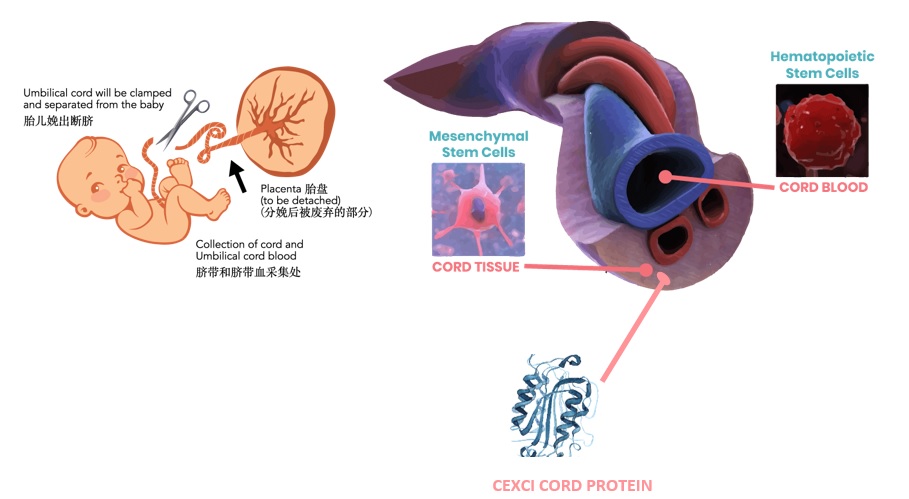
ThermoGenesis Holdings (US88603Y1055) specializes in the processing and banking of umbilical cord blood, leveraging mesenchymal stem cells (MSCs) for regenerative medicine. By concentrating these potent cells, the company aims to transition from a storage provider to a therapeutic leader in treating autoimmune and degenerative diseases globally.
The intersection of biotechnology and financial viability often hinges on a single factor: clinical utility. For patients and investors alike, the question isn’t just whether ThermoGenesis can store cells, but whether those cells can be reliably deployed as a “living drug” to reverse chronic pathology. As we move through April 2026, the shift toward cell-based therapies is no longer theoretical; it is a regulatory race between the FDA and EMA to standardize the “mechanism of action”—the specific biochemical process through which a drug produces its effect—for MSCs.
In Plain English: The Clinical Takeaway
- What it is: A move from simply “saving” stem cells in a freezer to using them to treat actual diseases.
- The Goal: Using cells from umbilical cords to reduce inflammation and repair damaged tissues in the body.
- The Bottom Line: While the technology is promising, it requires strict regulatory approval before it becomes a standard medical treatment.
The Biology of Regeneration: How Cord Blood MSCs Function
At the core of ThermoGenesis’s value proposition is the use of Mesenchymal Stem Cells (MSCs). Unlike hematopoietic stem cells, which create blood, MSCs are multipotent stromal cells capable of differentiating into bone, cartilage, and fat cells. More importantly, they possess a powerful immunomodulatory effect, meaning they can “calm down” an overactive immune system.
The clinical focus is currently on the paracrine effect. Instead of simply replacing dead cells, MSCs secrete cytokines and growth factors that signal the body’s own tissues to heal. Here’s critical for treating conditions like Graft-versus-Host Disease (GvHD), where the body’s immune system attacks its own organs after a transplant. By modulating the T-cell response, these therapies can prevent systemic organ failure.
However, the transition from “banking” to “therapy” requires a shift in processing. The company must ensure the purity and potency of the cells. Any contamination during the separation process can lead to adverse immune reactions or, in worst-case scenarios, the promotion of tumorigenesis—the uncontrolled growth of cells into tumors.
Regulatory Hurdles and the Global Access Gap
The path to commercialization is gated by the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). In the United States, cell therapies are regulated as “Biologics,” which requires rigorous Phase I, II, and III clinical trials to prove safety and efficacy. Many companies in this space struggle because they rely on “expanded” cells (growing cells in a lab), which the FDA scrutinizes heavily for genetic stability.
In Europe and Asia, the regulatory environment for stem cell clinics is often more fragmented, leading to a rise in “unproven stem cell therapies.” This creates a dangerous information gap where patients pay out-of-pocket for treatments that lack double-blind placebo-controlled trials—the gold standard of research where neither the patient nor the doctor knows who received the treatment, eliminating bias.
“The challenge for the next decade of regenerative medicine is not the scarcity of cells, but the standardization of the delivery. We must move from ‘artisan’ cell processing to industrial-grade pharmaceutical precision to ensure patient safety.” — Dr. Aris Thikiros, Regenerative Medicine Specialist.
Funding for these advancements is typically a mix of venture capital and public grants. It is essential to note that much of the early-stage research into cord blood processing is funded by the companies selling the banking services, creating a potential conflict of interest. Independent verification via peer-reviewed journals is the only way to decouple corporate optimism from clinical reality.
Comparative Efficacy of Cell Therapy Modalities
To understand if the cell therapy focus is “strong enough,” we must compare umbilical cord-derived MSCs with other common sources, such as bone marrow or adipose (fat) tissue.
| Cell Source | Immunogenicity (Risk of Rejection) | Proliferation Rate | Primary Clinical Application |
|---|---|---|---|
| Umbilical Cord Blood | Low (Allogeneic) | High | Autoimmune / Pediatric Disorders |
| Bone Marrow | Moderate (Autologous) | Medium | Hematologic Malignancies |
| Adipose Tissue | Low (Autologous) | Medium | Orthopedic / Plastic Surgery |
Bridging the Gap: From Storage to Systemic Treatment
For ThermoGenesis to dominate, it must solve the “delivery” problem. Injecting cells into the bloodstream often results in the “pulmonary trap,” where the majority of cells are filtered out by the lungs before they ever reach the target organ. To overcome this, researchers are exploring scaffold-based delivery and targeted chemotaxis—using chemical signals to “lure” the stem cells to the site of injury.
The epidemiological need is vast. With an aging global population and a rise in chronic inflammatory diseases, the demand for non-pharmacological interventions is peaking. If the company can successfully navigate the PubMed-documented hurdles of cell viability and dosage standardization, they move from a service-based revenue model to a high-margin therapeutic model.
Contraindications & When to Consult a Doctor
Cell therapy is not a universal cure and carries significant risks for certain populations. Patients with a history of active malignancy (cancer) should avoid MSC therapies, as these cells can potentially secrete factors that encourage the growth of existing tumors.

individuals with severe acute infections or those experiencing an anaphylactic reaction to bovine serum (often used in cell culture) are contraindicated. You must consult a board-certified hematologist or immunologist if you are considering stem cell treatments, especially if you have a compromised immune system or are currently undergoing chemotherapy.
Immediate medical intervention is required if, following a cell infusion, you experience shortness of breath, high fever, or sudden swelling in the extremities, which may indicate a pulmonary embolism or an acute inflammatory response.
The Clinical Verdict
The focus on cell therapy is a necessary evolution for any cord blood entity. However, the “strength” of this focus is measured by clinical data, not corporate press releases. The transition from a bank to a biotech firm requires an immense capital expenditure in GMP (Good Manufacturing Practice) facilities and a willingness to face the grueling scrutiny of Phase III trials.
For the patient, the promise is profound: a future where we treat the root cause of degeneration rather than managing symptoms. For the observer, the trajectory of ThermoGenesis will depend on whether they can produce a statistically significant improvement in patient outcomes that justifies the high cost of biological manufacturing.

