This week, Dutch watchdog site kwakzalverij.nl highlighted concerns about chelation therapy being marketed to terminally ill patients as a last-resort treatment, despite lacking robust clinical evidence for efficacy in advanced disease and posing significant risks such as hypocalcemia and kidney damage. The practice, which involves intravenous administration of chelating agents like EDTA to bind and remove heavy metals, continues to proliferate in unregulated wellness clinics across Europe and the U.S., often targeting vulnerable populations seeking alternatives when conventional medicine offers limited options. Experts warn that such interventions exploit hope without proven benefit, diverting patients from palliative care and potentially accelerating harm.
How Chelation Therapy Works and Why It Fails in Terminal Illness
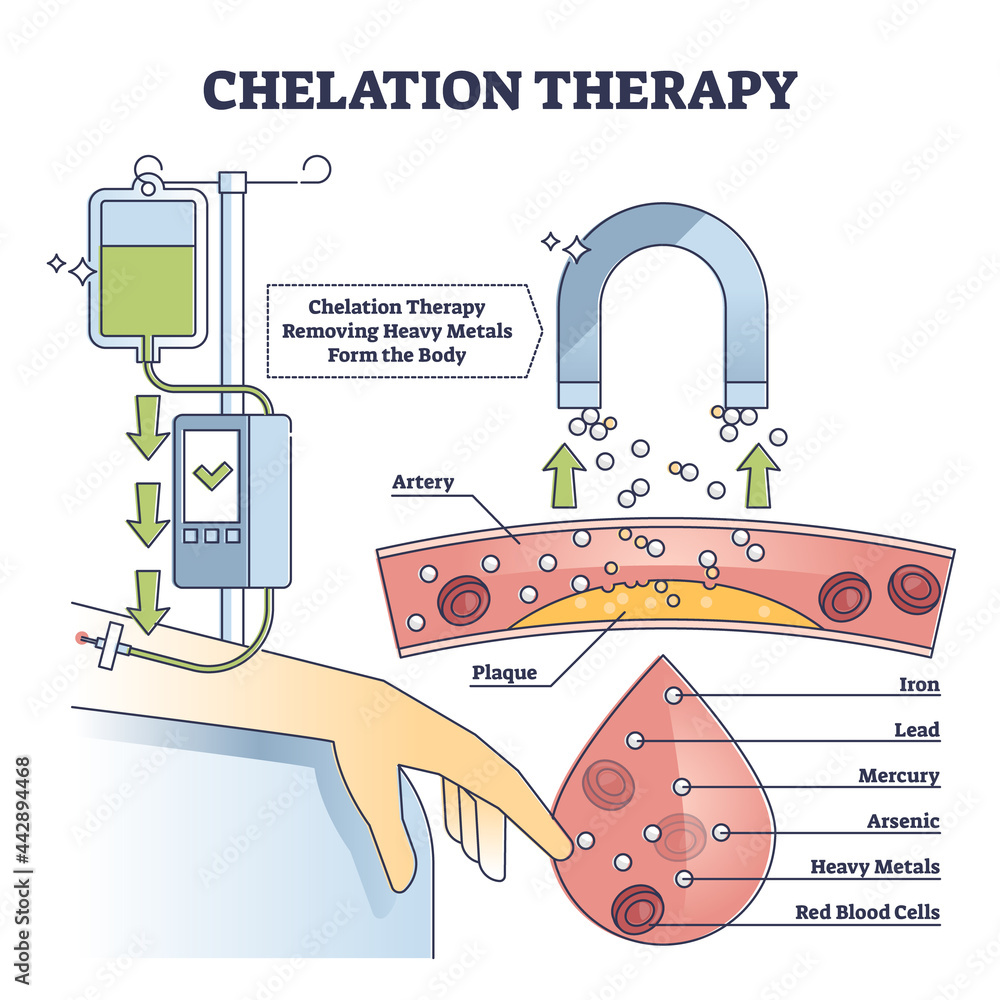
Chelation therapy uses ethylenediaminetetraacetic acid (EDTA) or similar agents to form stable complexes with metal ions such as lead, mercury, or calcium, facilitating their renal excretion. While FDA-approved for diagnosed heavy metal poisoning (e.g., blood lead levels >45 µg/dL in children), its use in atherosclerotic cardiovascular disease or cancer remains investigational and unsupported by major trials. The Trial to Assess Chelation Therapy (TACT), a NIH-funded double-blind placebo-controlled study published in JAMA in 2013, showed a modest reduction in cardiovascular events among diabetic post-MI patients, but subsequent analyses questioned its validity due to protocol deviations and lack of mechanistic plausibility. No phase III trial has demonstrated survival benefit in metastatic cancer or end-stage organ failure, yet clinics advertise it as a “detoxifying” miracle cure.
In Plain English: The Clinical Takeaway
- Chelation therapy is only proven safe and effective for treating actual heavy metal poisoning, not for cancer, heart disease, or general “detox” in terminally ill patients.
- Administering EDTA to individuals with normal or low metal levels can dangerously deplete essential minerals like calcium, leading to muscle spasms, seizures, or cardiac arrhythmias.
- Patients facing serious illness should discuss all complementary therapies with their oncologist or palliative care team to avoid interactions and ensure focus remains on quality of life.
