Families in North Macedonia are facing a public health crisis after Euromedics, a stem-cell storage provider, disappeared without accounting for umbilical cord blood samples collected since 2012. This loss deprives patients of potentially life-saving hematopoietic stem cells used to treat severe blood disorders, leukemias, and certain genetic immunodeficiencies.
This systemic failure is more than a corporate disappearance; It’s a stark warning regarding the lack of stringent regulatory oversight in private biobanking. When biological assets—literally the genetic blueprints of a child’s future health—are treated as commercial commodities without state-mandated auditing, the result is a catastrophic loss of medical agency. For the affected families, the “cold trail” isn’t just a legal frustration; it is the erasure of a biological insurance policy.
In Plain English: The Clinical Takeaway
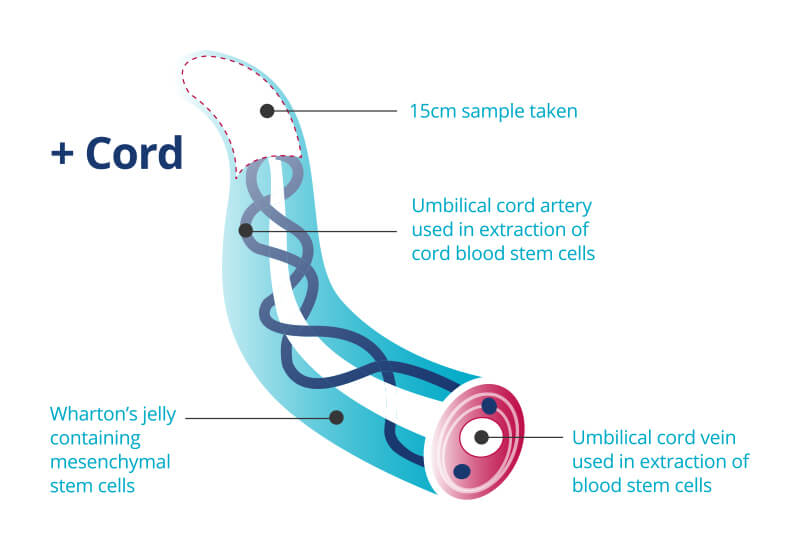
- What was lost: Cord blood contains hematopoietic stem cells (HSCs), which are “master cells” capable of regenerating the entire blood and immune system.
- The Risk: Private banks often lack the same rigorous oversight as public hospitals, meaning samples can be lost, degraded, or mismanaged without the family knowing.
- The Lesson: Biological storage should ideally be handled by accredited institutions that follow international standards (like AABB or FACT) to ensure the cells remain viable for decades.
The Biological Stakes: What is Lost in the Cold Trail?
To understand the gravity of the Euromedics collapse, one must understand the mechanism of action of umbilical cord blood. Cord blood is rich in hematopoietic stem cells (HSCs)—undifferentiated cells that have the unique ability to develop into red blood cells, white blood cells, and platelets. In clinical settings, these cells are utilized in “stem cell transplants” to replace diseased or damaged bone marrow.
The process requires precise cryopreservation, the act of freezing biological samples at ultra-low temperatures (typically -196°C in liquid nitrogen) to stop all metabolic activity. If the “cold chain” is broken—meaning the temperature fluctuates even slightly—the cells undergo lysis, where the cell membrane ruptures and the biological material becomes useless. For the families in North Macedonia, the uncertainty is twofold: they do not know if their samples exist, and if they do, they do not know if the cryopreservation was maintained.
“The unregulated proliferation of private cord blood banks creates a dangerous illusion of security. Without standardized auditing and mandatory reporting to national health registries, these biological assets are essentially invisible to the medical community until the moment they are needed, at which point the loss is irreversible.” — Dr. Elena Rossi, Hematology Consultant and advocate for standardized biobanking protocols.
The clinical utility of these cells is most profound in treating pediatric leukemias and sickle cell anemia. While bone marrow is a more common source of HSCs, cord blood is often preferred because it has a lower risk of Graft-versus-Host Disease (GvHD), a condition where the transplanted cells attack the recipient’s organs.
Regulatory Voids and the European Medicines Agency (EMA) Gap
The Euromedics situation highlights a critical geo-epidemiological divide in healthcare regulation. In the European Union, the European Medicines Agency (EMA) classifies stem cells as Advanced Therapy Medicinal Products (ATMPs). This classification mandates rigorous traceability, meaning every sample must be trackable from the point of collection to the point of administration.
North Macedonia, while aligning its laws with the EU acquis (the body of common rights and obligations), has historically lacked the enforcement mechanisms to hold private medical entities to these standards. This creates a “regulatory vacuum” where companies can operate with the appearance of medical legitimacy without the requisite oversight from a national health authority or the EMA. In contrast, systems like the NHS in the UK or the FDA-regulated labs in the US require strict adherence to Current Good Manufacturing Practice (cGMP) to ensure product purity and potency.
The following table summarizes the fundamental differences between the public banking model (which promotes transparency) and the private model (which led to the Euromedics crisis):
| Feature | Public Cord Blood Banks | Private Cord Blood Banks |
|---|---|---|
| Primary Goal | Altruistic donation for any compatible patient | Family-specific storage for the donor child |
| Funding Source | Government/Non-profit grants | Direct payment from parents |
| Regulatory Oversight | High (Strict government/WHO standards) | Variable (Often depends on local law) |
| Availability | Available to the general public | Restricted to the paying family |
| Risk Profile | Low risk of “disappearance” | Higher risk of corporate insolvency |
The Ethics of Private Banking and Corporate Accountability
The funding of private stem cell banks is almost entirely consumer-driven, creating a conflict of interest where marketing often outpaces clinical evidence. Many parents are sold the idea of “biological insurance,” yet the statistical probability of a child ever needing their own cord blood is remarkably low—estimated by some peer-reviewed studies to be less than 25% for the child or their siblings.

When a company like Euromedics ceases operations without a handover of biological materials, it is not merely a financial loss; it is a violation of bioethical autonomy. The lack of a centralized registry in North Macedonia meant that there was no “fail-safe” to alert the government when the company stopped meeting its storage obligations. This underscores the need for mandatory state-level biological registries to prevent the “orphaning” of genetic material.
Contraindications & When to Consult a Doctor
While stem cell therapy is revolutionary, it is not a universal cure. Patients and families should be aware of the following:
- Contraindications: Cord blood transplants are contraindicated in cases where the sample is contaminated with bacteria or viruses (e.g., CMV positive samples in certain neonates) or when the total nucleated cell (TNC) count is too low to achieve engraftment.
- Warning Signs: If you have stored biological materials in a private facility and have not received a verified “viability report” or “audit certificate” within the last 24 months, contact a healthcare provider to discuss the status of your samples.
- Professional Guidance: Always consult a board-certified hematologist or oncologist before making decisions about stem cell storage or transplantation to understand the actual statistical benefit based on your family’s medical history.
The tragedy in North Macedonia serves as a global case study in the necessity of treating biobanks not as warehouses, but as critical health infrastructure. Until international standards are legally binding across all borders, the risk of the “cold trail” remains a reality for thousands of families.
References
- PubMed: Hematopoietic Stem Cell Transplantation and Cord Blood Efficacy
- World Health Organization (WHO): Guidelines on Human Cell, Tissue and Organ Transplantation
- European Medicines Agency (EMA): Regulation of Advanced Therapy Medicinal Products (ATMPs)
- The Lancet: Longitudinal Studies on Cord Blood Banking and Pediatric Outcomes

