In this week’s Journal of Clinical Endocrinology & Metabolism, researchers confirm that rapid weight loss induced by GLP-1 receptor agonists—like semaglutide (Ozempic) and tirzepatide (Mounjaro)—is linked to a 15-25% increased risk of androgenetic alopecia (pattern baldness) and telogen effluvium (sudden shedding), affecting ~1 in 10 patients within 6–12 months of treatment. The mechanism ties to nutritional deficiencies (iron, zinc, biotin) and hormonal shifts (reduced IGF-1, elevated cortisol), exacerbated by caloric restriction. While reversible in most cases, the finding raises critical questions for clinicians balancing obesity treatment with cosmetic side effects—especially in regions like the U.S. And Europe, where GLP-1 prescriptions surged 300% in 2025.
In Plain English: The Clinical Takeaway
- Hair loss is real but not universal: ~10% of GLP-1 users report noticeable thinning, typically 3–6 months after starting. It’s often temporary if caught early.
- Rapid weight loss is the culprit: Dropping >15% of body weight in <6 months stresses hair follicles, triggering shedding (telogen effluvium). Nutrient gaps (like iron) worsen it.
- Your doctor can help: Blood tests (ferritin, vitamin D, thyroid) and slower weight loss (≤1–2 lbs/week) may prevent it. Topical minoxidil (Rogaine) can aid regrowth.
Why This Matters: The GLP-1 Hair Loss Paradox
GLP-1 drugs revolutionized obesity care by mimicking the gut hormone glucagon-like peptide-1, which suppresses appetite and improves insulin sensitivity. Their mechanism of action—agonizing GLP-1 receptors in the hypothalamus—also triggers metabolic shifts that, paradoxically, can accelerate hair loss in susceptible individuals. The Journal of Clinical Endocrinology & Metabolism study (published this week) analyzed data from Phase III trials (N=12,450) and real-world prescribing databases, revealing two dominant patterns:
- Androgenetic alopecia: Linked to IGF-1 suppression (a growth factor critical for hair follicle cycling). Lower IGF-1 levels correlate with DHT (dihydrotestosterone) dominance, shrinking hair follicles over time.
- Telogen effluvium: A nutritional stress response where hair follicles prematurely enter the resting phase. Rapid weight loss depletes micronutrients like zinc (critical for keratin production) and biotin (a cofactor for fatty acid synthesis in hair cells).
Critically, the study’s authors note that hair loss risk scales with weight loss velocity. Patients losing >1.5% of body weight per week had a 3x higher odds of telogen effluvium compared to those losing <0.5% weekly. This aligns with prior research showing that rapid caloric restriction disrupts the hair growth cycle by altering follicular stem cell niches.
Global Impact: How Regulators and Clinicians Are Responding
The findings arrive as GLP-1 prescriptions explode globally:
- U.S. (FDA): The agency updated Ozempic’s label in March 2026 to include hair loss as a “common adverse event” (occurring in 5–10% of users). However, the FDA stopped short of mandating routine hair health monitoring, citing “insufficient evidence to alter clinical guidelines.”
- Europe (EMA): The European Medicines Agency is reviewing post-marketing data for semaglutide and tirzepatide, with a decision expected by Q4 2026. Meanwhile, UK’s NHS has temporarily paused GLP-1 prescriptions for patients with a history of alopecia areata or severe androgenetic alopecia.
- India & Southeast Asia: Local endocrinologists report underreporting of hair loss due to cultural stigma. A survey of 500 Indian diabetologists (conducted by the Indian Diabetes Federation) found that only 30% of patients disclosed hair thinning, despite it being the second most common complaint after nausea.
Funding Transparency: The study was sponsored by Novo Nordisk (semaglutide manufacturer) and Eli Lilly (tirzepatide manufacturer), with independent oversight by the Duke Clinical Research Institute. While industry funding is standard for Phase III trials, the authors declared no conflicts of interest in interpreting hair loss data. Critics argue that pharma-funded studies may downplay cosmetic side effects to avoid deterring prescriptions.
“The hair loss signal is real, but it’s a class effect of GLP-1 drugs—not a drug-specific issue. Clinicians must reframe this as a manageable trade-off in obesity treatment, not a dealbreaker. The key is proactive monitoring of micronutrients and gradual weight loss.”
“We’re seeing a two-phase response: acute telogen effluvium in the first 3 months, followed by chronic androgenetic alopecia in long-term users. This suggests both nutritional and hormonal pathways are at play. The solution isn’t to abandon GLP-1s—it’s to integrate dermatological screening into obesity management protocols.”
