In April 2026, researchers reported promising early results from a Phase II clinical trial of a new Lyme disease vaccine candidate, VLA15, showing robust immune responses in healthy adults across endemic regions of the northeastern United States, though questions remain about long-term durability and real-world effectiveness in diverse populations.
Understanding the VLA15 Vaccine Candidate and Its Mechanism of Action
The VLA15 vaccine, developed by Valneva in collaboration with Pfizer, targets the outer surface protein A (OspA) of Borrelia burgdorferi, the spirochete bacterium transmitted by black-legged ticks (Ixodes scapularis) that causes Lyme disease. By inducing antibodies against OspA, the vaccine aims to neutralize the bacteria within the tick’s gut during feeding, preventing transmission to humans—a mechanism known as “transmission-blocking immunity.” This approach differs from traditional vaccines that act after pathogen entry into the host.
In Plain English: The Clinical Takeaway
- The VLA15 vaccine works by stopping Lyme disease bacteria in the tick before they can infect you, not by treating illness after a bite.
- Early trial data shows strong antibody production after three doses, but we still don’t know how long protection lasts or how well it works in children, older adults, or those with prior Lyme infection.
- If approved, this vaccine would be the first available for Lyme disease in the U.S. Since LYMErix was withdrawn in 2002, offering a new tool for prevention in high-risk areas.
Clinical Trial Progress and Epidemiological Context
As of April 2026, Valneva and Pfizer have completed Phase II trials (NCT04755133) involving over 1,800 participants aged 18–65 across Lyme-endemic regions in the U.S., Germany, and Sweden. The vaccine demonstrated a favorable safety profile, with most adverse events being mild to moderate injection-site reactions. Serological responses showed ≥90% seroconversion after three doses, meeting non-inferiority benchmarks compared to historical LYMErix data. However, no efficacy data against natural tick challenge has yet been published, as Phase III efficacy trials are ongoing.

Lyme disease remains the most common vector-borne illness in the Northern Hemisphere, with approximately 476,000 new cases diagnosed annually in the United States according to CDC surveillance data. The Northeast, Mid-Atlantic, and upper Midwest account for over 90% of cases, placing significant strain on regional healthcare systems. In New York State alone, where the University of Rochester Medical Center (URMC) is located, over 8,000 cases were reported in 2024, prompting renewed public health focus on prevention strategies.
Geo-Epidemiological Bridging: Impact on Regional Healthcare Systems
