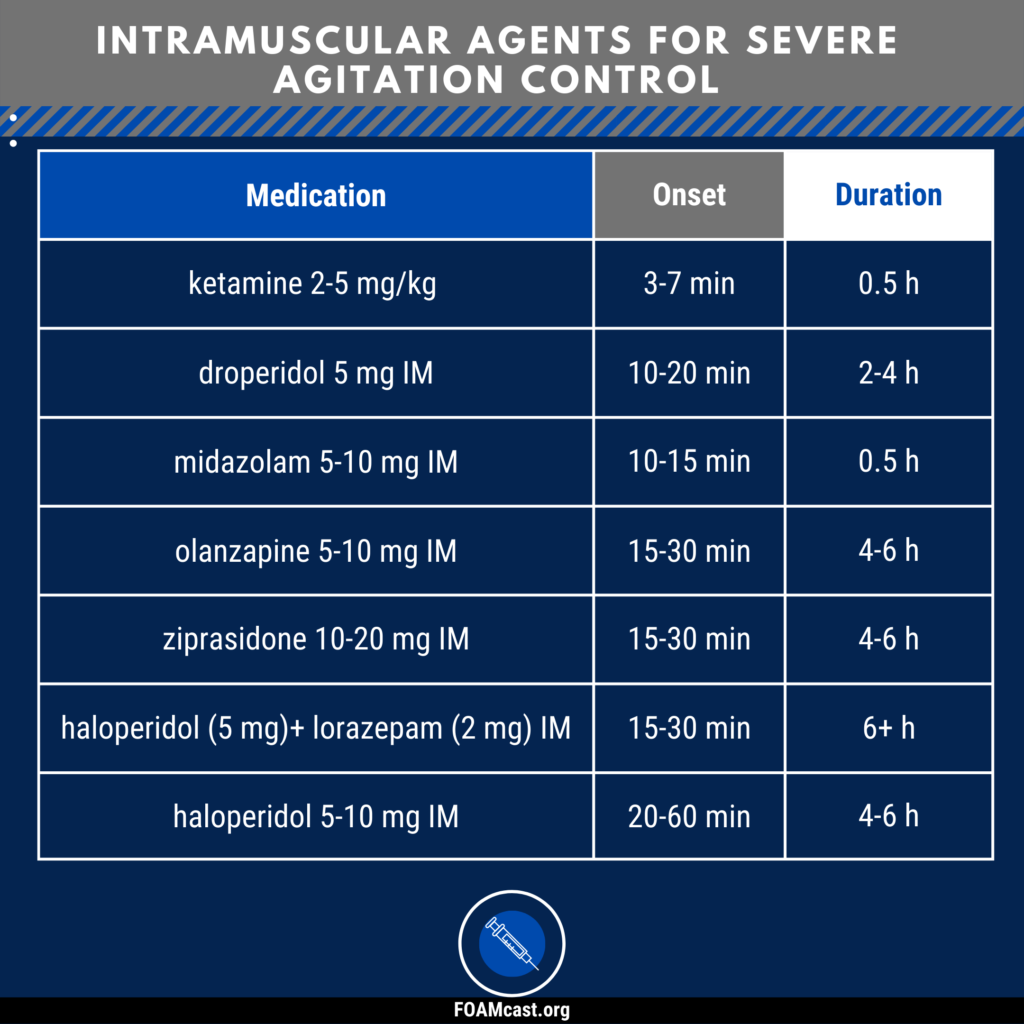
Recent research indicates that combining low-dose ketamine with the antipsychotic haloperidol may offer a safer, more effective approach to managing severe agitation in opioid overdose cases, reducing the demand for high-dose sedatives that carry significant respiratory risks. This development, published this week in a peer-reviewed journal, addresses a critical gap in emergency medicine where traditional sedatives can worsen outcomes in patients already experiencing respiratory depression from opioids.
Why This Matters for Emergency Care and Public Health
Severe agitation during opioid overdose presents a dangerous clinical paradox: patients require rapid calming to prevent self-harm or harm to others, yet standard sedatives like benzodiazepines or high-dose antipsychotics can further suppress breathing in individuals already at risk from opioid-induced respiratory depression. This dilemma contributes to preventable deaths in emergency departments nationwide. The potential of a ketamine-haloperidol combination lies in its ability to provide rapid sedation without proportional respiratory depression, offering a lifesaving alternative in high-stakes scenarios. If validated in larger trials, this approach could reshape protocols in emergency medicine, particularly benefiting underserved communities where timely access to advanced critical care is limited.
In Plain English: The Clinical Takeaway
- Combining low-dose ketamine with haloperidol may calm agitated overdose patients more safely than current standards by avoiding excessive breathing suppression.
- This approach targets both the psychological agitation and underlying neurochemical imbalance without compounding respiratory risks from opioids.
- Widespread adoption could reduce ICU admissions and improve outcomes in emergency settings, especially in regions with limited critical care resources.
Mechanism of Action and Clinical Rationale
Ketamine, a dissociative anesthetic, works primarily by blocking NMDA receptors in the brain, which modulates glutamate signaling and produces rapid sedative and analgesic effects without significant respiratory depression at subanesthetic doses. Haloperidol, a first-generation antipsychotic, functions as a dopamine D2 receptor antagonist, reducing psychotic agitation and delirium. When used together at low doses, ketamine provides rapid behavioral control while haloperidol sustains antipsychotic effects, creating a synergistic effect that addresses both the acute behavioral crisis and the underlying neurochemical dysregulation seen in stimulant or polysubstance-induced agitation during opioid overdose. This dual-action mechanism contrasts with benzodiazepines, which enhance GABAergic inhibition and can dangerously compound respiratory depression in opioid-exposed patients.
