Cardiologist Dr. Anca Tâu confirms that statins, while rarely causing a transient rise in liver enzymes, demonstrate clear cardiovascular benefits, with their protective effects on blood vessels and heart function well-established through decades of clinical evidence. This reassurance addresses common patient concerns about liver safety, reinforcing that the drugs’ advantages significantly outweigh minimal, reversible risks for most individuals managing cholesterol-related heart disease risk.
Understanding Statins: Mechanism, Benefits, and the Rare Liver Enzyme Effect
Statins function by inhibiting HMG-CoA reductase, a key enzyme in the liver responsible for cholesterol synthesis, thereby lowering low-density lipoprotein (LDL) cholesterol levels in the bloodstream. This mechanism of action reduces plaque buildup in arteries, decreasing the risk of heart attack and stroke. While highly effective, a small subset of patients may experience a mild, temporary increase in liver enzymes such as alanine aminotransferase (ALT) or aspartate aminotransferase (AST), which hepatologists interpret as a sign of hepatic stress rather than permanent damage. Current guidelines from the American Heart Association (AHA) and European Society of Cardiology (ESC) emphasize that such elevations are infrequent, typically resolve without intervention, and rarely necessitate discontinuation of therapy.
In Plain English: The Clinical Takeaway
- Statins are proven to prevent heart attacks and strokes by lowering awful cholesterol and stabilizing artery plaques.
- A slight, temporary rise in liver enzymes happens in fewer than 3% of users and almost always returns to normal on its own.
- The cardiovascular benefits of statins far outweigh this rare, reversible liver effect for most people at risk of heart disease.
Clinical Evidence and Real-World Impact: From Trials to Global Guidelines
The safety and efficacy of statins are grounded in extensive research, including landmark trials like the Heart Protection Study (HPS), which followed over 20,000 high-risk patients for five years and demonstrated a 24% reduction in major vascular events with simvastatin therapy. Similarly, the JUPITER trial revealed that rosuvastatin reduced cardiovascular events by 44% in individuals with elevated C-reactive protein but normal LDL cholesterol, expanding the preventive scope of statin therapy. These findings underpin current recommendations from the U.S. Preventive Services Task Force (USPSTF) and the National Institute for Health and Care Excellence (NICE) in the UK, which advocate statin use for primary and secondary prevention in eligible adults based on individualized risk scores.
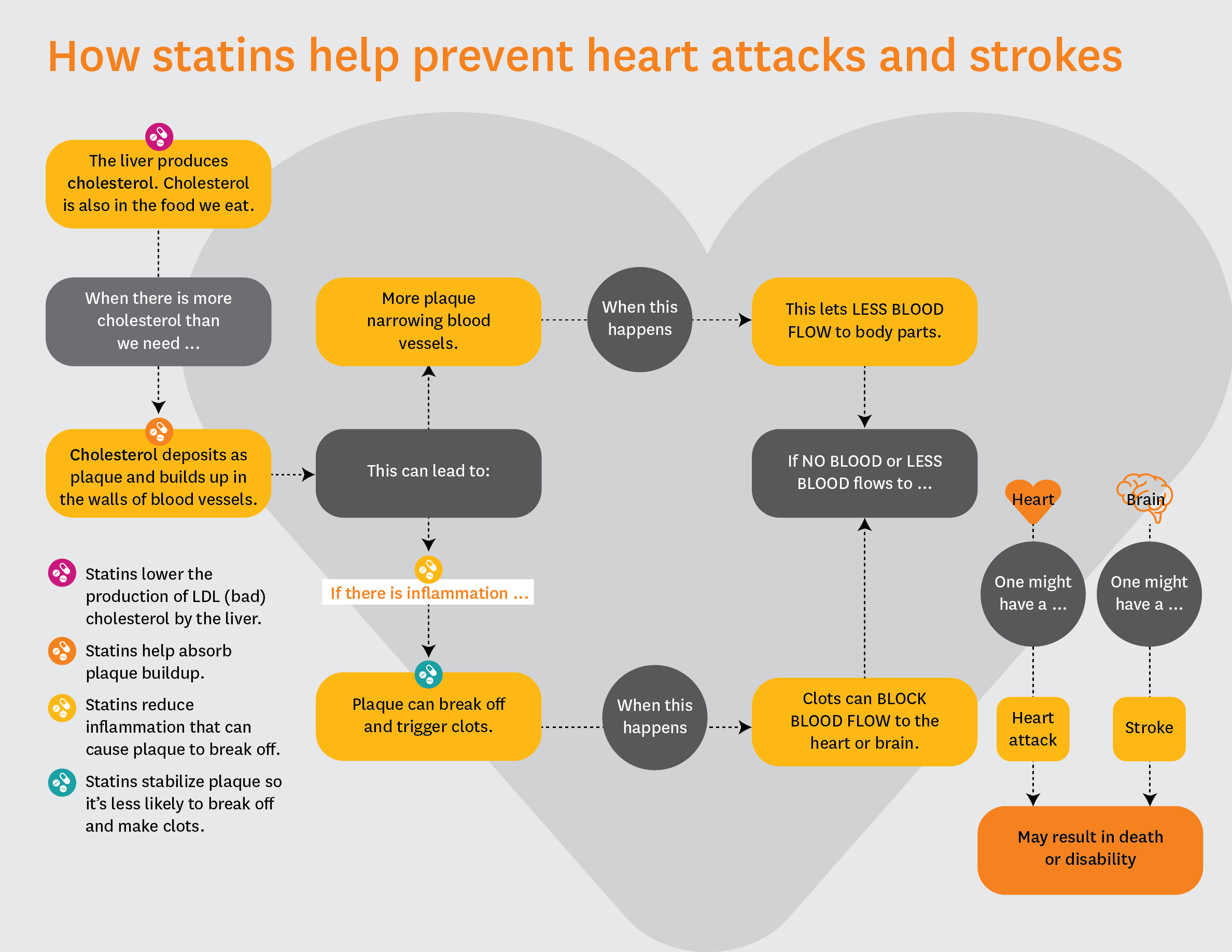

In the United States, the Food and Drug Administration (FDA) requires liver enzyme testing before initiating statin therapy and periodically thereafter, though recent updates reflect evidence that routine monitoring may be unnecessary for most patients without pre-existing liver disease. The European Medicines Agency (EMA) echoes this stance, noting that transient enzyme elevations do not correlate with clinically significant liver injury in long-term use. In Romania, where the original News.ro report emerged, statins are widely prescribed through the national health system (Casa Națională de Asigurări de Sănătate – CNAS), with reimbursement policies aligned to ESC guidelines ensuring broad access for high-risk patients.