In the United States, the Food and Drug Administration (FDA) regulates vaccine approval through its Center for Biologics Evaluation and Research (CBER). Should VLA15 proceed to Phase III and demonstrate efficacy, it would likely seek accelerated approval under the FDA’s Animal Rule or traditional pathways, pending demonstration of clinical benefit in preventing erythema migrans or disseminated disease. In Europe, the European Medicines Agency (EMA) would oversee evaluation via the centralized procedure, with potential implications for national immunization programs in high-incidence countries like Germany, Austria, and Slovenia.
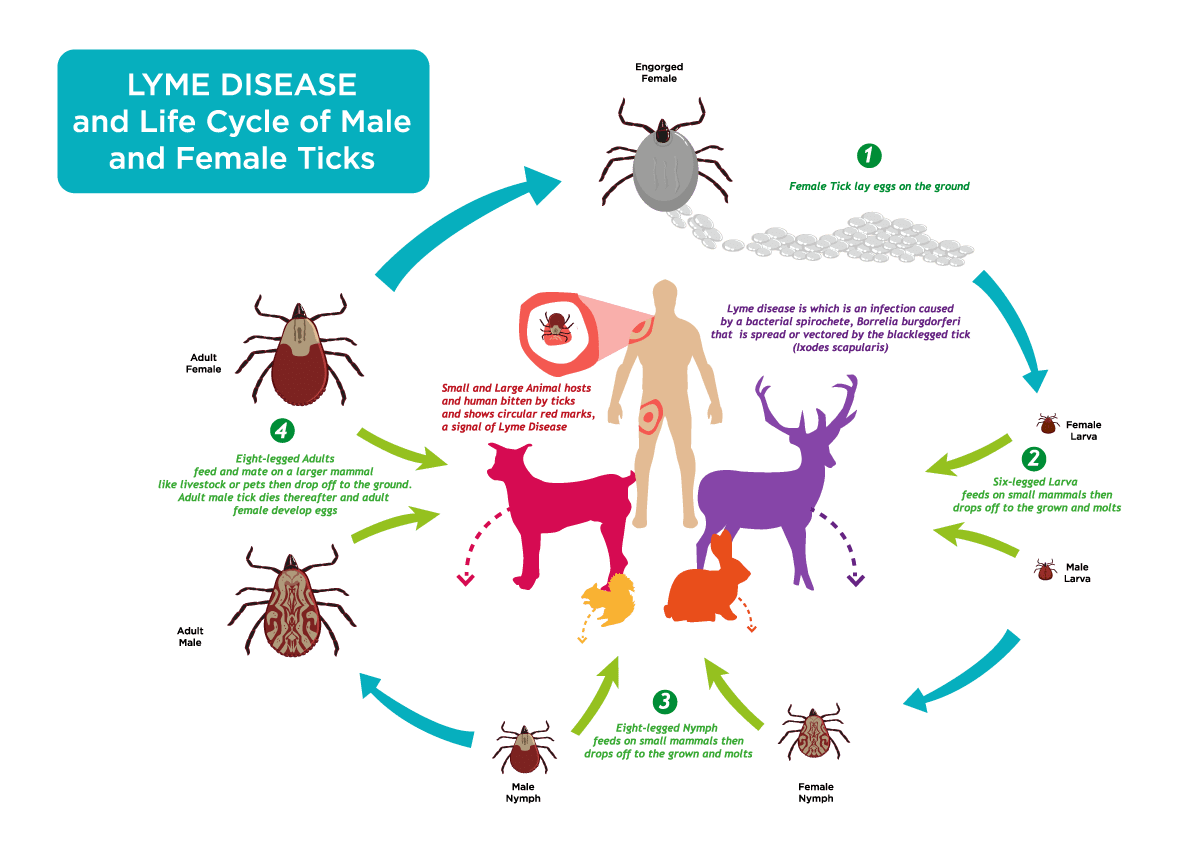
In the UK, where Lyme disease incidence is lower but rising—particularly in Scotland and southern England—the National Health Service (NHS) currently does not recommend routine vaccination due to limited endemic risk. However, a licensed vaccine could shift guidance for occupational groups (e.g., forestry workers) or travelers to endemic zones.
Funding, Bias Transparency, and Expert Perspectives
The VLA15 clinical program is primarily funded by Valneva and Pfizer, with additional support from the European Union’s Horizon 2020 research program (grant agreement No. 874735) for adjuvant optimization studies. While industry sponsorship is standard in vaccine development, independent oversight is maintained through Data Safety Monitoring Boards (DSMBs) and publication in peer-reviewed journals.

“The strength of the immune response seen in Phase II is encouraging, but we need Phase III data to confirm whether this translates to actual protection against infection in real-world settings—especially in pediatric populations who bear a disproportionate burden of Lyme disease.”
“Even with a highly effective vaccine, Lyme prevention will remain multi-faceted. Tick checks, prompt removal, and landscape management will continue to be essential, particularly as climate change expands tick habitats northward and westward.”
Comparative Overview: VLA15 vs. Historical and Alternative Approaches
| Characteristic | VLA15 (Valneva/Pfizer) | LYMErix (GSK, withdrawn 2002) | No Vaccine (Current Standard) |
|---|---|---|---|
| Target Antigen | OspA (six serotypes) | OspA (single serotype) | N/A |
| Dose Regimen | 3 doses (0, 1–2, 6 months) | 3 doses (0, 1, 12 months) | N/A |
| Primary Mechanism | Transmission-blocking (anti-tick gut) | Transmission-blocking (anti-tick gut) | N/A |
| Age Range Studied | 18–65 (Phase II) | 15–70 | All ages |
| Adjuvant | Aluminum hydroxide | Aluminum hydroxide | N/A |
| Current Status | Phase III ongoing (2026) | Withdrawn due to low demand and litigation concerns | Behavioral prevention only |
Contraindications & When to Consult a Doctor
Individuals with a known hypersensitivity to any component of the VLA15 vaccine, including aluminum hydroxide, should not receive it. Those with moderate to severe acute illness should delay vaccination until recovery. The vaccine is not currently recommended for individuals under 18 or over 65 pending Phase III data in these populations. Anyone experiencing symptoms such as expanding erythema migrans rash, facial palsy, arthritis, or cardiac arrhythmias following a tick bite should seek immediate medical evaluation, as early antibiotic treatment (e.g., doxycycline) remains highly effective for early Lyme disease.
