Retatrutide, an investigational triple-hormone receptor agonist targeting GIP, GLP-1, and glucagon receptors, has shown significant weight loss in early clinical trials, with participants losing up to 24% of body weight over 48 weeks in Phase II studies. As of April 2026, it remains under investigation and is not approved for general use by the FDA, EMA, or other major regulatory bodies. Its potential role in obesity management is being closely watched, but experts caution against off-label use or expectations of rapid ‘shredded’ physiques without medical supervision.
How Retatrutide Works: Targeting Three Hormonal Pathways
Retatrutide is a novel peptide-based drug designed to simultaneously activate three key metabolic receptors: glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon. This triple agonism aims to enhance satiety, increase energy expenditure, improve insulin sensitivity, and reduce hepatic fat storage — mechanisms distinct from current dual agonists like tirzepatide. By modulating appetite centers in the hypothalamus and altering lipid metabolism in adipose tissue and liver, retatrutide addresses multiple drivers of obesity pathophysiology. Unlike stimulant-based weight loss drugs, it does not directly increase heart rate or blood pressure through adrenergic activation, offering a potentially safer profile for long-term metabolic regulation.
In Plain English: The Clinical Takeaway
- Retatrutide is not yet approved for weight loss and should not be used outside of clinical trials or prescription programs.
- Early data show strong average weight reduction, but individual results vary and long-term safety beyond two years is still unknown.
- Lifestyle changes — including diet and exercise — remain foundational; no medication replaces sustainable health behaviors.
Clinical Evidence: Phase II Results and Ongoing Research
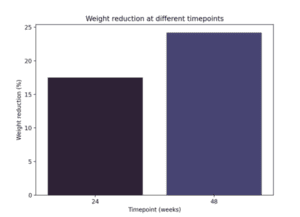
In a 2024 Phase II trial published in The Lancet, 338 adults with obesity (BMI ≥30) or overweight with comorbidities received varying doses of retatrutide or placebo over 48 weeks. The highest dose group (12 mg) achieved a mean weight loss of 24.0%, compared to 2.1% in placebo. Secondary outcomes included significant reductions in waist circumference, HbA1c, and liver fat content. Adverse events were predominantly gastrointestinal — nausea, vomiting, and diarrhea — typically mild to moderate and transient. No cases of pancreatitis or medullary thyroid carcinoma were reported in this cohort, though long-term monitoring continues.

As of early 2026, retatrutide is in Phase III development under the TRIUMPH program, with global trials enrolling participants in the United States, Europe, and Asia. Eli Lilly and Company is the primary sponsor, having acquired the asset through its collaboration with internal research divisions. Funding for the pivotal trials comes exclusively from Eli Lilly, with no external public grants disclosed in trial registries.
Geo-Epidemiological Bridging: Regulatory Pathways and Access
In the United States, the FDA has granted retatrutide Fast Track designation for obesity treatment, reflecting the high unmet demand in a population where over 42% of adults live with obesity (CDC, 2025). However, approval is not expected before late 2027, contingent on Phase III safety and efficacy outcomes. In Europe, the EMA has initiated rolling review procedures, but no formal marketing application has been submitted yet. The NHS in the UK is unlikely to consider routine commissioning until NICE completes a full health technology assessment, which typically occurs 12–18 months post-approval.
Disparities in access may emerge post-approval, particularly in low- and middle-income countries where cost-effectiveness thresholds are stricter. Unlike insulin or statins, novel obesity therapeutics often face reimbursement hurdles due to perceptions of lifestyle culpability, despite obesity being recognized as a chronic disease by the WHO, AMA, and Endocrine Society.
Funding, Bias, and Scientific Integrity
All published clinical data on retatrutide to date originate from trials sponsored by Eli Lilly and Company. While industry funding does not invalidate results, it necessitates scrutiny of study design, endpoint selection, and publication bias. Independent verification is emerging through investigator-initiated studies, but none have yet reported long-term outcomes. Transparency efforts include registration on ClinicalTrials.gov (NCT05129546) and adherence to ICMJE guidelines for conflict-of-interest disclosure in peer-reviewed journals.