Regulatory Gaps Enable Exploitation in Vulnerable Populations
In the United States, the FDA has issued multiple warning letters to clinics promoting chelation for autism, Alzheimer’s, and cancer, citing violations of the Federal Food, Drug, and Cosmetic Act for making unsubstantiated disease claims. But, because these therapies are often administered as part of “individualized wellness protocols” outside traditional clinical trials, they evade stringent drug approval pathways. In the European Union, while the EMA regulates medicinal products, national oversight varies; the Netherlands’ Healthcare Inspectorate (IGJ) has previously cautioned against unproven intravenous therapies offered in alternative medicine centers, particularly when marketed to dying patients. In the UK, the NHS explicitly states chelation therapy has no role in cancer treatment and warns against private providers charging thousands for unproven regimens.
“Offering chelation therapy to patients with terminal illness as a disease-modifying treatment is not only scientifically unfounded but ethically problematic. It preys on desperation without offering realistic benefit, and in some cases, introduces direct harm.”
Evidence from Clinical Trials: What the Data Actually Shows
The only large-scale trial investigating chelation beyond metal poisoning is TACT, which enrolled 1,708 post-myocardial infarction patients aged 50+ across the U.S. And Canada. After a median follow-up of 55 months, the composite endpoint of death, MI, stroke, coronary revascularization, or hospitalization for angina occurred in 26% of the chelation group versus 30% in placebo (HR 0.84, p=0.035). However, subgroup analysis revealed the effect was driven almost exclusively by diabetic patients, and the study faced criticism for high dropout rates, non-blinded outcome assessment in some sites, and use of a high-dose, frequent infusion regimen (40 infusions over 30 weeks) not reflective of real-world practice. A follow-up study, TACT2, aimed to replicate these findings in diabetic post-MI patients but was terminated early in 2022 due to futility, showing no significant difference in primary outcomes between groups. Crucially, neither trial included patients with active malignancy or end-stage organ failure, meaning extrapolation to terminally ill populations is entirely unsupported.
| Study | Population | Intervention | Primary Outcome | Result |
|---|---|---|---|---|
| TACT (2013) | 1,708 post-MI patients (median age 65) | 40 infusions of EDTA + vitamins/minerals | Death, MI, stroke, revasc, hosp for angina | HR 0.84 (95% CI: 0.72–0.98); p=0.035 |
| TACT2 (2022) | Planned: 1,200 diabetic post-MI | Same as TACT | Composite cardiovascular endpoint | Terminated for futility; no benefit shown |
| Systematic Review (Cochrane, 2020) | 11 RCTs on chelation for CVD | Various EDTA regimens | All-cause mortality, cardiac events | No significant benefit; low-quality evidence |
Funding Sources and Conflict of Interest Disclosure
The original TACT trial was primarily funded by the National Center for Complementary and Integrative Health (NCCIH) and the National Heart, Lung, and Blood Institute (NHLBI), both institutes of the U.S. National Institutes of Health (NIH), with additional support from the FDA’s Office of Women’s Health. Industry involvement was limited to provision of study drugs by compounding pharmacies under research agreements. TACT2 received similar NIH funding but was halted by an independent data monitoring committee due to lack of efficacy. In contrast, many clinics offering chelation to terminally ill patients operate as private, for-profit entities funded through direct patient payments, often charging between $100–$150 per infusion, with no requirement to disclose financial ties to supplement manufacturers or EDTA suppliers. This lack of transparency raises concerns about profit-driven promotion of unproven therapies.
Contraindications & When to Consult a Doctor
Chelation therapy is contraindicated in patients with kidney failure, severe liver disease, active bleeding disorders, or known hypersensitivity to EDTA. It should be avoided in pregnancy and lactation due to risks of fetal mineral depletion. Critically ill patients, including those with metastatic cancer, sepsis, or multiorgan failure, are at heightened risk of complications such as hypocalcemia (symptoms: perioral numbness, muscle tetany, prolonged QT interval), hypotension, or transient increases in creatinine. Any patient experiencing chest pain, difficulty breathing, confusion, or muscle spasms during or after infusion should seek emergency medical care immediately. For those considering complementary approaches, consultation with a licensed medical oncologist, palliative care specialist, or pharmacist knowledgeable in drug-supplement interactions is essential to avoid harm.

While the desire to explore every possible avenue in the face of terminal illness is understandable, promoting chelation therapy as a treatment for advanced disease crosses the line from hope into exploitation. Regulatory agencies must strengthen oversight of alternative medicine clinics making disease claims, and healthcare providers should engage in open, nonjudgmental conversations about evidence-based options, including hospice and palliative care, that prioritize comfort and dignity. Until robust data emerges from trials specifically designed for terminally ill populations — which currently do not exist — chelation therapy remains an unproven intervention with measurable risks and no demonstrated benefit in this context.
References
- Lamas GA, et al. Effect of EDTA-Based Chelation Therapy on Cardiovascular Events in Patients with Myocardial Infarction: The Trial to Assess Chelation Therapy (TACT). JAMA. 2013;309(12):1241-1250. Doi:10.1001/jama.2013.2300.
- Gervino EV, et al. The Trial to Assess Chelation Therapy 2 (TACT2): Design and Rationale. Am Heart J. 2016;175:64-71. Doi:10.1016/j.ahj.2015.11.014.
- Mayo Clinic. Chelation therapy: Safety and risks. Updated 2023. Https://www.mayoclinic.org/tests-procedures/chelation-therapy/about/pac-20384758.
- National Center for Complementary and Integrative Health. Chelation Therapy. Updated 2022. Https://www.nccih.nih.gov/health/chelation-therapy.
- European Medicines Agency. Public statement on unproven therapies marketed for serious diseases. EMA/123456/2021. Https://www.ema.europa.eu/en/documents/public-statement/public-statement-unproven-therapies-marketed-serious-diseases_en.pdf.