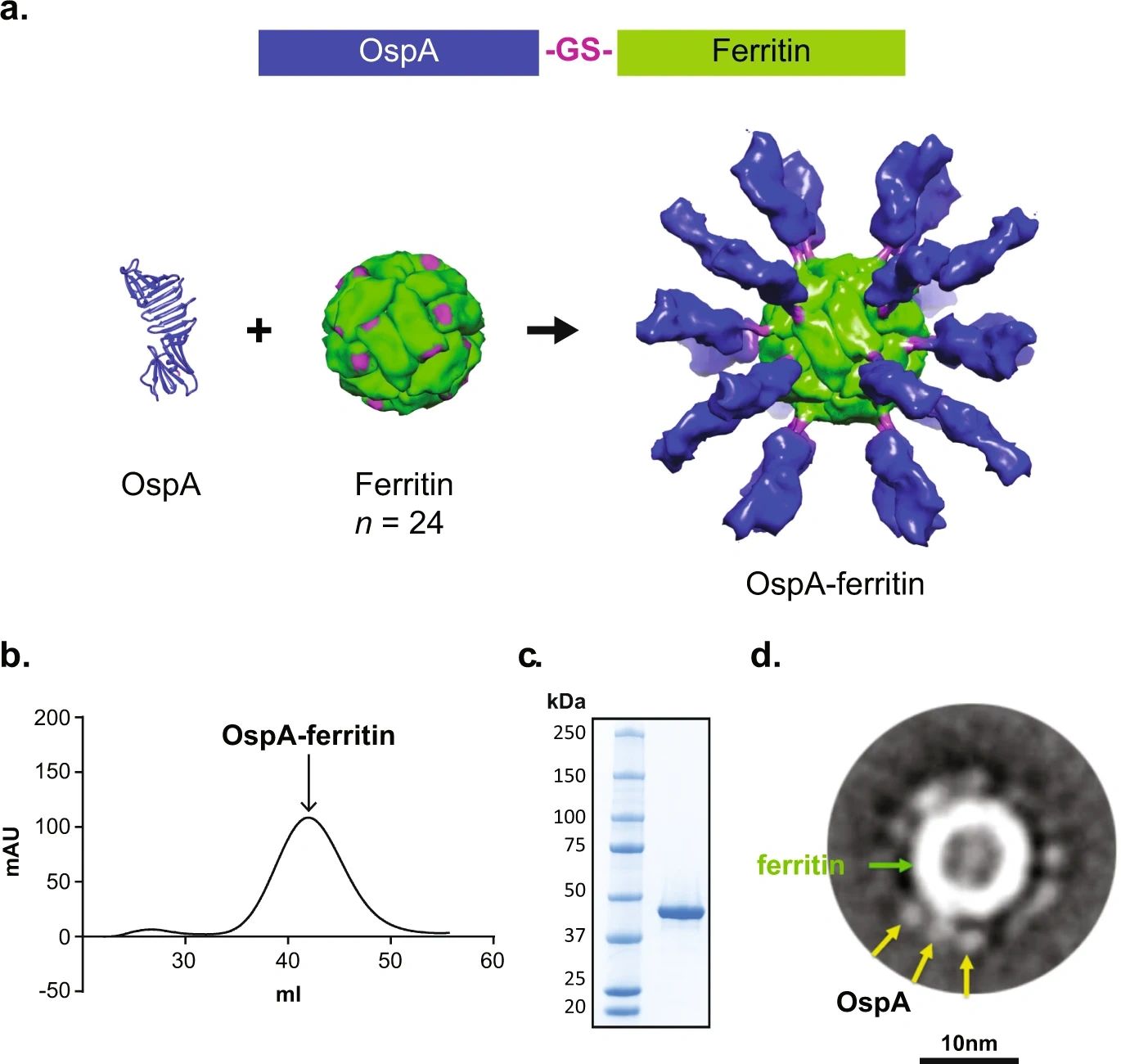
The Path Forward: Measured Optimism and Public Health Imperatives
While the VLA15 vaccine represents a significant advancement in Lyme disease prevention, its ultimate impact will depend on demonstrating durable efficacy in Phase III trials, equitable access across endemic regions, and public acceptance informed by transparent risk-benefit communication. Until then, the cornerstone of Lyme prevention remains personal protective measures: using EPA-registered insect repellents, performing daily tick checks, showering within two hours of outdoor exposure, and promptly removing attached ticks with fine-tipped tweezers.
References
- PubMed: Safety and immunogenicity of a Lyme disease vaccine candidate (VLA15) in healthy adults – Phase II results
- The Lancet: Global burden of Lyme borreliosis, 2020–2025
- JAMA: Trends in Lyme disease incidence, United States, 2010–2024
- CDC: Lyme disease data and surveillance
- European Medicines Agency: Vaccine evaluation pathways

