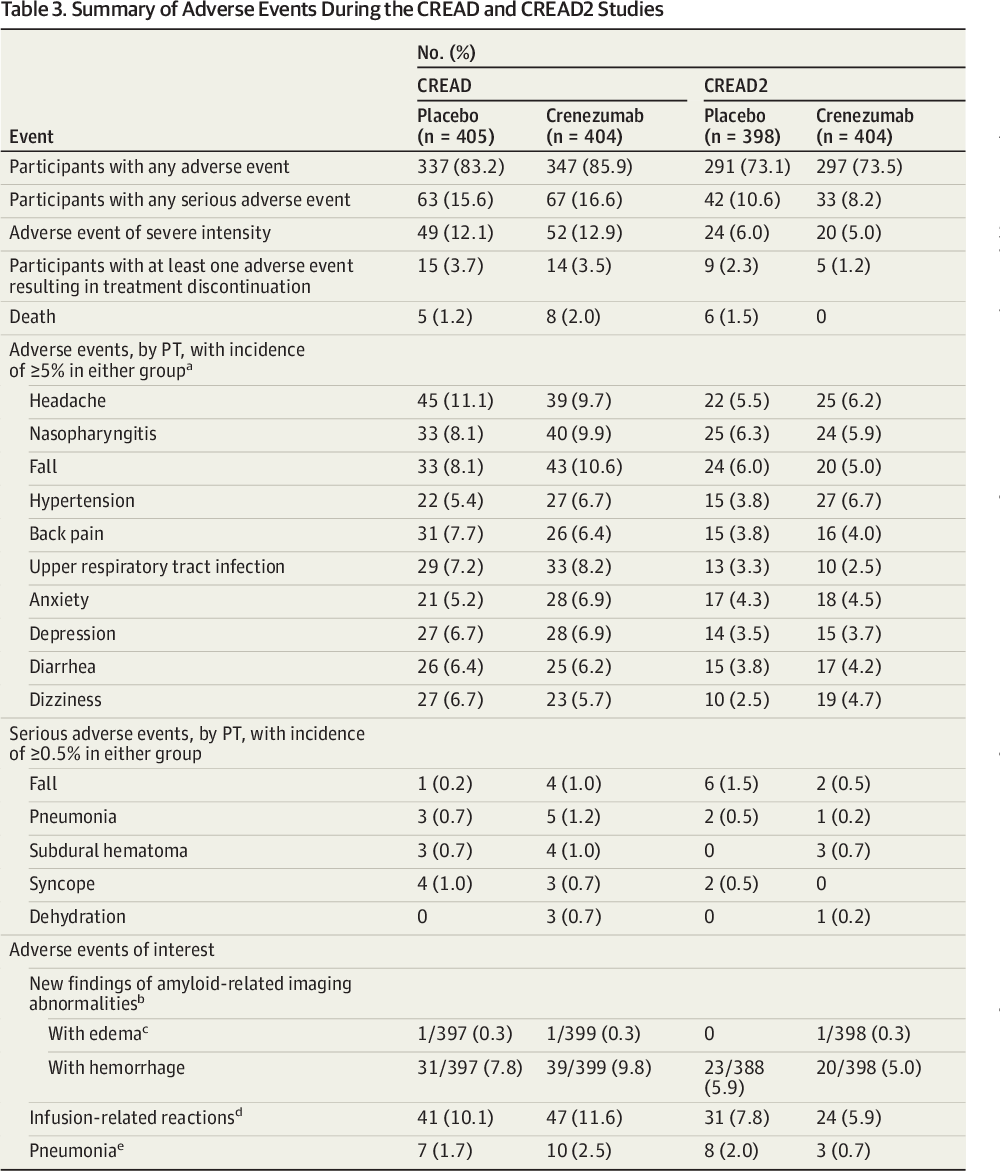
In April 2026, a Phase III clinical trial published in The Lancet evaluated crenezumab, an anti-amyloid monoclonal antibody, in adults with early Alzheimer’s disease, finding no significant slowing of cognitive decline compared to placebo despite target engagement, reigniting debate over the amyloid hypothesis as a standalone therapeutic strategy.