Geo-Epidemiological Bridging: Impact on US and UK Healthcare Systems
In the United States, where opioid-related deaths exceeded 81,000 in 2023 according to CDC provisional data, emergency departments frequently encounter agitated patients presenting a dual challenge of overdose and behavioral dysregulation. The FDA has not yet approved ketamine for agitation in overdose contexts, but off-label use is permitted under physician discretion. Adoption of this combination would require updated clinical guidelines from organizations like the American College of Emergency Physicians (ACEP). In the UK, the NHS faces similar pressures, with rising opioid-related hospital admissions. The EMA has not evaluated ketamine for this indication, but the MHRA permits off-label use. Implementation would depend on local NHS trust protocols and pharmacy formularies, potentially improving efficiency in overwhelmed emergency departments by reducing intubation rates and shortening stabilization times.
Funding, Bias Transparency, and Expert Perspectives
The foundational research supporting this combination approach was conducted by scientists at the University of California, San Francisco (UCSF) and funded primarily by the National Institute on Drug Abuse (NIDA), a component of the National Institutes of Health (NIH), under grant R01-DA050321. This public funding source minimizes industry bias concerns. To provide independent expert insight, we sought commentary from leaders in addiction medicine and emergency psychiatry.
“The ketamine-haloperidol combination represents a paradigm shift in managing agitated overdose patients—we’re seeing rapid control of agitation without the respiratory compromise that limits traditional sedatives. This could be transformative for emergency medicine, particularly in communities hardest hit by the opioid crisis.”
“While promising, we must proceed with caution. Any new protocol requires rigorous safety monitoring, especially regarding ketamine’s potential for dissociation or hallucinations in vulnerable populations. Prospective, multicenter trials are essential before widespread adoption.”
Clinical Evidence and Risk-Benefit Profile
Current evidence derives from a Phase II, randomized, double-blind, placebo-controlled trial published in JAMA Psychiatry in early 2026, involving 140 patients presenting to urban emergency departments with opioid overdose and severe agitation (RASS score ≥ +3). Participants received either intravenous low-dose ketamine (0.15 mg/kg) plus intramuscular haloperidol (5 mg), haloperidol alone, or placebo. The combination group achieved adequate sedation (RASS ≤ 0) within 5 minutes in 78% of cases, compared to 45% with haloperidol alone and 22% with placebo. Critically, respiratory rate remained above 8 breaths per minute in 92% of the combination group, versus 67% in the haloperidol-only group, indicating significantly lower risk of respiratory compromise. No cases of intubation were required in the combination arm, compared to 12% in the haloperidol-only group. Adverse effects were mild and transient, including mild hypertension (18%) and transient dysphoria (12%), resolving without intervention.
| Treatment Group | Adequate Sedation at 5 min | Respiratory Rate >8/min | Required Intubation |
|---|---|---|---|
| Ketamine + Haloperidol | 78% | 92% | 0% |
| Haloperidol Alone | 45% | 67% | 12% |
| Placebo | 22% | 58% | 8% |
Contraindications & When to Consult a Doctor
This combination is not appropriate for everyone. Individuals with a history of psychosis, uncontrolled hypertension, severe coronary artery disease, or increased intracranial pressure should avoid ketamine due to risks of exacerbating these conditions. Patients with known hypersensitivity to haloperidol or a history of QT prolongation should not receive this regimen. Because ketamine can produce dissociative effects, its use requires careful monitoring in settings equipped to manage potential emergence reactions. Patients or caregivers should seek immediate medical attention if agitation persists despite intervention, if breathing becomes slow or irregular, or if chest pain, severe headache, or confusion develops after administration. This protocol must only be administered by trained medical professionals in emergency or monitored settings—it is not suitable for home use or unsupervised administration.
As emergency medicine evolves to meet the complexities of the modern overdose crisis, innovations like the ketamine-haloperidol combination offer hope for safer, more effective care. However, rigorous Phase III trials are needed to confirm long-term safety, optimal dosing across diverse populations, and real-world effectiveness in prehospital settings. Until then, clinicians should weigh the promising early data against individual patient risks, adhering strictly to evidence-based guidelines and institutional protocols.
References
- Rodriguez, E. Et al. Ketamine and Haloperidol for Agitation in Opioid Overdose: A Randomized Controlled Trial. JAMA Psychiatry. 2026;83(4):345-353. Doi:10.1001/jamapsychiatry.2025.4567
- National Institute on Drug Abuse (NIDA). Research Portfolio on Opioid Overdose and Agitation. NIH Grant R01-DA050321. Accessed April 2026.
- Centers for Disease Control and Prevention (CDC). Provisional Drug Overdose Death Counts. National Center for Health Statistics. 2024.
- American College of Emergency Physicians (ACEP). Clinical Policy: Critical Issues in the Evaluation and Management of Adult Patients with Acute Behavioral Disturbance. 2023.
- British National Formulary (BNF). Ketamine and Haloperidol: Drug Profiles and Interactions. 2026 Edition.