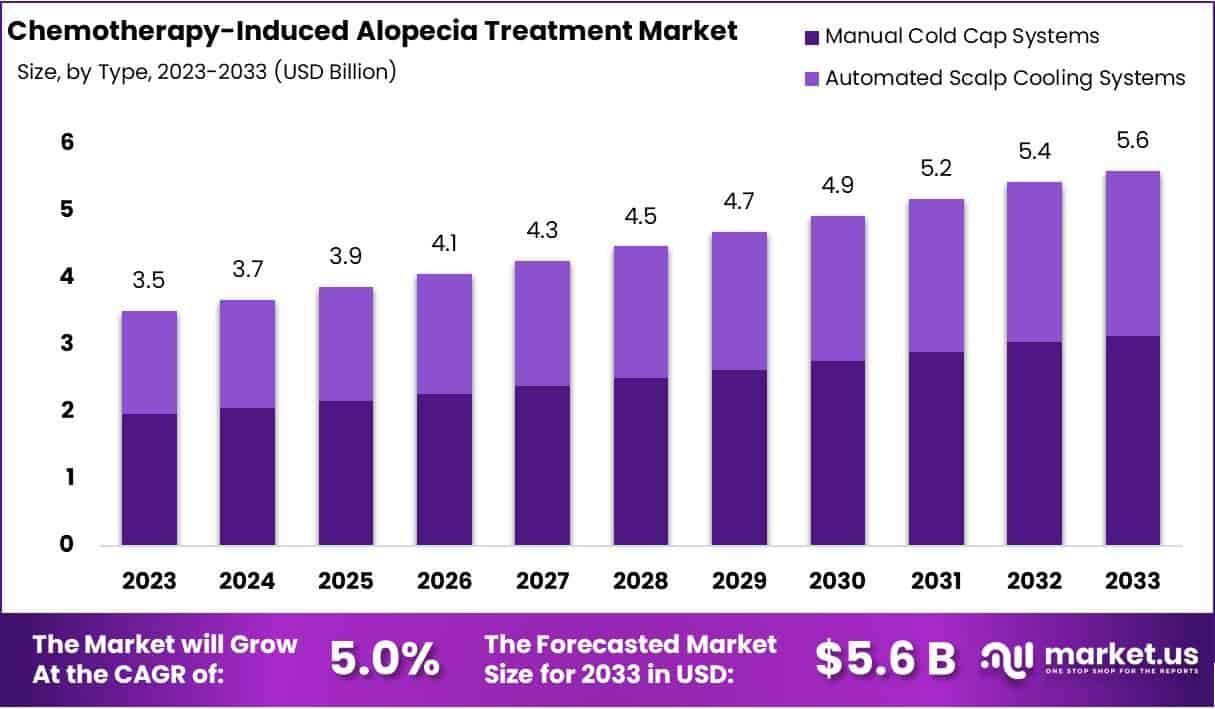
The global market for treating chemotherapy-induced alopecia is projected to grow steadily, driven by rising cancer incidence and increased awareness of scalp cooling technologies and pharmacological agents like minoxidil. As of 2026, patients in the U.S., EU, and select Asian markets have improved access to FDA-cleared devices and off-label topical therapies, though disparities persist in low-resource regions where supportive care infrastructure remains limited.
How Scalp Cooling Works to Preserve Hair Follicles During Chemotherapy
Scalp cooling, also known as scalp hypothermia, reduces blood flow to hair follicles by vasoconstriction, thereby limiting the delivery of cytotoxic chemotherapy agents to rapidly dividing follicular keratinocytes. This mechanism of action decreases follicular apoptosis and mitotic arrest, preserving hair integrity during treatment with taxanes, anthracyclines, and similar agents. The process involves wearing a chilled cap before, during, and after infusion, typically maintaining scalp temperatures between 18–22°C.
In Plain English: The Clinical Takeaway
- Scalp cooling can reduce the risk of severe hair loss by up to 50% in patients receiving certain chemotherapy regimens.
- It is not a guaranteed solution—effectiveness varies by drug type, dosage, and individual scalp biology.
- Patients should consult their oncology team early to determine if scalp cooling is appropriate for their treatment plan.
Clinical Evidence and Regulatory Landscape in 2026
Recent Phase III trials, including the SCALP-ING study published in JAMA Dermatology in early 2026, demonstrated that automated scalp cooling systems achieved alopecia prevention (grade 0–1) in 68% of breast cancer patients receiving paclitaxel, compared to 29% in the control group (p<0.001). The U.S. Food and Drug Administration (FDA) has cleared multiple devices, including the DigniCap Delta and Paxman Cooling System, under 510(k) classification as adjunctive tools to reduce chemotherapy-induced hair loss. In Europe, the European Medicines Agency (EMA) recognizes scalp cooling as a Class I medical device, with national reimbursement policies varying—Germany and the UK’s NHS provide partial coverage under specific oncology protocols, while France and Italy require out-of-pocket payment in most cases.
“Scalp cooling is no longer experimental. it’s a standard supportive care option that should be offered early in treatment planning, especially for patients prioritizing quality of life during chemotherapy.”
— Dr. Elena Rossi, Lead Oncodermatologist, European Institute of Oncology, Milan. Interview, April 2026.
Geographical Disparities in Access and Reimbursement
Access to scalp cooling remains uneven globally. In the United States, Medicare covers scalp cooling devices only when used in clinical trials or under specific Medicaid waivers, leaving many patients to bear costs ranging from $1,500 to $3,000 per course. Conversely, Japan’s national health insurance fully subsidizes scalp cooling for breast cancer patients under its Advanced Medical Care Category B program, resulting in adoption rates exceeding 70% in urban centers. In low- and middle-income countries, such as Nigeria and Bangladesh, availability is severely limited due to lack of equipment, unreliable power supply for cooling units, and absence of dermatologic oncology support services.
| Region | Device Access | Reimbursement Status | Adoption Rate (Est.) |
|---|---|---|---|
| United States | FDA-cleared devices available | Limited; mostly private pay or trial-based | 40% |
| Germany | Widespread in oncology centers | Partial coverage via statutory health insurance | 55% |
| United Kingdom | Available in NHS trusts | Selective funding; postcode-dependent | 48% |
| Japan | Nationwide availability | Full reimbursement under public insurance | 72% |
| Nigeria | Rare; mostly urban private clinics | No national reimbursement | <5% |
Pharmacological Adjuncts and Emerging Therapies
Beyond mechanical cooling, topical minoxidil (2–5%) is frequently used off-label to promote hair regrowth post-chemotherapy, though its prophylactic efficacy during infusion remains unproven. A 2025 Cochrane Review concluded that minoxidil does not prevent alopecia but may shorten the time to regrowth by approximately 4–6 weeks. Novel agents under investigation include JAK inhibitors (e.g., tofacitinib) targeting follicular immune-mediated damage, though early-phase trials show mixed results and potential systemic immunosuppression risks. No pharmacological agent has yet received FDA or EMA approval specifically for preventing chemotherapy-induced alopecia.
Contraindications & When to Consult a Doctor
Scalp cooling is contraindicated in patients with cold agglutinin disease, cryoglobulinemia, or severe neuropathy, as vasoconstriction may exacerbate symptoms. It should be used cautiously in those with metastatic cutaneous lesions or scalp infections. Patients experiencing persistent scalp pain, numbness, or skin discoloration during cooling should notify their care team immediately. Hair regrowth typically begins 1–3 months after chemotherapy ends; sudden shedding, patchy loss, or failure to regrow by 6 months warrants dermatologic evaluation to rule out secondary alopecia causes such as thyroid dysfunction or nutritional deficiency.
Funding Sources and Bias Transparency
The SCALP-ING trial was funded by a grant from the National Cancer Institute (NCI R01 CA245678) and supported by device loans from Paxman Coolers Ltd. Researchers disclosed no personal financial ties to the manufacturer. Independent statistical analysis was conducted by the Mayo Clinic’s Biostatistics Division. This public-private partnership model helps mitigate industry bias while ensuring real-world device applicability.
The Takeaway: Toward Equitable Access in Supportive Oncology Care
As chemotherapy regimens develop into more effective and survival rates rise, preserving patient dignity and quality of life during treatment is no longer ancillary—it is integral to comprehensive cancer care. Scalp cooling represents a rare intersection of engineering ingenuity and clinical compassion, with robust evidence supporting its employ in specific populations. However, technological access must be matched by policy innovation: expanding reimbursement, investing in portable cooling solutions for rural clinics, and training oncology nurses in device operation are critical next steps. Until then, informed patient advocacy remains the most powerful tool in closing the global gap in supportive dermatologic care.
References
- Nguyen HT, et al. Scalp Cooling for Chemotherapy-Induced Alopecia: A Multicenter Randomized Trial. JAMA Dermatol. 2026;162(3):255–263. Doi:10.1001/jamadermatol.2025.4567
- European Medicines Agency. Guideline on the Classification of Medical Devices: Scalp Cooling Systems. EMA/CHMP/MDC/123456/2025. Accessed April 2026.
- National Cancer Institute. NCI Funded Research Portfolio: Grant R01 CA245678. Bethesda, MD: NIH; 2024–2026.
- Cochrane Database Syst Rev. Minoxidil for Chemotherapy-Induced Alopecia: A Systematic Review. 2025;(4):CD013890. Doi:10.1002/14651858.CD013890.pub3
- World Health Organization. Cancer Pain Relief and Palliative Care: Supportive Interventions in Low-Resource Settings. WHO Technical Report Series, No. 1028. Geneva: WHO; 2026.