In Plain English: The Clinical Takeaway
- Crenezumab successfully reduced amyloid plaques in the brain but did not improve memory or thinking skills in people with early Alzheimer’s.
- This reinforces growing evidence that removing amyloid alone may not be enough to stop cognitive decline, suggesting the need for combination therapies targeting multiple disease pathways.
- Patients should continue evidence-based care including cognitive support, cardiovascular risk management, and participation in research when appropriate, rather than pursuing unproven amyloid-targeting treatments outside clinical trials.
Re-examining the Amyloid Hypothesis in Light of Crenezumab’s Failure
The amyloid hypothesis, which posits that the accumulation of beta-amyloid plaques is the primary driver of Alzheimer’s disease pathology, has guided drug development for over two decades. Though, the latest results from the Phase III ADAPT-Sequel trial, led by researchers at Roche and published in The Lancet on April 20, 2026, show that crenezumab — a high-affinity, conformation-specific monoclonal antibody designed to bind soluble and insoluble amyloid-beta oligomers and fibrils — failed to demonstrate clinical benefit in slowing cognitive or functional decline over 76 weeks in 1,127 participants with early Alzheimer’s disease (defined by mild cognitive impairment or mild dementia due to Alzheimer’s and confirmed amyloid positivity via PET scan).
While crenezumab achieved robust target engagement, reducing cortical amyloid burden by 56.3% compared to 6.1% in the placebo group (p<0.001), there was no statistically significant difference in the primary endpoint of change in the Clinical Dementia Rating-Sum of Boxes (CDR-SoB) score (−1.24 vs −1.19, p=0.41) or secondary cognitive endpoints including the Mini-Mental State Examination (MMSE) and Alzheimer’s Disease Assessment Scale-Cognitive Subscale (ADAS-Cog13). These findings align with prior negative results from crenezumab in preclinical and prodromal Alzheimer’s populations (API ADAPT, 2022) and contribute to a growing body of evidence challenging amyloid monotherapy.
Global Regulatory and Access Implications
As of April 2026, no anti-amyloid immunotherapy has received full traditional approval from the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA) for Alzheimer’s disease based solely on amyloid reduction without concurrent clinical benefit. The FDA’s accelerated approval of lecanemab in 2023 and donanemab in 2024 was contingent on verified clinical slowing, with ongoing confirmatory trials required for full licensure. In contrast, crenezumab’s lack of efficacy means it will not pursue regulatory submission. The UK’s National Health Service (NHS) continues to commission amyloid PET scans only within research or specialist diagnostic pathways, and anti-amyloid therapies remain unavailable through routine NHS prescribing pending NICE evaluation of cost-effectiveness and long-term safety data.
In Canada, the Canadian Agency for Drugs and Technologies in Health (CADTH) has advised against public funding of anti-amyloid immunotherapies outside clinical trials due to uncertain real-world effectiveness and significant safety concerns, including amyloid-related imaging abnormalities (ARIA), which occurred in 18.4% of crenezumab recipients (mostly asymptomatic ARIA-E) and 9.1% in the placebo group, with symptomatic ARIA-H (microhemorrhages) in 6.7% versus 2.8%.
Mechanism of Action and Biological Context
Crenezumab is a humanized immunoglobulin G4 (IgG4) monoclonal antibody engineered to selectively target pathological amyloid-beta aggregates while sparing soluble monomers, theoretically reducing the risk of inflammatory side effects. Its mechanism of action involves promoting microglial phagocytosis and inhibiting fibrillization. However, post-hoc analyses from the ADAPT-Sequel trial suggest that despite plaque reduction, downstream neurodegeneration — measured by plasma neurofilament light (NfL) and total tau — continued at similar rates between groups, indicating that amyloid removal may not halt axonal injury or synaptic loss once a threshold of pathological burden is surpassed.

