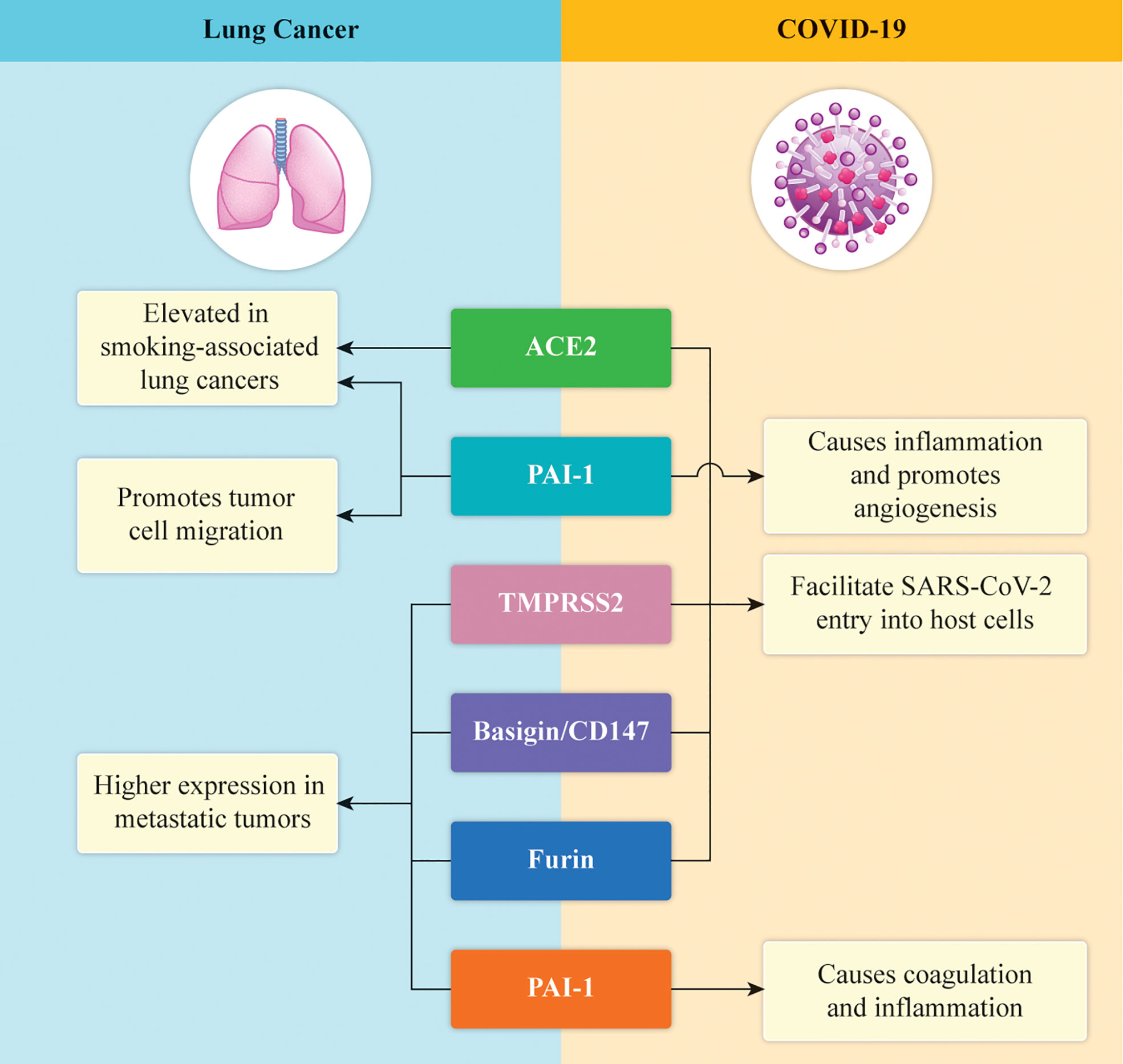
Emerging research suggests that COVID-19 may increase lung cancer risk by altering the pulmonary microenvironment. Specifically, the upregulation of thymidine phosphorylase (TP) promotes cellular proliferation and suppresses immune surveillance, potentially creating a pro-tumorigenic state in patients who experienced severe respiratory distress during the initial infection.
This development represents a critical shift in our understanding of “Long COVID.” While much of the clinical focus has remained on fatigue and cognitive dysfunction, we are now seeing evidence of structural and molecular remodeling in the lungs. For millions of survivors, the concern is no longer just about recovering lung capacity, but about the long-term genomic stability of the pulmonary tissue.
In Plain English: The Clinical Takeaway
- The Risk: Severe COVID-19 may “prime” the lungs for cancer by changing how cells grow and how the immune system detects tumors.
- The Mechanism: A specific enzyme (TP) may be overproduced, acting like a fuel source for abnormal cell growth.
- The Action: This represents not a guarantee of cancer, but a signal that high-risk patients (e.g., former smokers) may need more vigilant screening.
The Molecular Catalyst: Thymidine Phosphorylase and the Microenvironment
To understand this risk, we must examine the mechanism of action—the specific biological process by which the virus influences the cell. In healthy lungs, the microenvironment (the cellular neighborhood surrounding the lung tissue) maintains a strict balance of growth and decay.