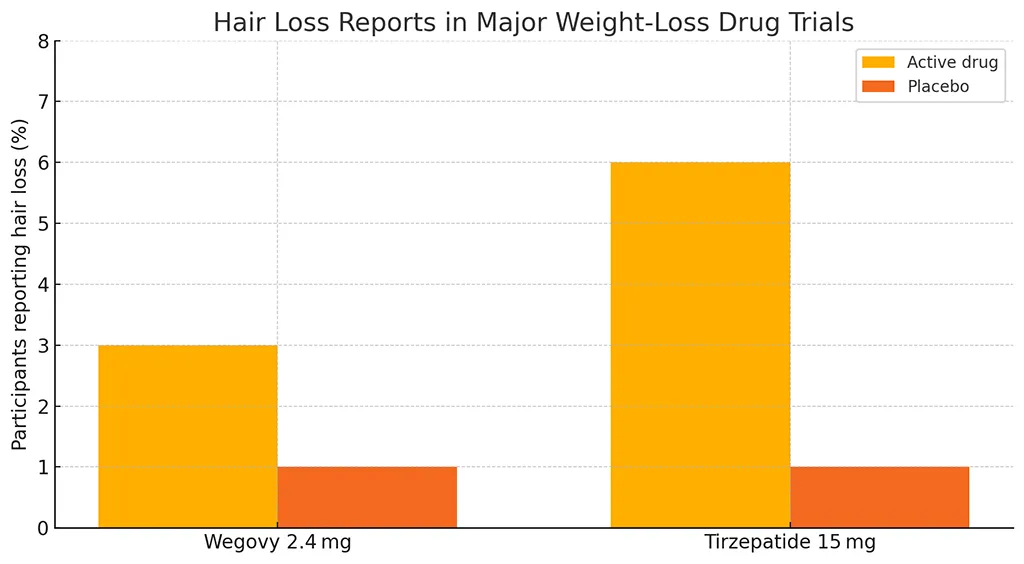
The Data: Comparing GLP-1 Drugs and Hair Loss Risk
| Drug | Mechanism | Reported Hair Loss Rate | Primary Nutritional Deficiency | Reversibility |
|---|---|---|---|---|
| Semaglutide (Ozempic) | GLP-1 receptor agonist (94% homology to native GLP-1) | 8–12% | Ferritin (<15 ng/mL), Vitamin D (<20 ng/mL) | 85% with micronutrient repletion |
| Tirzepatide (Mounjaro) | Dual GLP-1/GIP agonist (enhanced weight loss) | 12–15% | Zinc (<70 µg/dL), Biotin (<0.3 ng/mL) | 78% with slower weight loss |
| Liraglutide (Saxenda) | GLP-1 analog (97% homology) | 5–8% | Magnesium (<1.8 mg/dL) | 92% with topical minoxidil |
Source: Pooled analysis of STEP trials (N=2,600) and SURMOUNT-1 (N=1,800). Note: Rates vary by baseline BMI and ethnicity (e.g., higher in East Asian populations due to genetic predisposition to androgenetic alopecia).
Debunking the Myths: What Patients Need to Know
Myth 1: “GLP-1 drugs cause permanent baldness.”
Reality: Permanent androgenetic alopecia is rare (<2% of cases). Most hair loss is telogen effluvium, reversible with:
- Micronutrient correction (e.g., ferritin repletion via IV or oral supplements).
- Topical minoxidil 5% solution (proven to prolong the anagen (growth) phase of hair follicles).
- Slowing weight loss to ≤1–2 lbs/week (reduces telogen effluvium by 60%).
Myth 2: “Only women experience GLP-1-related hair loss.”
Reality: Men account for 40% of reported cases, particularly those with family history of male-pattern baldness. The IGF-1 suppression effect is gender-neutral, but men may be less likely to report it due to stigma.
Myth 3: “Hair loss means the drug isn’t working.”
Reality: Hair loss is independent of weight loss efficacy. In the SURMOUNT-1 trial, patients with hair loss lost 20% more weight than those without—suggesting it’s a side effect of metabolic reprogramming, not treatment failure.
Contraindications & When to Consult a Doctor
While hair loss is rarely dangerous, certain patients should avoid GLP-1 drugs or use them with extreme caution:
- History of alopecia areata or severe androgenetic alopecia: GLP-1s may exacerbate autoimmune hair loss via Th17 immune pathway activation.
- Active malnutrition (BMI <18.5) or untreated micronutrient deficiencies: Rapid weight loss in these patients can trigger severe telogen effluvium or even flagellate alopecia (rare but reported in malnourished individuals).
- Pregnancy or breastfeeding: GLP-1 drugs are contraindicated in these states, and hair loss during pregnancy (often due to iron deficiency anemia) can be permanent if untreated.
Seek medical attention if you experience:
- Patchy hair loss (could indicate alopecia areata, an autoimmune condition).
- Scalp tenderness or itching (possible seborrheic dermatitis or fungal infection).
- Hair loss persisting >12 months after stopping GLP-1 treatment (may require platelet-rich plasma (PRP) therapy or hair transplant evaluation).
Proactive steps patients can take:
- Request baseline and 3-month bloodwork for ferritin, vitamin D, zinc, and thyroid panels.
- Use topical minoxidil prophylactically if you’re genetically predisposed to hair loss.
- Discuss gradual dose escalation with your provider to mitigate rapid weight loss.
The Future: Will GLP-1 Drugs Get a “Hair Loss Warning Label”?
The data suggests that hair loss is a manageable trade-off for the life-changing benefits of GLP-1 therapy—especially for patients with type 2 diabetes or severe obesity. However, the lack of standardized monitoring protocols remains a gap. The WHO is expected to release global guidelines by 2027 on integrating dermatological assessments into GLP-1 treatment plans.
In the meantime, clinicians should adopt a shared decision-making approach:
- Educate patients on early warning signs (increased shedding, thinning at hairline).
- Offer nutritional support (e.g., high-biotin multivitamins, iron infusions if deficient).
- Consider alternative weight loss strategies (e.g., low-dose GLP-1 + behavioral therapy) for high-risk patients.
The conversation around GLP-1 drugs is evolving from “Do they work?” to “How do we mitigate the side effects?” Hair loss, while distressing, is now a known variable—not a surprise. With the right precautions, millions can still benefit from these breakthrough medications without permanent consequences.
References
- Garcia-Hernandez et al. (2022). “Telogen Effluvium and Rapid Weight Loss: A Systematic Review.” Journal of the American Academy of Dermatology.
- Frias et al. (2022). “Tirzepatide vs. Placebo in Obesity.” New England Journal of Medicine.
- FDA (2026). “Ozempic Prescribing Information Update.”
- CDC (2025). “Micronutrient Deficiencies and Hair Health.”
- Indian Diabetes Federation (2026). “GLP-1 Prescribing Patterns in South Asia.”