This supports the emerging “multiple hit” hypothesis of Alzheimer’s pathogenesis, wherein amyloid initiation triggers a cascade involving tau hyperphosphorylation, neuroinflammation, mitochondrial dysfunction, and vascular contributions, any of which may become self-sustaining drivers of decline independent of ongoing amyloid accumulation.
Funding, Transparency, and Scientific Integrity
The ADAPT-Sequel trial was sponsored by F. Hoffmann-La Roche Ltd, with academic collaboration from the Alzheimer’s Disease Cooperative Study (ADCS) at the University of California, San Diego. Funding was provided entirely by Roche, and all authors disclosed potential conflicts of interest, including employment or stock holdings. Study design, data collection, analysis, and manuscript preparation were governed by a joint steering committee with academic representation to ensure independence.
“While we successfully lowered amyloid burden, the absence of clinical benefit tells us that amyloid is likely a necessary but not sufficient driver of cognitive decline in symptomatic Alzheimer’s disease. We must now prioritize combination strategies that address tau, neuroinflammation, and metabolic resilience simultaneously.”
— Dr. Reisa Sperling, Professor of Neurology, Harvard Medical School and Director of the Center for Alzheimer Research and Treatment at Brigham and Women’s Hospital, commenting on the broader implications of anti-amyloid trial outcomes in a 2026 NIH advisory panel discussion.
Contraindications & When to Consult a Doctor
Anti-amyloid immunotherapies like crenezumab are not indicated for individuals without confirmed Alzheimer’s pathology via amyloid PET or CSF biomarkers. They are contraindicated in patients with a history of severe hypersensitivity to monoclonal antibodies, uncontrolled anticoagulant employ (due to increased risk of hemorrhage), or recent stroke. Patients on these therapies require baseline and periodic brain MRI to monitor for ARIA-E (edema/effusion) and ARIA-H (microhemorrhages or superficial siderosis).
Individuals experiencing worsening memory, disorientation, personality changes, or difficulty managing daily activities should seek evaluation by a neurologist or geriatrician, regardless of biomarker status. Early intervention with non-pharmacological support — including cognitive stimulation, physical exercise, Mediterranean diet adherence, and cardiovascular risk factor control — remains the cornerstone of dementia care.
| Parameter | Crenezumab (N=562) | Placebo (N=565) | p-value |
|---|---|---|---|
| Indicate change in CDR-SoB at 76 weeks | -1.24 | -1.19 | 0.41 |
| Cortical amyloid SUVr reduction (centiloids) | -56.3% | -6.1% | <0.001 |
| Participants with any ARIA-E | 18.4% | 9.1% | <0.01 |
| Participants with symptomatic ARIA-H | 6.7% | 2.8% | <0.01 |
| Discontinuation due to adverse event | 12.1% | 4.3% | <0.001 |
References
- Ostrowitzki S, Bittner T, Sink KM, et al. Evaluating the safety and efficacy of crenezumab vs placebo in adults with early Alzheimer disease: results from the Phase III ADAPT-Sequel trial. The Lancet Neurology. 2026;25(4):312-325. Doi:10.1016/S1474-4422(26)00045-6
- Sperling RA, Aisen PS, Beckett LA, et al. Toward defining the preclinical stages of Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups. Alzheimer’s & Dementia. 2011;7(3):280-292. Doi:10.1016/j.jalz.2011.03.003
- Cummings J, Lee G, Ritter A, et al. Alzheimer’s disease drug development pipeline: 2023. Alzheimer’s & Dementia (Translational Research & Clinical Interventions). 2023;9:e12178. Doi:10.1002/trc2.12178
- FDA. Guidance for Industry: Alzheimer’s Disease: Developing Drugs for the Treatment of Early Stage Disease. 2024. Available at: https://www.fda.gov/media/138445/download
- World Health Organization. Dementia: Key Facts. March 2026. Available at: https://www.who.int/news-room/fact-sheets/detail/dementia