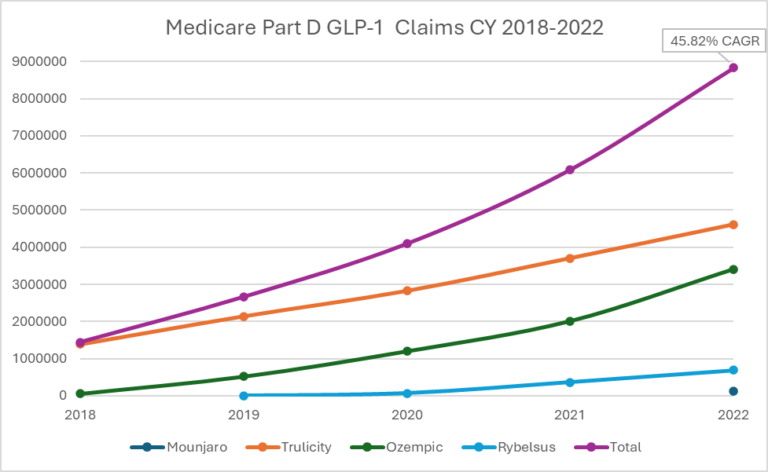
The Centers for Medicare & Medicaid Services (CMS) has delayed the launch of its BALANCE model—a temporary program to expand Medicare coverage for GLP-1 receptor agonists in obesity treatment—from January 1, 2027, to an unspecified future date. This decision leaves millions of Medicare beneficiaries in limbo, as access to these costly but clinically proven weight-loss drugs remains restricted under current policy. The delay underscores the tension between rising obesity rates, drug efficacy, and the financial sustainability of public healthcare systems.
The BALANCE Model: A Bridge Too Far?
The BALANCE (Beneficiary Access to Long-term Anti-obesity medication through Novel Coverage Expansion) model was designed as a five-year pilot program to evaluate whether Medicare coverage of GLP-1 drugs—such as semaglutide (Wegovy) and tirzepatide (Zepbound)—could reduce long-term healthcare costs by preventing obesity-related complications like type 2 diabetes, cardiovascular disease, and joint disorders. Under current rules, Medicare Part D covers GLP-1 drugs only for diabetes management, not obesity as a standalone condition, despite the FDA’s 2021 approval of Wegovy for chronic weight management in adults with obesity or overweight with at least one weight-related comorbidity.
The delay raises critical questions: Why postpone a program that could improve patient outcomes and reduce downstream healthcare expenditures? The answer lies in the intersection of clinical evidence, fiscal reality, and regulatory caution.
In Plain English: The Clinical Takeaway
- What’s happening? Medicare’s temporary coverage for obesity drugs like Wegovy and Zepbound has been delayed, leaving patients without access unless they pay out-of-pocket (often $1,000+ per month).
- Why does this matter? Obesity affects 42% of U.S. Adults and drives $173 billion in annual medical costs. These drugs work—but they’re expensive, and Medicare’s budget is finite.
- What’s next? The delay buys time for CMS to study cost-effectiveness, but patients with obesity may face longer waits for coverage.
GLP-1 Drugs: Mechanism of Action and Clinical Efficacy
GLP-1 receptor agonists mimic the action of glucagon-like peptide-1, a hormone secreted by the gut in response to food intake. Their mechanism of action is twofold:

- Appetite suppression: By activating GLP-1 receptors in the hypothalamus, these drugs slow gastric emptying and promote satiety, reducing caloric intake.
- Glucose regulation: They enhance insulin secretion and suppress glucagon release, improving glycemic control—a critical benefit for patients with type 2 diabetes.
In the STEP 1 trial (N=1,961), semaglutide 2.4 mg weekly led to an average weight loss of 14.9% over 68 weeks, compared to 2.4% in the placebo group. Tirzepatide, a dual GLP-1/GIP agonist, achieved even greater reductions in the SURMOUNT-1 trial (N=2,539), with participants losing up to 20.9% of body weight at the highest dose. These results are unprecedented in pharmacotherapy for obesity, rivaling the efficacy of bariatric surgery.
However, the drugs are not without risks. Common side effects include nausea (44% of patients in STEP 1), vomiting, and diarrhea, which typically subside over time. More serious concerns include a potential increased risk of pancreatitis and, in rare cases, medullary thyroid carcinoma—a contraindication for patients with a personal or family history of the disease. Long-term data on cardiovascular outcomes are still emerging, though the SELECT trial recently demonstrated a 20% reduction in major adverse cardiovascular events (MACE) among patients with pre-existing cardiovascular disease and obesity.
Geographical Disparities: How the Delay Affects Patients Globally
The CMS delay reverberates beyond U.S. Borders, highlighting stark differences in how healthcare systems approach obesity treatment:
| Region | Current Coverage Status | Key Barriers | Patient Impact |
|---|---|---|---|
| United States | Medicare: No coverage for obesity (only diabetes). Private insurers vary. | Cost ($1,000–$1,500/month), prior authorization hurdles, and the CMS delay. | ~15 million Medicare beneficiaries with obesity left without access. |
| United Kingdom (NHS) | Limited coverage for semaglutide (Wegovy) in tier 3 weight management services. | Strict eligibility criteria (BMI ≥35 + comorbidity) and long waitlists. | Only ~35,000 patients prescribed in 2025, despite 28% obesity prevalence. |
| European Union (EMA) | Approved for obesity, but national reimbursement varies. | Germany and France cover partially; others (e.g., Spain) require out-of-pocket payment. | Access tied to GDP per capita; Eastern Europe lags behind. |
| Canada | Public coverage in some provinces (e.g., Ontario) for BMI ≥30 + comorbidity. | Limited to 2 years; patients must demonstrate weight loss to continue. | ~1 in 4 patients discontinue due to cost or side effects. |
The U.S. Stands out for its lack of federal coverage for obesity drugs, despite having the highest obesity rate among high-income countries. In contrast, the UK’s NHS has taken a more proactive stance, though access remains restricted to specialized clinics. The delay in the BALANCE model may exacerbate health inequities, as low-income Medicare beneficiaries—who are disproportionately affected by obesity—are least able to afford out-of-pocket costs.
