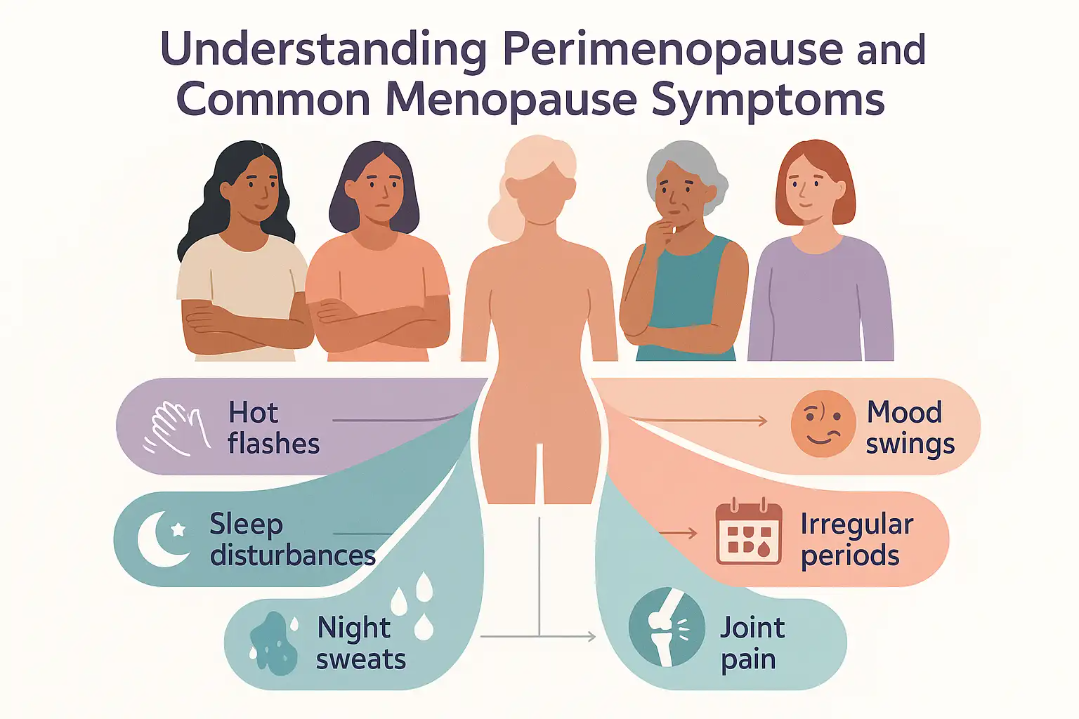
Perimenopause, the transitional phase before menopause, affects 85% of women, marked by hormonal fluctuations causing symptoms like hot flashes, fatigue, and brain fog. Understanding its clinical nuances and regional healthcare implications is critical for informed decision-making.
How Perimenopause Alters Hormonal Dynamics
Perimenopause, spanning 4–8 years before menopause, is driven by declining ovarian reserve, leading to erratic estrogen levels. This hormonal instability disrupts the hypothalamic-pituitary-ovarian axis, triggering vasomotor symptoms (hot flashes, night sweats) in 75% of women. A 2023 PubMed study found that 25% of women experience no vasomotor symptoms, highlighting individual variability.

Estrogen’s role in regulating body temperature, mood, and vaginal health means its decline impacts multiple systems. For instance, reduced estrogen thins vaginal epithelium, causing dryness and increased infection risk. A 2025 JAMA study linked perimenopausal estrogen fluctuations to a 30% increased risk of urinary tract infections.
In Plain English: The Clinical Takeaway
- Hot flashes are caused by unstable estrogen, not a “menopause disease.”
- Vaginal dryness can be managed with localized estrogen therapy, not just lifestyle changes.
- Brain fog may stem from sleep disruption, not cognitive decline.
Regional Healthcare Implications and Treatment Access
The U.S. Food and Drug Administration (FDA) classifies hormone replacement therapy (HRT) as a Tier 2 intervention, requiring careful risk-benefit analysis. In contrast, the European Medicines Agency (EMA) emphasizes non-hormonal first-line treatments like selective serotonin reuptake inhibitors (SSRIs). A 2024 Lancet review noted that 60% of UK NHS patients receive non-hormonal alternatives due to cost and safety concerns.
Global disparities in access persist. In low-income regions, 40% of women lack access to even basic hormonal therapies, per the World Health Organization (WHO). This underscores the need for region-specific guidelines, such as the WHO’s 2025 framework prioritizing affordable, low-risk interventions.
| Treatment | Efficacy (Symptom Reduction) | Common Side Effects | Regulatory Status |
|---|---|---|---|
| Hormone Replacement Therapy (HRT) | 70–80% reduction in hot flashes | Increased clot risk, breast cancer (long-term) | FDA-approved, EMA-restricted |
| SSRIs (e.g., gabapentin) | 40–60% reduction in vasomotor symptoms | Drowsiness, weight gain | Approved for off-label use |
| Lifestyle Modifications | 20–30% symptom reduction | Minimal | First-line recommendation |
Funding Transparency and Research Integrity
Much of the foundational research on perimenopause is funded by government agencies. The Women’s Health Initiative (WHI), a landmark 1990s study funded by the National Institutes of Health (NIH), revealed HRT’s risks, reshaping clinical practice. However, industry-funded trials, such as those evaluating bioidentical hormones, require scrutiny. A 2025