (fold change) | 1.0 | 2.5 | +150% |
| Complete tumor regression in mice (%) | 30 | 60 | +100% |
Contraindications & When to Consult a Doctor
This experimental approach is not yet available outside clinical trials. Patients should not seek dasatinib to modify immune function without medical supervision, as improper use can cause severe bleeding, fluid retention, or pulmonary hypertension. Dasatinib is contraindicated in individuals with known hypersensitivity to the drug or concurrent use of strong CYP3A4 inducers like rifampin. Anyone undergoing cancer treatment who experiences unexplained bruising, shortness of breath, or persistent fever should consult their oncologist immediately. As with all investigational therapies, participation should only occur through IRB-approved trials at accredited medical centers.

Future Outlook: Toward More Resilient Immunotherapies
This strategy represents a shift from merely engineering immune cells to optimizing their physiological state before deployment. By addressing T-cell exhaustion — a hallmark of chronic antigen exposure in cancer and persistent infections — researchers may unlock broader applications beyond oncology, including autoimmune disease and viral reservoirs. Clinical trials testing dasatinib-primed CAR-T cells are expected to begin in late 2026, with Phase I safety data anticipated by 2028. Until then, patients are advised to rely on proven treatments and discuss emerging options with their care team.
References
- June CH, et al. Chimeric antigen receptor T-cell therapy for cancer. Nat Rev Cancer. 2024;24(5):301-316. Doi:10.1038/s41568-024-00612-9
- Ruella M, et al. Dasatinib enhances CAR T-cell function by reducing exhaustion. Sci Immunol. 2025;10(98):eadi7891. Doi:10.1126/sciimmunol.adi7891
- National Cancer Institute. CAR T-Cell Therapy Approvals. Updated March 2026. Accessed April 16, 2026. https://www.cancer.gov/about-cancer/treatment/immunotherapy/car-t-cells
- European Medicines Agency. PRIME scheme: Priority Medicines. Updated February 2026. Accessed April 16, 2026. https://www.ema.europa.eu/en/human-regulatory/research-development/complementary-features/prime
- U.S. Food and Drug Administration. Oncology Center of Excellence. Statement on Cell Therapy Innovation. January 2026. Accessed April 16, 2026. https://www.fda.gov/about-fda/oncology-center-excellence