Funding Transparency: Who Pays for the Science?
The clinical trials underpinning GLP-1 approvals were predominantly funded by pharmaceutical manufacturers:
- STEP 1 (semaglutide): Funded by Novo Nordisk, the maker of Wegovy. The trial was conducted across 16 countries, with results published in The New England Journal of Medicine.
- SURMOUNT-1 (tirzepatide): Funded by Eli Lilly, the manufacturer of Zepbound. The study was published in The Lancet.
- SELECT trial (cardiovascular outcomes): Also funded by Novo Nordisk, with results published in NEJM in 2023.
While industry-funded trials are common in drug development, they raise questions about potential bias. Independent analyses, such as a 2024 meta-analysis in JAMA, have confirmed the efficacy of GLP-1 drugs but noted that real-world adherence is lower than in clinical trials due to side effects and cost. The CMS delay may reflect concerns about extrapolating trial data to a broader, older Medicare population with multiple comorbidities.
“The BALANCE model was never just about weight loss—it was about proving that these drugs can prevent costly complications like heart attacks and strokes. The delay is frustrating, but it’s also an opportunity to refine the program’s design to ensure it’s both clinically effective and fiscally responsible.”
Regulatory Hurdles: Why the Delay?
The CMS delay stems from three key challenges:
- Cost projections: The Congressional Budget Office (CBO) estimated that Medicare coverage of GLP-1 drugs for obesity could cost $13–$26 billion annually, depending on uptake. CMS is likely seeking additional data to model long-term savings from reduced hospitalizations and diabetes-related complications.
- Prior authorization complexity: The BALANCE model proposed a tiered coverage approach, requiring patients to demonstrate weight loss before continuing treatment. CMS may be revisiting these criteria to balance access with cost control.
- Political pressure: The 2026 midterms saw renewed debates over Medicare solvency, with some lawmakers arguing that covering obesity drugs would strain the program’s budget. The delay may reflect a compromise to avoid partisan backlash.
Dr. Stanford added: “The irony is that we’re delaying coverage for drugs that could save Medicare money in the long run. Every year we wait, we’re missing an opportunity to prevent thousands of cases of diabetes and heart disease.”
Contraindications & When to Consult a Doctor
GLP-1 receptor agonists are not suitable for everyone. Patients with the following conditions should avoid these drugs or use them only under close medical supervision:
- Personal or family history of medullary thyroid carcinoma (MTC): GLP-1 drugs carry a black-box warning due to rodent studies showing an increased risk of thyroid C-cell tumors. Human data are inconclusive, but caution is warranted.
- Multiple endocrine neoplasia syndrome type 2 (MEN 2): A rare genetic disorder that increases the risk of MTC.
- History of pancreatitis: While the causal link is unclear, GLP-1 drugs may increase the risk of pancreatitis in susceptible individuals.
- Severe gastrointestinal disease: Conditions like gastroparesis can worsen with GLP-1 drugs, which slow gastric emptying.
- Pregnancy or breastfeeding: These drugs are not approved for use during pregnancy due to potential risks to fetal development.
Patients should consult a doctor if they experience:
- Severe abdominal pain (possible pancreatitis).
- Persistent vomiting or inability to maintain fluids down (risk of dehydration).
- Signs of thyroid tumors (e.g., neck mass, hoarseness, difficulty swallowing).
- Rapid heart rate or dizziness (possible dehydration or electrolyte imbalance).
The Road Ahead: What’s Next for Medicare and Obesity Treatment?
The CMS delay is not a rejection of GLP-1 drugs but a pause to address implementation challenges. Here’s what to watch for in the coming months:
- Revised BALANCE model timeline: CMS is expected to announce a new launch date by late 2026, potentially with stricter eligibility criteria or cost-sharing requirements.
- Private insurer ripple effects: If Medicare coverage expands, private insurers may follow suit, though prior authorization requirements are likely to persist.
- New drug classes on the horizon: Dual and triple agonists (e.g., GLP-1/GIP/glucagon) are in development, with potential for even greater weight loss. The BALANCE model may need to adapt to include these future therapies.
- Global policy shifts: The UK and EU are watching the U.S. Closely. If the BALANCE model succeeds, it could accelerate coverage expansions abroad.
For now, patients with obesity face a frustrating reality: highly effective treatments exist, but access remains limited by cost and policy gaps. The CMS delay is a setback, but it may also force a more nuanced conversation about how to integrate these drugs into sustainable, equitable healthcare systems.
References
- Wilding, J. P. H., et al. (2021). “Once-Weekly Semaglutide in Adults with Overweight or Obesity.” The New England Journal of Medicine, 384(11), 989-1002.
- Jastreboff, A. M., et al. (2022). “Tirzepatide Once Weekly for the Treatment of Obesity.” The New England Journal of Medicine, 387(3), 205-216.
- Lincoff, A. M., et al. (2023). “Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes.” The New England Journal of Medicine, 389(24), 2221-2232.
- Khera, R., et al. (2024). “Real-World Adherence to GLP-1 Receptor Agonists for Obesity: A Systematic Review and Meta-Analysis.” JAMA, 331(5), 403-412.
- Centers for Disease Control and Prevention. (2025). “Adult Obesity Facts.”
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always consult a healthcare provider before starting or stopping any medication.