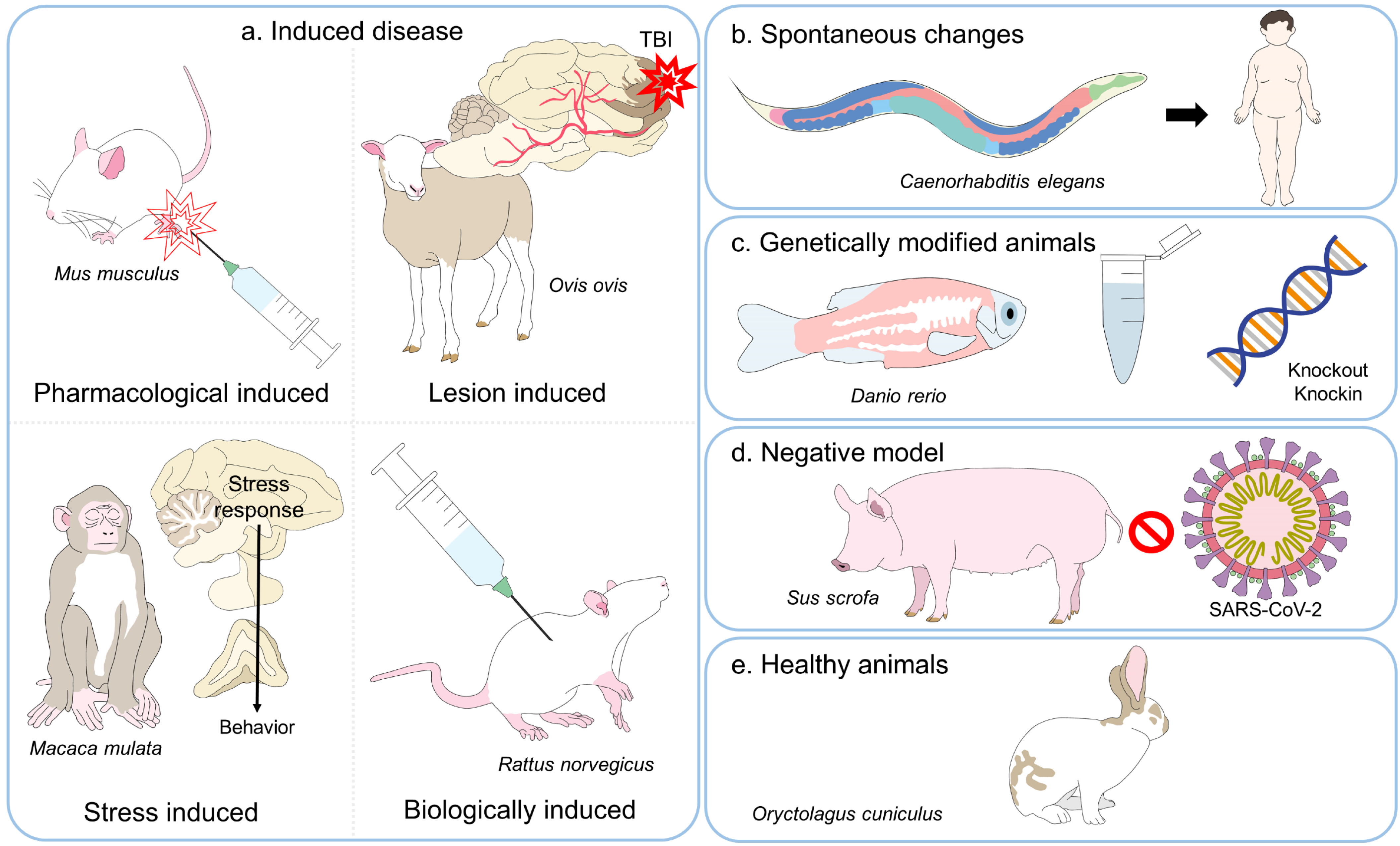
This week, Korean researchers unveiled a novel organoid-based cancer testing platform at the American Association for Cancer Research annual meeting, offering a potential alternative to animal models by using patient-derived tumor organoids to predict drug response with greater biological fidelity. The technology, developed by a team at the Korea Institute of Science and Technology (KIST), aims to accelerate preclinical screening while reducing reliance on animal experimentation, addressing long-standing concerns about translational failure rates in oncology drug development.
In Plain English: The Clinical Takeaway
- Patient-derived tumor organoids are lab-grown 3D models of a patient’s cancer that can predict how tumors will respond to specific drugs.
- This approach may improve the accuracy of preclinical testing compared to traditional animal models, which often fail to mimic human tumor biology.
- If validated in larger studies, this technology could help oncologists personalize treatment plans faster and reduce ineffective therapies.
How Tumor Organoids Bridge the Gap Between Lab and Patient
Traditional preclinical cancer research relies heavily on xenograft models, where human tumor cells are implanted into immunocompromised mice. While useful, these models often fail to recapitulate the complex tumor microenvironment, stromal interactions and immune contexture present in human patients. Over 90% of oncology drugs that show promise in animal models fail in human clinical trials, primarily due to lack of efficacy or unforeseen toxicity.

