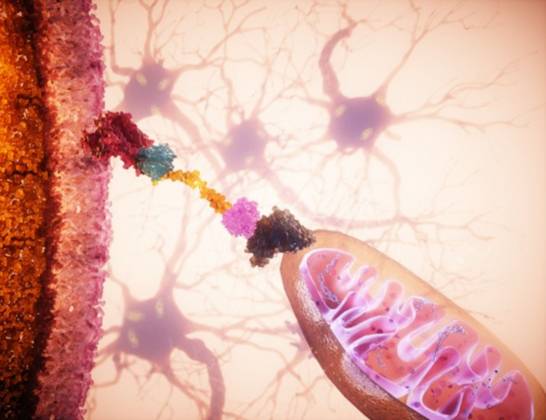
Researchers have developed a “cellular GPS” system capable of directing healthy mitochondria to damaged cells, potentially halting neurodegeneration. By leveraging targeted delivery mechanisms, this breakthrough aims to restore cellular energy production in tissues affected by mitochondrial dysfunction, offering a new therapeutic pathway for metabolic and degenerative diseases.
For decades, the medical community has viewed mitochondrial decay as an inevitable slide toward cellular senescence—the process where cells stop dividing and lose function. When the mitochondria, the “powerhouses” of our cells, fail, the result is often catastrophic, leading to the cognitive decline seen in Alzheimer’s or the muscle wasting of mitochondrial myopathies. This new research transforms the mitochondria from a static organelle into a deliverable therapeutic agent.
In Plain English: The Clinical Takeaway
- Cellular Energy Restore: Instead of just treating symptoms, scientists can now potentially “swap” broken cellular batteries for healthy ones.
- Targeted Delivery: The “GPS” ensures that healthy mitochondria go exactly where they are needed, reducing systemic waste and side effects.
- Disease Reversal: This could shift the goal from “slowing down” brain decay to actively repairing the damage in neurons.
The Molecular Mechanism: How the ‘Cellular GPS’ Navigates the Cytosol
The core of this innovation lies in the mechanism of action—the specific biochemical process through which a drug or therapy produces its effect. In this case, the researchers utilized specialized ligands (molecules that bind to a specific target) to coat healthy mitochondria, effectively tagging them for delivery.

These tagged mitochondria are then guided through the extracellular matrix and across the plasma membrane via endocytosis. Once inside, the “GPS” utilizes the cell’s own microtubule network—the internal scaffolding of the cell—to migrate toward the areas of highest metabolic stress. This is a critical leap forward; previously, mitochondrial transplantation was haphazard and lacked the precision required for clinical application in the human brain.
By restoring the oxidative phosphorylation (the process where cells utilize oxygen to create ATP, or energy), the treatment prevents the trigger of apoptosis, which is programmed cell death. In simpler terms, it stops the cell from “committing suicide” due to a lack of energy.
Global Regulatory Landscapes and Patient Access
While the results are promising, the path from laboratory bench to bedside is governed by rigorous regulatory frameworks. In the United States, the FDA (Food and Drug Administration) would likely classify this as a Biologic or an Advanced Therapy Medicinal Product (ATMP). This means the therapy must undergo three distinct phases of clinical trials to prove safety and efficacy.

In Europe, the EMA (European Medicines Agency) maintains similar strictures, particularly regarding the sourcing of “donor” mitochondria to avoid immunogenic responses—where the body’s immune system attacks the foreign mitochondria. The UK’s NHS is likely to monitor these developments closely, as mitochondrial diseases represent a significant burden on long-term neurological care systems.
The primary hurdle for global access remains the “cold chain” requirement. Because mitochondria are highly sensitive to temperature and pH changes, the logistics of transporting these “live” cellular components to clinics worldwide will require an infrastructure similar to that used for mRNA vaccines.
“The ability to selectively relocate organelles is not just a feat of bioengineering; it is a fundamental shift in how we approach regenerative medicine. We are moving from molecular pharmacology to organelle-based therapy.”
Clinical Comparison: Mitochondrial Transfer vs. Traditional Antioxidants
To understand the impact, we must compare this “GPS” approach to current standards of care, such as the administration of Coenzyme Q10 or other mitochondrial antioxidants.

| Feature | Traditional Antioxidants | Cellular GPS Transfer |
|---|---|---|
| Approach | Chemical support of existing organelles | Physical replacement of damaged organelles |
| Precision | Systemic (affects all cells) | Targeted (cell-specific) |
| Efficacy | Low to Moderate (Symptomatic) | High Potential (Regenerative) |
| Risk Profile | Low / Well-tolerated | Moderate (Immune response risk) |
Funding, Bias, and the Path to Peer Review
Transparency is the bedrock of medical journalism. Much of the foundational research into mitochondrial transfer has been funded by a combination of governmental grants (such as the NIH in the US and ERC in Europe) and private biotech venture capital. While the excitement is palpable, it is essential to note that many of these studies are currently in in vitro (test tube) or animal model stages.
The “Information Gap” in current reporting is the lack of long-term longitudinal data. We do not yet know if the transplanted mitochondria remain stable over years or if the cell eventually rejects them. For a treatment to be viable for a condition like Parkinson’s, the “GPS” must not only deliver the mitochondria but ensure their long-term survival within the host cell.
Contraindications & When to Consult a Doctor
As this technology is currently in the experimental phase, it is not available for general clinical use. However, patients dealing with mitochondrial disorders should be aware of the following:
- Autoimmune Conditions: Patients with systemic lupus erythematosus (SLE) or other autoimmune disorders may be at higher risk for adverse reactions to exogenous organelle transfer.
- Acute Infections: Any therapy involving cellular modulation is typically contraindicated during active systemic infections.
- When to seek support: If you or a loved one are experiencing unexplained muscle weakness, severe ataxia (loss of muscle coordination), or rapid cognitive decline, consult a neurologist immediately to discuss current evidence-based options.
The trajectory of this research suggests a future where we no longer accept the “inevitability” of neurodegeneration. By treating the cell as a modular system where broken parts can be replaced, we are entering the era of precision regenerative medicine. While we must remain objective about the timeline, the scientific foundation is undeniably robust.