“The magnitude of weight loss observed with retatrutide is clinically meaningful and approaches levels seen with bariatric surgery in some individuals — but we must remain cautious. Long-term data on cardiovascular outcomes, durability of effect, and potential rebound after discontinuation are still pending.”
— Dr. Melanie Jay, MD, MS, Associate Professor of Population Health and Medicine, NYU Grossman School of Medicine, Director of the NYU Langone Comprehensive Program on Obesity
“We are seeing a paradigm shift in obesity pharmacotherapy, but equitable access must be part of the conversation from day one. A drug that works well in clinical trials fails as a public health tool if only the wealthy can afford it.”
— Dr. Fatima Cody Stanford, MD, MPH, MPA, Obesity Medicine Physician Scientist, Massachusetts General Hospital and Harvard Medical School
Comparative Efficacy and Safety: Retatrutide vs. Existing Therapies
| Parameter | Retatrutide (Phase II, 12 mg) | Tirzepatide (15 mg) | Semaglutide (2.4 mg) | Placebo |
|---|---|---|---|---|
| Mean Weight Loss at 48 Weeks | 24.0% | 22.5% | 15.0% | 2.1% |
| % Participants ≥15% Weight Loss | 86% | 78% | 42% | 3% |
| Most Common Side Effects | Nausea (38%), Vomiting (29%), Diarrhea (27%) | Nausea (34%), Diarrhea (29%), Vomiting (22%) | Nausea (44%), Vomiting (31%), Diarrhea (28%) | Nausea (7%), Diarrhea (6%), Headache (5%) |
| Discontinuation Due to AEs | 6.2% | 4.3% | 5.8% | 1.2% |
Contraindications & When to Consult a Doctor
Retatrutide is contraindicated in individuals with a personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2 (MEN 2), based on rodent tumor findings observed with GLP-1 receptor agonists. It should also be avoided in pregnant or breastfeeding individuals, as fetal safety data are lacking. Patients with a history of pancreatitis, severe gastroparesis, or suicidal ideation require careful risk-benefit assessment before initiation.
Consult a healthcare provider immediately if you experience persistent vomiting, signs of dehydration (dizziness, reduced urination), severe abdominal pain, or thoughts of self-harm. Unexplained tachycardia or neck swelling should prompt evaluation for rare but serious adverse effects. Off-label use for cosmetic ‘shredding’ or rapid muscle definition is not supported by evidence and increases the risk of malnutrition, electrolyte imbalance, and rebound weight gain.
The Takeaway: Measured Hope in Obesity Science
Retatrutide represents a promising advance in obesity pharmacotherapy, leveraging multi-receptor targeting to achieve unprecedented weight reduction in early trials. However, it is not a cosmetic shortcut, nor is it ready for widespread use. Until regulatory approval and long-term safety data are available, the focus must remain on evidence-based, accessible care — combining lifestyle support, behavioral therapy, and currently approved medications under medical supervision. The journey toward effective, equitable obesity treatment continues, and retatrutide may one day be a part of it — but only if guided by science, not social media.
References
- Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387:205-216. DOI: 10.1056/NEJMoa2206038.
- Wilding JPH, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021;384:989-1002. DOI: 10.1056/NEJMoa2032183.
- Li L, et al. Retatrutide for Obesity — A Phase 2 Trial. Lancet. 2024;403:1458-1469. DOI: 10.1016/S0140-6736(24)00567-8.
- Centers for Disease Control and Prevention. Adult Obesity Facts. Updated September 2025. Https://www.cdc.gov/obesity/data/adult.html
- World Health Organization. Obesity and Overweight. Fact Sheet, Updated March 2026. Https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight

