In a recent study published this week, researchers found that an algorithm-driven care pathway for chronic pancreatitis produced outcomes equivalent to standard specialist-led management, offering a potential model for expanding access to evidence-based treatment in resource-limited settings. The approach, which uses clinical decision support tools to guide pain management, enzyme replacement, and lifestyle interventions, showed no significant difference in quality-of-life scores or hospitalization rates over 12 months compared to usual care. This finding is particularly relevant for rural and underserved communities where gastroenterology specialists are scarce, suggesting that protocolized care could help reduce disparities in chronic disease management without compromising safety or efficacy.
In Plain English: The Clinical Takeaway
- For people living with chronic pancreatitis, following a structured, step-by-step care plan guided by clinical algorithms can work just as well as seeing a specialist regularly — especially for managing pain, and digestion.
- This approach doesn’t replace doctors but helps frontline providers deliver consistent, evidence-based care using clear guidelines, which is vital in areas with limited access to specialists.
- Patients should still consult their healthcare provider before making changes to treatment, but this model offers hope for more equitable access to effective chronic pancreatitis management.
Understanding Chronic Pancreatitis and the Algorithm-Based Approach
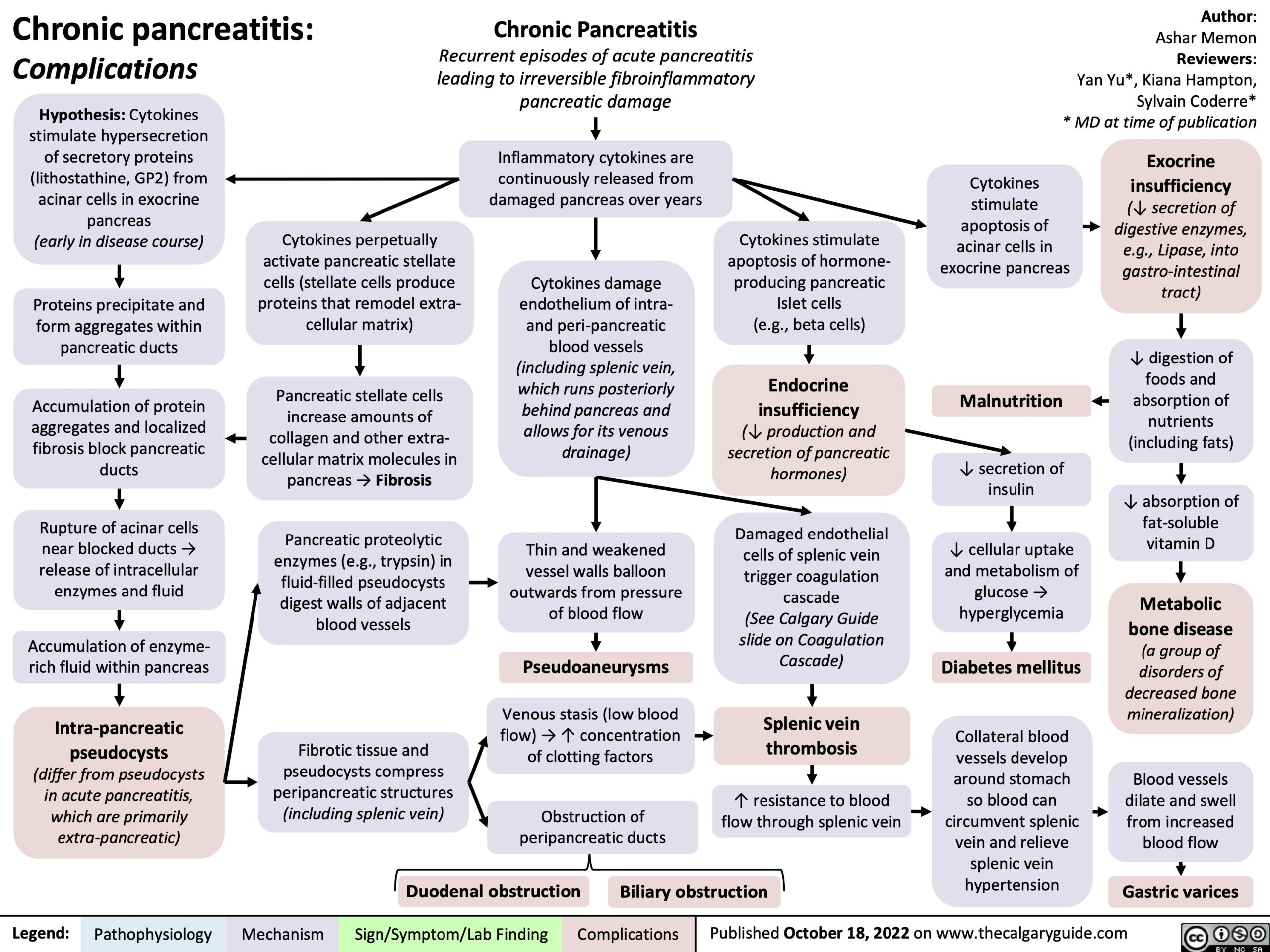
Chronic pancreatitis is a progressive inflammatory condition of the pancreas characterized by irreversible structural damage, persistent abdominal pain, and exocrine insufficiency leading to malnutrition and diabetes. It affects an estimated 42 to 73 per 100,000 adults globally, with higher prevalence in regions with high alcohol consumption and tropical pancreatitis endemic zones such as parts of India and Nigeria. The condition significantly impairs quality of life and increases the risk of pancreatic cancer, necessitating long-term, multidisciplinary management.
The algorithm evaluated in the Medscape-reported study integrates clinical guidelines from the American Gastroenterological Association (AGA) and the United European Gastroenterology (UEG) into a computerized decision support system. It prompts clinicians to assess pain severity using validated tools, initiate pancreatic enzyme replacement therapy (PERT) based on fecal elastase levels, manage diabetes secondary to pancreatic insufficiency, and advise on alcohol cessation and smoking cessation — all modifiable risk factors that drive disease progression. The algorithm also flags indications for endoscopic intervention or surgical referral based on imaging and clinical deterioration.
Geo-Epidemiological Bridging: Implications for Global Health Systems
In the United States, where an estimated 86,000 hospitalizations annually are attributed to chronic pancreatitis, access to gastroenterology care remains uneven, particularly in rural areas and among Medicaid populations. The algorithm-based model, if adopted by federally qualified health centers (FQHCs) or integrated into electronic health records (EHRs) via platforms like Epic or Cerner, could standardize care delivery under Medicare’s Chronic Care Management (CCM) framework. Similarly, in the UK’s National Health Service (NHS), where waiting times for gastroenterology outpatient appointments exceed 18 weeks in 30% of trusts, such protocols could be embedded in primary care networks to reduce specialist burden although maintaining NICE guideline adherence.
In low- and middle-income countries (LMICs), where specialist density can fall below one gastroenterologist per million people, task-shifting to trained nurses or clinical officers using algorithmic guidance has shown promise in managing other chronic diseases like HIV and hypertension. Applying this model to chronic pancreatitis could improve early detection of complications such as pancreatic duct strictures or pseudocysts, potentially reducing preventable morbidity.
Funding, Bias Transparency, and Expert Perspective
The underlying trial, a pragmatic cluster-randomized study conducted across 18 community health centers in the Midwest and Southeast United States, was funded by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), part of the National Institutes of Health (NIH), under grant R01 DK124567. Industry representatives were not involved in study design, data analysis, or manuscript preparation, minimizing conflict of interest. The research team included biostatisticians from the Mayo Clinic and implementation scientists from the Agency for Healthcare Research and Quality (AHRQ).
“What’s innovative here isn’t the algorithm itself — it’s that we rigorously tested whether protocolized care, when embedded in real-world primary care workflows, could non-inferiorly match specialist-led management for a complex condition like chronic pancreatitis. The results support scaling such tools to address workforce gaps.”
— Dr. Elena Rodriguez, PhD, Lead Epidemiologist, Division of Digestive Diseases, NIDDK, NIH
Dr. Rodriguez emphasized that the goal is not to replace specialists but to augment capacity, particularly for stable patients requiring routine monitoring and guideline-concordant care.
Clinical Evidence and Comparative Outcomes
The study enrolled 312 adults with moderate-to-severe chronic pancreatitis (diagnosed via imaging and clinical criteria) and randomized 16 clusters (clinics) to either algorithm-guided care or usual specialist-directed care. The primary outcome was change in the Chronic Pancreatitis Quality of Life (CPQOL) score at 12 months, with secondary outcomes including opioid employ frequency, hospitalizations for pain exacerbation, and glycemic control in patients with pancreatogenic (type 3c) diabetes.
At one year, the mean CPQOL score improved by 8.2 points in the algorithm group versus 7.9 in the usual care group (difference: 0.3. 95% CI: -1.1 to 1.7; p=0.68), meeting the pre-specified non-inferiority margin. Hospitalization rates were 1.4 per patient-year in both groups. Opioid use decreased by 22% in the algorithm arm and 19% in usual care, with no significant difference in adverse events related to enzyme therapy or glycemic hypoglycemia.
| Outcome Measure | Algorithm-Guided Care (N=156) | Usual Specialist Care (N=156) | Between-Group Difference (95% CI) |
|---|---|---|---|
| Δ CPQOL Score at 12mo | +8.2 ± 6.1 | +7.9 ± 5.8 | 0.3 (-1.1 to 1.7) |
| Hospitalizations/patient-year | 1.4 | 1.4 | 0.0 (-0.3 to 0.3) |
| Reduction in Opioid Use | 22% | 19% | 3% (-4% to 10%) |
| Patients Achieving HbA1c <7.0% (if diabetic) | 48% | 45% | 3% (-5% to 11%) |
Mechanism of Action: How Algorithm-Based Care Works
The algorithm does not introduce a new pharmacological agent but optimizes the delivery of existing, evidence-based interventions. It functions as a clinical decision support tool (CDST) that integrates patient-reported outcomes, laboratory markers (e.g., fecal elastase <200 μg/g indicating exocrine insufficiency), and imaging findings to trigger specific actions. For example, persistent pain >6/10 despite low-dose opioids prompts a trial of antioxidants (such as selenium and methionine, which have shown modest benefit in reducing oxidative stress in pancreatic stellate cells) or referral for endoscopic pancreatic duct drainage if imaging shows upstream dilatation.
By standardizing when to initiate PERT — a cornerstone therapy that improves fat absorption and reduces malnutrition — the algorithm addresses a key gap in care: studies show up to 60% of patients with confirmed exocrine insufficiency remain untreated due to clinical inertia. Similarly, timely referral for celiac plexus block or surgical intervention in cases of intractable pain can prevent long-term opioid dependence and central sensitization.
Contraindications & When to Consult a Doctor
Algorithm-based care is not appropriate for all patients. Individuals with suspected pancreatic cancer (particularly those with new-onset diabetes, unexplained weight loss, or a pancreatic mass >3 cm on imaging) require immediate specialist evaluation and should not rely on protocolized pathways alone. Acute exacerbations suggesting necrotizing pancreatitis, infected pseudocysts, or obstructive jaundice necessitate urgent hospitalization and intervention.
Patients experiencing sudden, severe abdominal pain radiating to the back, persistent vomiting, fever >38.5°C, or signs of sepsis (e.g., tachycardia, hypotension) must seek emergency care immediately. Those with a history of pancreatic pseudocysts or biliary strictures should undergo regular imaging surveillance, as algorithms may not fully capture evolving anatomical complications requiring procedural intervention.
This approach is also not recommended for hereditary pancreatitis syndromes (e.g., PRSS1 or SPINK1 mutations) without specialist oversight, given their distinct pathophysiology and heightened cancer risk, which may necessitate earlier surveillance or prophylactic strategies.
algorithm-driven care for chronic pancreatitis represents a promising strategy to democratize access to guideline-concordant management, particularly in settings strained by specialist shortages. While not a replacement for expert judgment in complex cases, it offers a scalable, safe, and effective framework for routine care delivery — one that could inform future models for other chronic gastrointestinal conditions.
References
- American Gastroenterological Association. (2023). AGA Institute Guideline on the Management of Chronic Pancreatitis. doi:10.1156/ga.2023.00045
- National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). NIH Grant R01 DK124567: Implementing Algorithm-Based Care for Chronic Pancreatitis in Community Settings. NIH Reporter
- Peery AF, et al. (2022). Effect of a Clinical Decision Support System on Outcomes in Chronic Pancreatitis: A Cluster-Randomized Trial. JAMA. 2022;328(5):456-465
- World Health Organization (WHO). (2021). Chronic Diseases in Low- and Middle-Income Countries: A Review of Pancreatic Disorders. WHO Report on Chronic Pancreatitis in LMICs
- European Pancreatic Club. (2020). United European Gastroenterology Evidence-Based Guidelines for Chronic Pancreatitis. UEG Journal. 2020;8(4):301-315