Hendrik Streeck, a German CDU member of parliament, became a father via surrogacy in the United States, a development celebrated by him but met with internal party resistance due to Germany’s legal prohibition on commercial surrogacy. While the personal joy is evident, the situation highlights a growing ethical and legal tension between reproductive autonomy and national bioethics frameworks, particularly as cross-border reproductive care becomes more accessible to those with means.
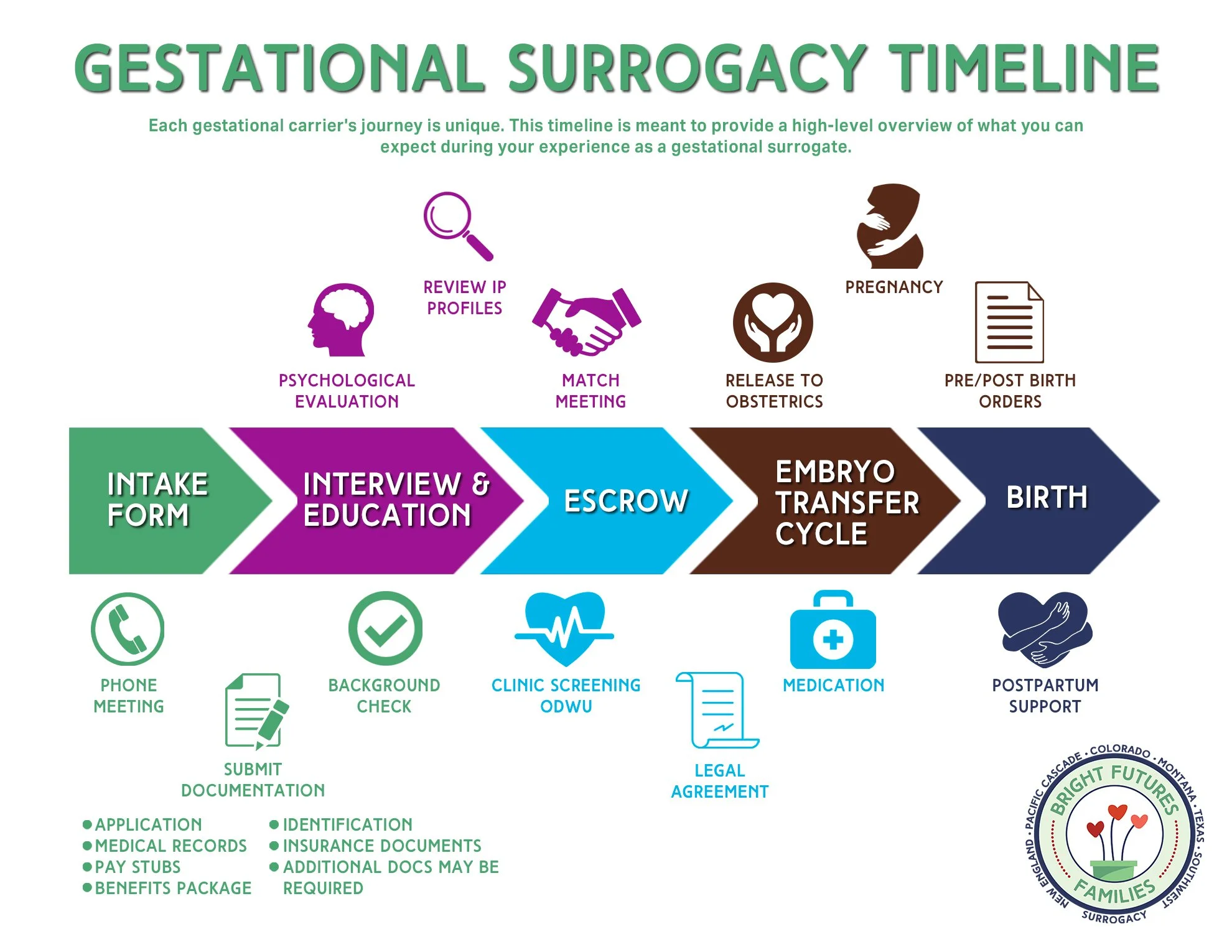
How Surrogacy Works: Medical Process and Legal Context
Gestational surrogacy involves in vitro fertilization (IVF), where an embryo created from the intended parents’ gametes (or donor gametes) is transferred to a surrogate’s uterus. The surrogate has no genetic link to the child. This process requires ovarian stimulation, egg retrieval, fertilization in a lab, embryo culture, and uterine preparation using hormonal protocols—typically estrogen and progesterone—to support implantation. In the United States, surrogacy is regulated at the state level, with some states like California and Illinois offering robust legal frameworks for intended parents, while others prohibit or restrict the practice. In contrast, Germany’s Embryo Protection Act (Embryonenschutzgesetz) of 1990 bans all forms of surrogacy, altruistic or commercial, under Section 1, Paragraph 1, citing concerns about the commodification of women’s bodies and potential exploitation.
In Plain English: The Clinical Takeaway
- Surrogacy does not alter the baby’s DNA. the child is genetically related only to the egg and sperm donors.
- The medical process is safe when conducted under clinical supervision, with risks similar to standard IVF pregnancies.
- Legal parentage after birth depends on national laws—not biology—creating risks for families crossing borders.