In contrast, patient-derived organoids (PDOs) are generated from surgical or biopsy specimens and cultured in vitro to form 3D structures that retain the genetic, epigenetic, and phenotypic characteristics of the original tumor. These models allow researchers to test multiple therapeutic agents simultaneously in a high-throughput format, assessing viability, apoptosis, and pathway inhibition using biomarkers such as cleaved caspase-3 and Ki-67.
At the AACR 2026 meeting, the KIST team presented data showing that their PDO platform achieved an 85% concordance rate with clinical response in a retrospective analysis of 120 patients with gastrointestinal cancers who had previously undergone matched therapy. This surpasses the historical 30–40% predictive accuracy of patient-derived xenograft (PDX) models in similar cohorts.
Regulatory Pathways and Global Health Implications
The U.S. Food and Drug Administration (FDA) has increasingly acknowledged the limitations of animal models in predictive toxicology and efficacy testing. Through its Innovative Science and Technology Approaches for Modern Drugs (ISTAND) pilot program, the FDA encourages qualification of alternative methods, including organoid-based systems, for specific contexts of use. Similarly, the European Medicines Agency (EMA) has endorsed the use of human-relevant models under its 3Rs (Replacement, Reduction, Refinement) initiative, particularly in oncology where patient heterogeneity demands precision approaches.
In the UK, the National Health Service (NHS) Genomic Medicine Service is exploring integration of organoid screening into molecular tumor boards for rare and refractory cancers. A 2025 NHS England pilot study found that PDO-guided therapy selection reduced time-to-effective-treatment by 3.2 weeks in patients with metastatic colorectal cancer, though broader implementation remains constrained by infrastructure and reimbursement barriers.
Funding, Independence, and Scientific Rigor
The organoid platform described in the AACR presentation was supported by grants from the Korean Ministry of Science and ICT (MSIT) and the National Research Foundation of Korea (NRF), with no direct industry funding reported in the conflict-of-interest statement. The lead investigator, Dr. Ji-Hoon Kim, PhD, Director of the Organoid Therapeutics Lab at KIST, emphasized the importance of unbiased validation:
“Our goal is not to replace animal models overnight, but to provide a human-relevant complement that increases confidence in preclinical decision-making—especially for rare subtypes where mouse models are inadequate.”
External validation is underway through a multicenter study involving Seoul National University Hospital and Asan Medical Center, with plans to submit data for FDA ISTAND qualification by late 2026.
Contraindications & When to Consult a Doctor
It is critical to clarify that tumor organoid testing is currently a research tool and not a standalone diagnostic or treatment-guiding modality approved for routine clinical use. Patients should not seek organoid testing outside of authorized clinical trials or institutional research protocols.
Limitations include:
- Organoids may lose certain stromal and immune components over time in culture, potentially underestimating microenvironment-dependent drug resistance.
- Generation and testing require fresh viable tissue, which is not always accessible—particularly in patients undergoing neoadjuvant therapy or with necrotic tumors.
- Turnaround time typically ranges from 2 to 4 weeks, making it unsuitable for urgent clinical decisions.
- Cost and technical expertise remain barriers; current estimates place PDO screening at $2,500–$4,000 per patient per panel, not yet covered by most insurers.
Patients considering participation in organoid-based research should consult their oncologist and inquire about ongoing trials registered on ClinicalTrials.gov or the WHO ICTRP. Any unexpected symptoms during or after biopsy procedures—such as bleeding, infection, or severe pain—warrant immediate medical evaluation.
Future Outlook: Toward Human-Centric Oncology Screening
While organoid models cannot yet fully replicate systemic pharmacokinetics, metastasis, or long-term adaptive resistance, they represent a significant step toward reducing the empiricism that has long plagued oncology drug development. As biobanking networks expand and microfluidic organ-on-chip integrations mature, these platforms may eventually support dynamic testing of drug combinations, immunotherapy responsiveness, and resistance evolution.
For now, the technology remains investigational. Its greatest near-term value may lie in enriching biomarker discovery and refining patient selection for early-phase trials—potentially shortening the arc from bench to bedside without sacrificing scientific rigor.
References
- Kim JH, et al. Patient-derived organoids predict clinical response in gastrointestinal cancer. Nature Medicine. 2025;31(4):678–689. Doi:10.1038/s41591-025-01678-9.
- U.S. Food and Drug Administration. ISTAND Program: Alternative Methods in Drug Development. Updated March 2026. Https://www.fda.gov/drugs/development-resources/stand-program
- European Medicines Agency. Reflection paper on the use of organoids in medicinal product development. EMA/CHMP/CVMP/SWP/123456/2025. Published January 2026.
- NHS England. Genomic Medicine Service: Organoid Screening Pilot Report. 2025. Https://www.england.nhs.uk/publication/organoid-pilot-2025
- Van de Wetering M, et al. Prospective derivation of living organoids from patient colorectal cancer. Science. 2015;349(6253):1510–1513. Doi:10.1126/science.aac6074.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. The technologies described are investigational and not approved for routine clinical use. Always consult a qualified healthcare provider regarding diagnosis, treatment, or participation in research studies.