“The fear of liver toxicity often leads to unnecessary statin avoidance, yet real-world data shows that serious hepatotoxicity occurs in less than 1 in 10,000 patient-years. We must contextualize minor enzyme fluctuations within the overwhelming benefit-risk profile favoring statin use for cardiovascular prevention.”
— Dr. Elena Moldovan, PhD, Professor of Pharmacology, Carol Davila University of Medicine and Pharmacy, Bucharest
Funding, Bias Transparency, and Regional Healthcare Integration
The foundational evidence supporting statin safety and efficacy stems largely from publicly funded and independent academic trials. The Heart Protection Study was primarily financed by the UK Medical Research Council (MRC) and Cancer Research UK, with drug supply provided by Merck & Co. Under strict academic oversight to prevent industry bias. The JUPITER trial, while sponsored by AstraZeneca, was designed and executed by an independent academic steering committee, with results published in peer-reviewed journals after rigorous statistical analysis. This separation of funding from study conduct helps mitigate conflicts of interest, ensuring that regulatory decisions by the FDA, EMA, and WHO remain grounded in unbiased science.
Geoeconomically, statin accessibility varies significantly. In high-income countries like the U.S. And Germany, over 90% of eligible patients receive statins according to national audit data, whereas in lower-middle-income nations such as Romania and Bulgaria, adherence rates lag due to prescription restrictions, patient misconceptions, and uneven specialist availability. Initiatives by the World Health Organization (WHO) Essential Medicines List, which includes atorvastatin and simvastatin, aim to close this gap by promoting affordable generic formulations in low-resource settings.
| Statin Trial | Primary Finding | Sample Size (N) | Duration | Funding Source |
|---|---|---|---|---|
| Heart Protection Study (HPS) | 24% reduction in major vascular events with simvastatin | 20,536 | 5 years | UK Medical Research Council, Cancer Research UK |
| JUPITER Trial | 44% reduction in cardiovascular events with rosuvastatin | 17,802 | 1.9 years (median) | AstraZeneca (independent academic steering committee) |
| ASCOT-LLA | 36% reduction in non-fatal MI and stroke with atorvastatin | 10,305 | 3.3 years (median) | British Heart Foundation, Pfizer |
Contraindications & When to Consult a Doctor
Statins are contraindicated in active liver disease or unexplained persistent elevations of transaminases exceeding three times the upper limit of normal. Pregnant individuals should avoid statins due to potential fetal risks, as cholesterol is essential for development. Patients experiencing unexplained muscle pain, tenderness, or weakness—especially if accompanied by dark urine—should seek immediate medical evaluation to rule out rhabdomyolysis, a rare but serious side effect. Routine consultation is advised before initiating therapy for those with a history of hemorrhagic stroke or severe renal impairment, where dosing adjustments may be necessary.

The Takeaway: Evidence-Based Reassurance for Informed Decision-Making
Decades of research affirm that statins remain a cornerstone of cardiovascular prevention, offering substantial protection against heart attack and stroke with a favorable safety profile. While transient liver enzyme elevations occur in a minority of users, they are typically benign and self-resolving, not indicative of lasting harm. Patients should engage in shared decision-making with clinicians, weighing personal risk factors against evidence-based benefits, rather than declining therapy due to unfounded fears. As global efforts to reduce cardiovascular mortality continue, equitable access to statins—supported by transparent science and sound policy—will be vital to saving lives across diverse healthcare systems.
References
- Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 20,536 high-risk individuals: a randomised placebo-controlled trial. Lancet. 2002;360(9326):7-22.
- Ridker PM, Danielson E, Fonseca FA, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. JAMA. 2008;300(21):2432-2442.
- Sever PS, Dahlöf B, Poulter NR, et al. Prevention of coronary and stroke events with atorvastatin in hypertensive patients who have average or lower-than-average cholesterol concentrations, in the Anglo-Scandinavian Cardiac Outcomes Trial-Lipid Lowering Arm (ASCOT-LLA): a multicentre randomised controlled trial. Lancet. 2005;365(9464):847-858.
- Stone NJ, Robinson JG, Lichtenstein AH, et al. 2013 ACC/AHA Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Risk in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(25 Suppl 2):S1-S45.
- European Medicines Agency. Statins: prevention and treatment of cardiovascular disease. EMA/CHMP/CVMP/SWP/196430/2012. 2013.