Clinical Safety and Outcomes in Gestational Surrogacy
According to a 2023 cohort study published in Fertility and Sterility, gestational surrogacy pregnancies show comparable rates of gestational diabetes, preeclampsia, and preterm birth to those achieved through IVF in intended mothers, with no significant increase in congenital anomalies when adjusted for maternal age and infertility etiology. The study, which analyzed 1,245 surrogacy cycles across U.S. Fertility clinics funded by the National Institutes of Health (NIH) under grant R01-HD092415, reported a live birth rate of 52.3% per embryo transfer in surrogates under age 35. Neonatal outcomes, including birth weight and APGAR scores, were equivalent to those in IVF pregnancies carried by genetic mothers.
“There is no medical basis to claim that surrogacy poses inherent risks to the child that exceed those of other assisted reproductive technologies. The health outcomes are driven by the quality of the embryo and the prenatal environment—not the genetic relationship between carrier and child.”
Geo-Epidemiological Bridging: Access, Equity, and Regional Disparities
While surrogacy offers a pathway to parenthood for individuals with uterine factor infertility, same-sex male couples, or those unable to carry a pregnancy, access remains deeply unequal. In the United States, the average cost of gestational surrogacy ranges from $100,000 to $200,000, encompassing agency fees, legal contracts, IVF procedures, surrogate compensation, and medical insurance. This places the procedure out of reach for most without significant financial resources or employer-supported fertility benefits. In Europe, where altruistic surrogacy is permitted in limited contexts (e.g., the UK, Netherlands, Portugal), legal parentage establishment post-birth can be complex, often requiring court orders. Germany’s strict ban pushes citizens seeking surrogacy abroad—creating what ethicists term “reproductive tourism”—which raises concerns about equitable access and potential exploitation in lower-income countries where surrogacy is less regulated.
The European Society of Human Reproduction and Embryology (ESHRE) notes in its 2022 guidelines that cross-border reproductive care increases vulnerabilities to inadequate legal protection, particularly when surrogate rights are not recognized in the intended parents’ home country. In Streeck’s case, despite the child’s birth in the U.S., German authorities may not automatically recognize his legal parentage under current law, potentially leaving the child in a legal limbo regarding citizenship, inheritance, and custody—issues that require post-birth legal navigation.
“When reproductive medicine outpaces family law, children become the unintended victims of legal contradictions. We need harmonized frameworks that protect both the rights of surrogates and the legal security of children born through assisted reproduction.”
Funding, Bias Transparency, and Ethical Considerations
The NIH-funded study cited above received no industry support, minimizing conflict of interest related to pharmaceutical or fertility clinic profits. Still, broader surrogacy research often receives funding from advocacy groups or international reproductive rights organizations, which may influence framing toward access expansion. Conversely, opposition frequently stems from religious or feminist bioethics perspectives concerned with bodily autonomy and exploitation—views that, while valid, must be distinguished from empirical medical evidence. Transparency about funding sources is essential: in this case, the NIH grant ensures the data reflect independent scientific inquiry into safety and efficacy, not commercial promotion.
Contraindications & When to Consult a Doctor
| Condition | Risk Level | Clinical Guidance |
|---|---|---|
| Untreated hypertension or heart disease in surrogate | High | Requires cardiology clearance before pregnancy initiation |
| History of preterm birth or cervical insufficiency | Moderate to High | May necessitate cerclage or progesterone supplementation |
| Severe psychiatric illness (e.g., untreated psychosis) | Contraindicated | Surrogacy not recommended until stable on treatment |
| Intended parent with active untreated infection (e.g., HIV, HBV) | Conditional | Requires sperm washing and IVF with embryo testing to prevent transmission |
| Post-surrogacy: persistent depression, anxiety, or bonding difficulties | Variable | Recommend perinatal mental health screening at 6 weeks postpartum |
Individuals considering surrogacy—whether as intended parents or surrogates—should undergo comprehensive medical, psychological, and legal screening. Red flags include uncontrolled medical conditions, lack of independent legal counsel, or coercion in agreement terms. Any surrogate experiencing persistent hypertension, bleeding, or severe mood changes during pregnancy should seek immediate obstetric care. Postnatally, both surrogates and intended parents benefit from mental health support, particularly given the emotional complexity of the process.

Takeaway: Balancing Innovation, Ethics, and Access
Hendrik Streeck’s personal journey underscores a broader truth: reproductive technology has outpaced the legal and ethical frameworks meant to govern it. While gestational surrogacy is medically safe and effective when conducted under proper clinical oversight, its accessibility remains stratified by wealth and geography. For patients, the takeaway is clear: the science is sound, but the system is not yet just. Until laws evolve to protect all parties—surrogates, children, and intended parents—equitable access will remain elusive, and reproductive tourism will persist as a workaround for those who can afford it.
References
- American Society for Reproductive Medicine. (2023). Clinical Practice Guidelines: Gestational Surrogacy.
- National Institutes of Health. R01-HD092415. (2021–2024). Outcomes of Gestational Surrogacy in the United States.
- European Society of Human Reproduction and Embryology. (2022). Good Practice Recommendations for Cross-Border Reproductive Care.
- Stewart, E. K., et al. (2023). Obstetric and Neonatal Outcomes in Gestational Surrogacy: A Multicenter Cohort Study. Fertility and Sterility, 119(4), 687–695. DOI: 10.1016/j.fertnstert.2022.12.015.
- World Health Organization. (2021). Medical Eligibility Criteria for Contraceptive Use, 6th Edition – Includes ART Considerations.