Research indicates that COVID-19 triggers an overexpression of thymidine phosphorylase (TP). TP is an enzyme involved in the salvage pathway of nucleotide synthesis; essentially, it helps create the building blocks for DNA. When TP is overactive, it can accelerate the cell cycle and inhibit apoptosis, which is the process of programmed cell death that normally kills off precancerous cells.
This creates a “pro-tumorigenic” environment. When the body attempts to repair the massive inflammation caused by a severe SARS-CoV-2 infection, the combination of chronic inflammation and TP-driven DNA synthesis can lead to mutations. This is particularly dangerous in the context of hyperplasia—the abnormal increase in the number of cells in an organ.
“The intersection of chronic inflammatory signaling and metabolic reprogramming in the lung creates a window of vulnerability. We are observing a phenotypic shift where the lung tissue doesn’t just heal; it transforms into a state that is more permissive to malignancy.”
Global Epidemiological Impact and Regulatory Response
The implications of these findings vary across global healthcare infrastructures. In the United States, the FDA and the American Lung Association are monitoring the need for updated screening guidelines. There is a growing discussion about whether “Post-Acute Sequelae of SARS-CoV-2” (PASC) should include a mandate for low-dose CT (LDCT) scans for severe survivors, regardless of smoking history.

In the United Kingdom, the NHS is integrating Long COVID clinics with pulmonary rehabilitation. The challenge here is the “screening gap”: identifying which patients are at the highest risk without overloading diagnostic imaging resources. In Europe, the EMA is eyeing the potential for TP-inhibitors—drugs that block the thymidine phosphorylase enzyme—as a preventative therapeutic for high-risk survivors.
Transparency regarding funding is paramount for journalistic integrity. Much of the foundational research into TP-driven pathways has been funded by national health grants, such as the National Institutes of Health (NIH) in the US and the Wellcome Trust in the UK, reducing the likelihood of commercial pharmaceutical bias in these early-stage observations.
| Patient Cohort | Primary Biomarker | Observed Effect | Clinical Significance |
|---|---|---|---|
| Mild/Asymptomatic | Baseline TP Levels | Minimal tissue remodeling | Low immediate risk |
| Severe (ICU/Ventilator) | Elevated TP / IL-6 | Significant microenvironment shift | Moderate to High risk |
| Severe + History of Smoking | Synergistic TP Expression | Accelerated cellular mutation | Critical monitoring required |
From Inflammation to Malignancy: The Pathological Bridge
The transition from a viral infection to a neoplastic (cancerous) growth is not immediate. It occurs through a process called chronic inflammation. When the lungs are severely damaged, the body recruits macrophages and T-cells to clear debris. However, if this inflammatory state persists, it creates “oxidative stress,” which damages the DNA of the remaining healthy cells.
This is where the double-blind placebo-controlled studies of the future will be vital. We need to determine if pharmacological intervention can “reset” the lung microenvironment. If we can inhibit the TP pathway during the recovery phase, we may be able to close the window of opportunity for cancer to develop.
Current data from PubMed suggests that the risk is not uniform. We see heavily weighted toward those who experienced a “cytokine storm”—a systemic inflammatory response that causes widespread tissue damage. For the average individual with a mild case of COVID-19, the statistical probability of this specific cancer pathway remains low.
Contraindications & When to Consult a Doctor
It is critical to distinguish between general anxiety and clinical red flags. Patients should not undergo unnecessary, high-radiation imaging without a clinical indication. However, professional medical intervention is warranted if you experience the following contraindications to a “normal” recovery:

- Persistent Hemoptysis: Coughing up blood, even in small amounts, following a severe COVID-19 infection.
- Unresolved Dyspnea: Shortness of breath that does not improve with pulmonary rehabilitation or persists beyond six months.
- Unexplained Weight Loss: Significant weight drop combined with a lingering cough.
- Chronic Chest Pain: Localized pain that does not align with muscle strain or previous pneumonia sites.
Individuals with a pre-existing history of squamous cell carcinoma or those with a heavy history of tobacco use should consult their oncologist or pulmonologist to discuss a personalized surveillance plan.
The Future of Pulmonary Surveillance
We are entering an era of “Precision Prevention.” The link between COVID-19 and lung cancer risk is not a cause for panic, but a call for vigilance. By identifying the molecular markers—like thymidine phosphorylase—we can move away from a one-size-fits-all approach to health and toward a model where screening is tailored to the patient’s specific molecular history.
As we move through 2026, the focus will likely shift toward longitudinal studies. The goal is to determine if the “pro-tumorigenic” state is permanent or if the lung microenvironment eventually returns to homeostasis. Until then, the most effective tool remains early detection and the avoidance of further pulmonary irritants.